Grantee Research Project Results
2016 Progress Report: The Impact of PAH Exposure on Childhood Growth Trajectories and Visceral Adipose Tissue
EPA Grant Number: R836154C002Subproject: this is subproject number 002 , established and managed by the Center Director under grant R836154
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: The Columbia Center for Children’s Environmental Health
Center Director: Perera, Frederica P.
Title: The Impact of PAH Exposure on Childhood Growth Trajectories and Visceral Adipose Tissue
Investigators: Rundle, Andrew
Institution: Columbia University in the City of New York , Columbia University Mailman School of Public Health
Current Institution: Columbia University Mailman School of Public Health
EPA Project Officer: Callan, Richard
Project Period: September 1, 2015 through August 31, 2019 (Extended to August 31, 2020)
Project Period Covered by this Report: September 1, 2015 through August 31,2016
RFA: Children's Environmental Health and Disease Prevention Research Centers (2014) RFA Text | Recipients Lists
Research Category: Children's Health , Endocrine Disruptors , Human Health
Objective:
Aim 1
We will follow-up 350 CCCEH cohort children to ages 15 to 17 years old and measure abdominal subcutaneous adipose and visceral adipose tissue mass using MRI and fat mass index (FMI) using bio-impedance and anthropometric outcomes of height, weight, and waist circumference. Hypothesis 1: After adjustment for effects of postnatal and concurrent PAH exposures and other covariates, maternal exposure to higher concentrations of airborne PAH during pregnancy will be associated with greater child adiposity at ages 15 to 17 as assessed by abdominal visceral adipose tissue mass measured by MRI, FMI measured by bio-impedance, BMI z-score, and waist circumference.
Aim 2
We will characterize growth and adiposity trajectories from childhood into adolescence. Hypothesis 2: After adjustment for the effects of postnatal PAH exposures and other covariates, prenatal exposure to higher concentrations of airborne PAH will be associated with: BMI growth trajectories from age 5 to 17; and FMI growth trajectories from age 7 to 17.
Aim 3
We will apply cluster analysis to the anthropometric, eating behavior, physical activity and neurodevelopment and behavioral outcomes data collected at ages 15 to 17. Hypothesis 3: A cluster characterized by greater adiposity, higher hedonic eating behavior, sedentary pursuits, poorer neuropsychological function on measures of self-regulation and poorer behavioral and emotional function will be identified. We hypothesize that after adjustment for the effects of postnatal and concurrent PAH exposures and other covariates, membership in this cluster will be associated with maternal exposure to higher concentrations of airborne PAH during pregnancy.
Aim 4
All three projects in this P50 share a common cohort in which we will explore how prenatal PAH exposure dysregulates neurodevelopment (project 1) and physical growth (project 2) via its mediational effects on the brain (project 3). Hypothesis 4: Differences in brain structure and perfusion (by MRI at age 9-12 years) will mediate the effects of prenatal PAH exposures on neurodevelopment (assessed in Project 1), and obesity/adiposity (assessed in Project 2) at 15-17 years. This mediational hypothesis will be formally tested in Project 3.
Progress Summary:
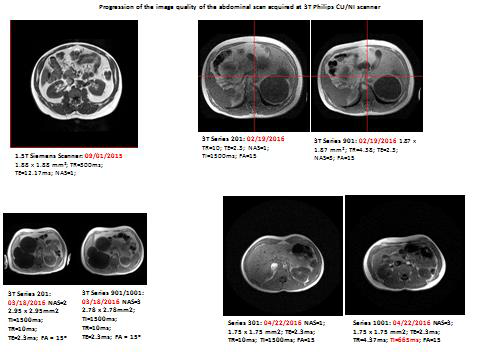
In our first year we have been working to establish our cohort follow-up protocol for assessing both the obesity (Project 2) and neurodevelopmental outcomes (Project 1). The P50 project protocol will collect data for both the obesity and neurodevelopment outcomes in a single integrated clinical visit and we have been establishing the process and flow of this clinical visit. A major activity has been to establish a MRI image capture protocol for the abdomen using the 3 Tesla (T) MRI machines available to us. At Columbia University (CU) abdominal scans for measuring body composition are conducted on a 1.5T MRI machine. However, MRI brain scans are typically performed using a 3T machine and the prior brain scans that were performed on our study subjects at age 9-11 years were acquired with a 3T machine. Maintaining the consistency of the scanner technology for the new age 16-18 brain scans with the technology we used for the age 9-11 brain scans is a priority for the Center’s research. Because of the P50 budget limitations and the logistics of MRI scheduling and the scheduling of, and the time required for, the participant visit, we cannot scan each participant on a 1.5T machine and then on a 3T machine. Thus we have worked to develop a MRI imaging protocol that can acquire brain and abdominal images on a single 3T scanner. The initial abdominal scan images acquired on the 3T scanner had poor contrast between adipose and non-adipose tissues and had areas of non-homogeneity in pixel intensity within adipose tissues which reduced the validity and reliability of image segmentation and adipose tissue area quantification. CCCEH investigators have been working to develop MRI pulse sequences with improved image quality and this work has involved researchers at the University of Southern California (USC), The Mailman School of Public Health, and the New York Obesity Research Center and consultations with a physicist at the New York Psychiatric Institute MRI Lab. Our progress in improving image quality is depicted in Figure 1.
Figure 1. Development of pulse sequences for abdominal image acquisition on the 3T scanner.
Figure 1 depicts representative images of the progress we have made in acquiring abdominal MRI images on the 3T MRI scanner. Our objective was to acquire images that have similar contrast-to-noise (CNR) and signal-to-noise ratio (SNR) as the image acquired on the 1.5T scanner (Top left image).
The images on the top right are from the CU 3T scanner and reflect the first step in protocol testing. The first of the pair of images presented has both poorer SNR and CNR than seen in the 1.5T scan. The second image with NAS=3, has adequate SNR, but both SNR and CNR were lower than that in the 1.5T data.
The images shown in the bottom left, had excellent SNR but at the expense of spatial resolution and they also had lower tissue contrast than the 1.5T data.
The set of images in the bottom right shows that the image with NAS=1 has excellent SNR and sufficient contrast as well for tissue segmentation. The image on the right has higher CNR and SNR but required NAS = 3. Although the last set of images has excellent SNR and CNR for accurate tissue segmentation, we are performing a final set of adjustments to the pulse sequence parameters to further increase the CNR.
Another MRI technology issue that we have been addressing is that MRI imaging of the abdomen requires that the subject holds his/her breath during the scans. However, many of our participants have asthma and breath maneuvers can be a challenge for this population. We have successfully reduced the duration of the breath hold maneuver by 50% compared to the original 3T MRI pulse sequence protocol. In addition we have established a secure protocol to incorporate the MRI images into the Data Core and to distribute MRI images to USC and CU teams.
We have selected and pilot tested questionnaire instruments to measure Hedonic eating, physical activity, food insecurity, weight cycling, weight loss surgery and use of supplemental nutrition programs. We have also pilot tested the combined Project 1 and 2 protocol, except the MRI image collection, on teens and mothers who are not part of the analytical cohort. We have worked closely with the Data Core to establish methods for direct data entry, for data security and for distributing data to study investigators. Methods development, training and pilot testing will help ensure robust and unbiased results. We have been contacting the cohort members and informing them of the upcoming project recruitment phase and the responses have been universally positive.
As a result of our efforts to maximize image quality described above specific to the abdominal scan protocol for the 3T scanner, the planned follow-up of the cohort and data collection has been delayed. We anticipate that we will begin collecting data from the study participants next month (June 2016) and we will accelerate our planned study subject recruitment and follow-up to make up for the delays we have experienced.
Data analysis of trajectories of child growth, adiposity and self-regulation through age 12 and physical activity at age 12 are underway and preliminary results were reported during the Children’s Centers webinar (Rundle, “Exposures to Polycyclic Aromatic Hydrocarbons and Childhood Growth Trajectories and Body Composition: Linkages to Disrupted Self-Regulatory Processes”, 1/13/16).
Future Activities:
In the coming reporting period we will continue follow-up of the cohort members as they reach ages 16-18 years and expect to complete 135 follow-up visits. The next reporting period will focus on intensive data collection from the study subjects. Clinical visits during which the study participants visit the Center’s clinical offices, complete the study instruments, and undergo anthropometric and fitness assessments and then have MRI scans at the CU Neurological Institute will be completed on weekdays and weekends. The recruitment and data collection will follow the approaches described in our grant application. We have been developing and testing rigorous data collection methods and the use of these approaches in the coming reporting period will ensure scientific rigor and robust results. The Data Core has established protocols for data acquisition, cleaning, storage and distribution to study investigators that will also ensure scientific rigor.
Our original proposal called for follow-up and data collection from 100 subjects in year one, two and three of the project and from 50 participants in year four. As a result of the challenges we experienced in developing a single MRI scan protocol that acquires both brain and abdominal images on a 3T MRI machine, the launch of participant recruitment and data collection was delayed. We anticipate beginning data collection in June of 2016 and thus expect to collect data from 30 participants in year 1 of the project. In year two and three we plan to complete recruitment at data collection for 135 participants to bring us back onto schedule by year four of the project.
Journal Articles:
No journal articles submitted with this report: View all 3 publications for this subprojectRelevant Websites:
Columbia Center for Children’s Environmental Health Exit
Progress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R836154 The Columbia Center for Children’s Environmental Health Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R836154C001 The Impact of PAH Exposure on Adolescent Neurodevelopment: Disruption of Self-Regulatory Processes
R836154C002 The Impact of PAH Exposure on Childhood Growth Trajectories and Visceral Adipose Tissue
R836154C003 An MRI Study of the Effects of Prenatal and Early Childhood PAH Exposure on Brain Maturation and Its Mediating Influences on Adverse Adolescent Outcomes
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
Main Center: R836154
13 publications for this center
10 journal articles for this center