Grantee Research Project Results
Final Report: Rapid Methods to Estimate Exposure to SVOCs in Indoor Environments
EPA Grant Number: R835606Title: Rapid Methods to Estimate Exposure to SVOCs in Indoor Environments
Investigators: Little, John C , Marr, Linsey C.
Institution: Virginia Tech
EPA Project Officer: Aja, Hayley
Project Period: July 1, 2014 through June 30, 2017 (Extended to June 30, 2018)
Project Amount: $900,000
RFA: New Methods in 21st Century Exposure Science (2013) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Expressed briefly, the major goals of the project were to:
- Develop novel method to measure the gas-phase SVOC concentration (y0) adjacent to the material surface in a consumer product;
- Measure occurrence of SVOCs in consumer products (C0) and use resulting data to establish the nature of equilibrium relationship between C0 and y0;
- Conduct single-source experiments to characterize emissions of various SVOCs from the representative sources;
- Develop novel method to determine surface/air partition coefficients (Ks) for selected SVOCs and six interior surfaces including airborne particles (Kp) and dust (Kdust), and compare results to available correlations between Ks, Kp, and Kdust and vapor pressure of SVOCs;
- Validate the single-source model using results from the chamber experiments for representative sources and the single-phase model using data collected from the literature; and
- Evaluate assumptions on which the rapid single-source and single-phase methods are based and characterize the uncertainty inherent in model predictions, especially for the low volatility SVOCs.
Summary/Accomplishments (Outputs/Outcomes):
Project Goal 1:
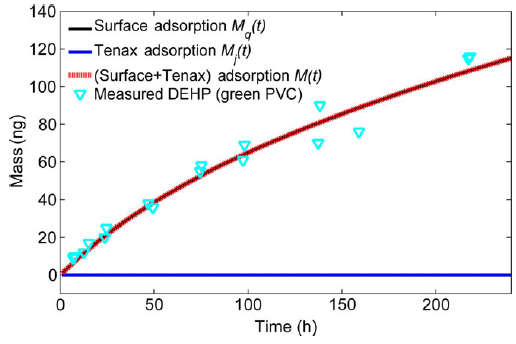
To easily measure key emission parameters for exposure assessment, a simple and rapid method based on passive sampling technique was developed to measure the gas-phase SVOC concentration (y0) immediately adjacent to the material surface in a consumer product (Figure 1). The y0 measurement method was shown to be applicable to chemicals with a wide range of vapor pressures. Detailed results can be found in Wu, Xie et al. (2015) .
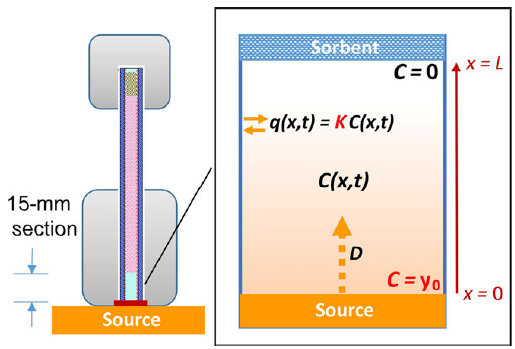
Figure 1: Left: Schematic representation of the diffusive sampler and diffusion model. Right: Measured DEHP in the tube as a function of time and best-fit model curves for green PVC flooring. The black line overlaps with the red line. (Wu, Xie et al. 2015)
Project Goal 2:
To reduce the required time and improve the accuracy of this method, a method which uses solid-phase microextraction (SPME) to measure y0 was established (Figure 2). The experimental time was shortened from several days (even several months) to about 1 day, with relative errors of less than 5%. The measured y0values agree well with the results obtained by independent methods (Cao, Zhang et al. 2017).
Figure 2: a) Schematic representation of the experimental setup and b) photographs of the setup. (Cao, Zhang et al. 2017)
The occurrence of phthalates (C0) in consumer products was evaluated by measuring the mass content C0 of several plasticizers in children's backpacks and toys (Xie, Wu et al. 2015). Additionally, surface wipes taken from the products were used to evaluate the mass transfer of plasticizers to clothing and human skin. The data also provided a knowledge base for the investigation of the relationship between C0 and y0.
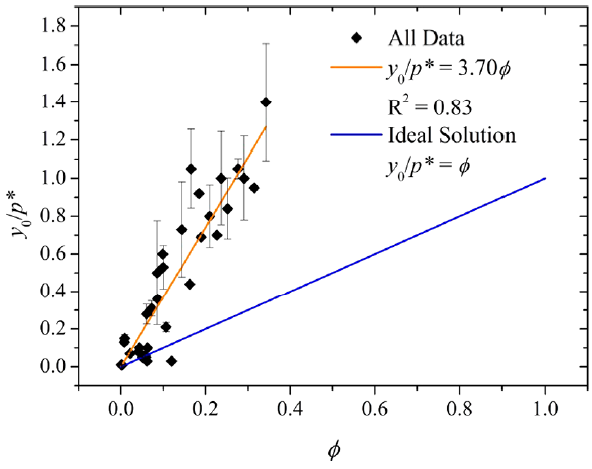
The diffusion chamber setup developed in Wu, Eichler et al. (2017) was adapted and used to measure y0 for the backpacks and toys investigated by Xie, Wu et al. (2015). We were able to establish an equilibrium relationship between C0 and y0 for phthalates and phthalate alternatives in PVC products based on a data set consisting of our own measurements and appropriate literature data (Eichler, Wu et al. 2018). The results show that the material/gas equilibrium relationship follows Henry's Law and that the Henry's Law constant consists of the respective pure liquid vapor pressure and an activity coefficient which accounts for the non-ideality of the mixture (Figure 3). The estimation of y0 based on this correlation significantly facilitates exposure and risk assessment.
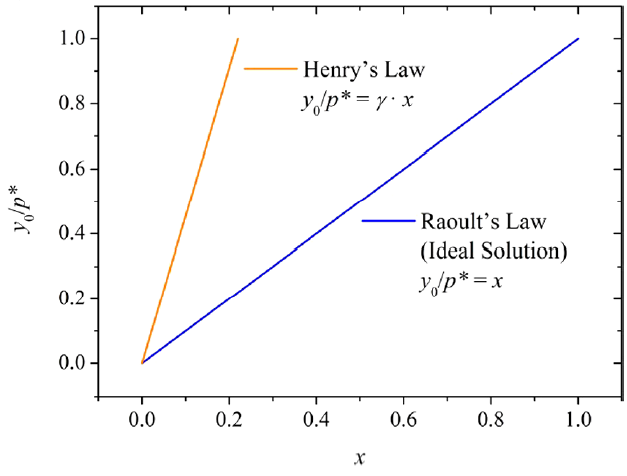
Figure 3: Left: Relationship between y0 normalized by the vapor pressure (p*) and the mole fraction x for ideal and non-ideal solutions. Right: Relationship between y0 (μg/m3) normalized by p* (μg/m3) and the volume fraction ϕ, including data measured in this study and additional data from literature and other measurements (Ntotal = 34, T = 25 °C). Errors were calculated if data were available. The standard error of the fitted slope is 0.18. (Eichler, Wu et al. 2018)
Project Goal 3:
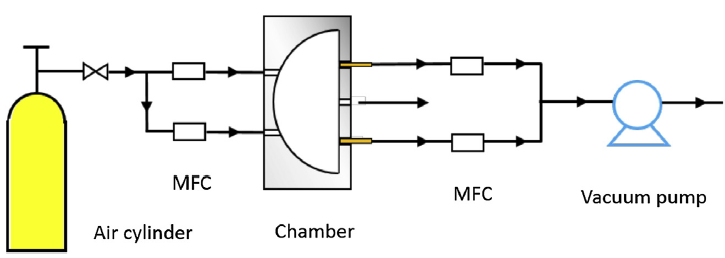
Our proposed emission model was successfully fitted to emission profiles obtained from using a reference method to characterize emissions of SVOCs in small chamber tests. An inter-laboratory study including six laboratories used the chamber test method to evaluate emission of DEHP from flooring material (Figure 4). The results showed good agreement, suggesting that the chamber test method can serve as reference method for emission tests. (Wu, Cox et al. 2016)
Figure 4: Schematic representation of the chamber test setup evaluated in an inter-laboratory study. (Wu, Cox et al. 2016)
Project Goal 4:
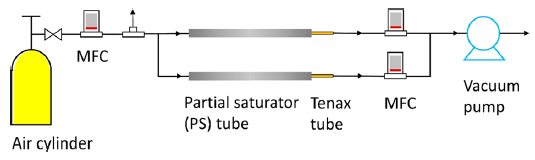
We also developed a novel method to measure the vapor pressure of phthalates and alternative plasticizers (Figure 5). Detailed results can be found in Wu, Eichler et al. (2016) . The measured vapor pressures ranged from about 10-2 to 10-7 Pa. Compared to the traditional gas saturation method, the model-based approach is advantageous in terms of both predictability and simplicity. The knowledge provides new insight into experimental design and a sound basis for further method development.
Figure 5: Schematic representation of the PS tube setup. (Wu, Eichler et al. 2016)
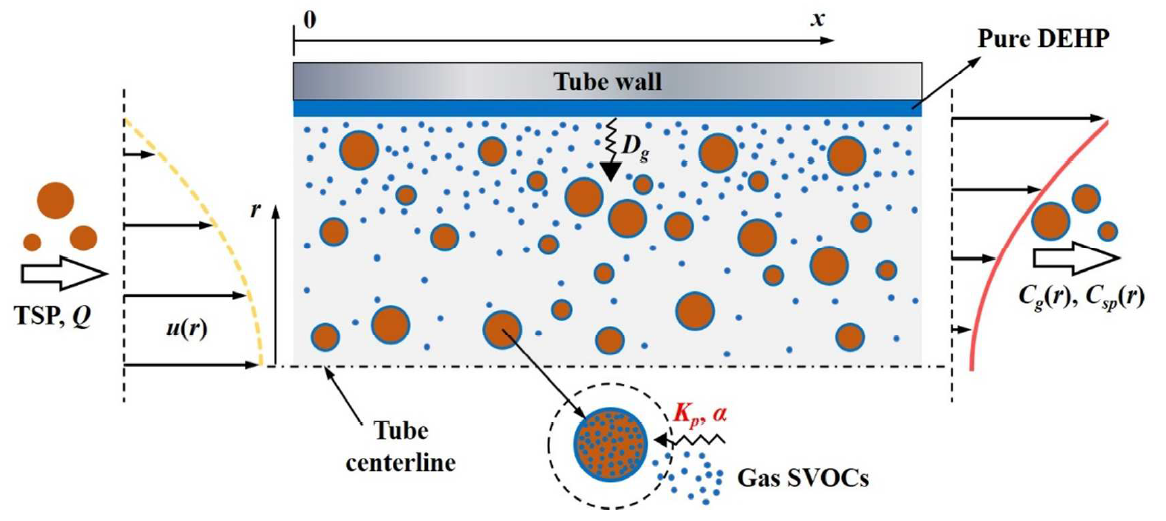
Another key element of our research regarding project goal 4 is the development of methods to characterize particle-enhanced mass transfer of SVOCs. A specially-designed tube chamber with laminar flow regime was used to investigate the particle-mediated mass transfer of SVOCs. We were able to determine the particle/air partition coefficient (Kp) and the vapor pressure of DEHP using this chamber experiment. The experimental quantification was combined with a computational fluid dynamics (CFD) model analysis of transport phenomena. The results showed that SVOC transport between emission source and air is significantly enhanced in the presence of particles (Wu, Eichler et al. 2018). We then further optimized the applicability and accuracy of the tube chamber method by developing a dynamic method combining the tube chamber with an SVOC mass transfer model (Figure 6). This approach does not only allow us to measure Kp, but also to measure the mass accommodation coefficient (α), another parameter important for characterizing the gas-particle interaction of SVOCs (Cao, Eichler et al. In preparation).
Figure 6: Schematic representation of SVOC mass transfer (in the presence of particles) in a tube chamber coated with a thin layer of pure SVOC liquid. The blue dots represent the SVOC molecules. Because the tube and the internal mass transfer process are symmetrical, only half the tube is depicted. (Cao, Eichler et al. 2018)
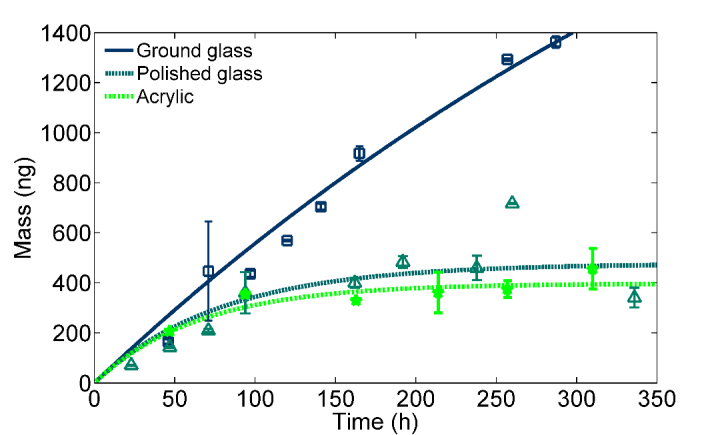
Sorption of SVOCs to interior surfaces greatly affects the fate and transport of SVOCs and thus human exposure. We developed an experimental method based on diffusion modeling to determine the surface/air partition coefficient Ks of di-2-ethylhexyl phthalate (DEHP) on typical impervious indoor surfaces. The diffusion model is similar to the model developed in (Wu, Liu et al. 2016). The results show that the surface roughness plays an important role in a surface's tendency to adsorb SVOCs (Figure 7). We also showed that the presence of an organic layer on the surfaces resulted in a nearly identical Ks that is close to reported values of the octanol/air partition coefficients. The knowledge provides a sound basis for simplification of estimating Ks of SVOCs for interior surfaces. (Wu, Eichler et al. 2017)
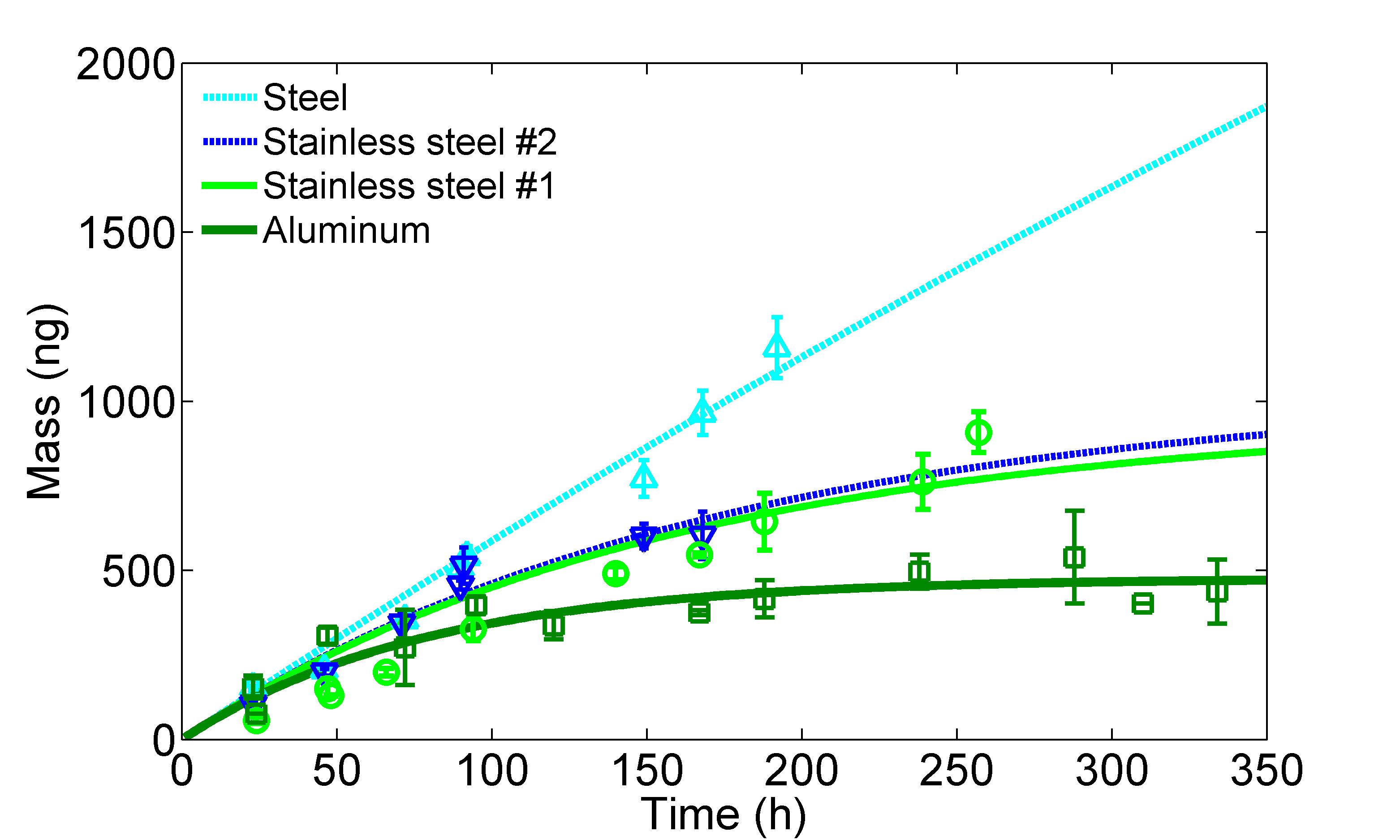
Figure 7: Measured DEHP mass accumulated on the impervious surface as function of time and best-fit model curves for (to the left) aluminum, stainless steel #1, stainless steel #2, and steel, and (to the right) ground glass, polished glass, and acrylic. (Wu, Eichler et al. 2017)
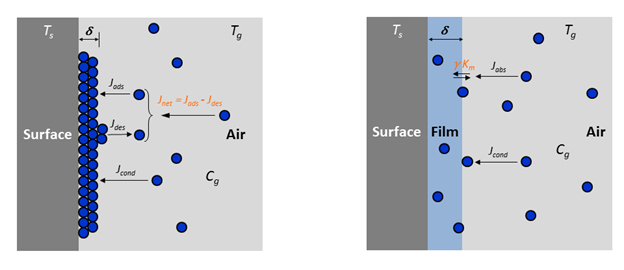
In Wu, Eichler et al. (2017), we were able to show the effect of a thin layer of organic "grime" on surfaces on Ks. To utilize this knowledge for modeling exposure to SVOCs, we developed a mass transfer model accounting for adsorption, condensation, and absorption of SVOCs on vertical indoor surfaces. In a manuscript currently under review, we present the model with an example, discuss gaps and uncertainties, and highlight possible aspects for further research (Figure 8) (Eichler, Cao et al. Under review).
Figure 8: Mechanisms of initial organic film formation and subsequent growth on a vertical surface: a) Stage 1 - initial formation governed by adsorption/desorption and condensation; b) Stage 2 - governed by absorption and condensation. (Eichler, Cao et al. Under review)
Our findings are novel and the developed methods provide a solid and consistent basis for further research. The presented results not only improve the general understanding of the fate and transport of SVOCs in the indoor environment, but also serve as key knowledge for modeling and prediction of human exposure to SVOCs. Exposure modeling is essential for the assessment of the risk that SVOC-product combinations might pose to human health.
Project Goals 5 & 6:
As part of the validation process of the single-source and the single-phase approaches, extensive literature research has been conducted. During the literature research it became apparent that a consistent review and analysis of existing chemical assessment policies and methods in place worldwide had not been conducted before. Therefore, we are working on closing this gap. The paper (Eichler, Hubal et al. In preparation) focuses on the policy landscape in the U.S., but also describes important regulations and associated research efforts in Europe, Canada, Australia, and Asia. It highlights the global shift in chemical assessment from hazard-driven, single-chemical assessments towards risk-based, high-throughput modeling approaches. Those approaches address the high complexity of human exposure to chemicals posed by the omnipresence of chemicals in consumer products and materials and thus in indoor environments. The relevance of and difficulties associated with the current policy landscape were illustrated by the example of phthalates.
References:
Cao, J., C. M. A. Eichler, Y. Wu and J. C. Little (In preparation). "Dynamic method to measure partition coefficient and mass accommodation coefficient for gas-particle interaction of phthalates." Environmental Science & Technology.
Cao, J., X. Zhang, J. C. Little and Y. Zhang (2017). "A SPME-based method for rapidly and accurately measuring the characteristic parameter for DEHP emitted from PVC floorings." Indoor Air 27: 417-426.
Eichler, C. M. A., J. Cao, G. Isaacman-VanWertz and J. C. Little (Under review). "Modeling the Formation and Growth of Organic Films on Indoor Surfaces " Indoor Air.
Eichler, C. M. A., E. Hubal and J. C. Little (In preparation). "Exposure to Chemicals in Consumer Products: The Policy Landscape." Environmental Science & Technology.
Eichler, C. M. A., Y. Wu, J. Cao, S. Shi and J. C. Little (2018). "Equilibrium relationship between SVOCs in PVC products and the air in contact with the product." Environmental Science & Technology 52(5): 2918-2925.
Wu, Y., S. S. Cox, Y. Xu, Y. Liang, D. Won, X. Liu, P. A. Clausen, L. Rosell, J. L. Benning, Y. Zhang and J. C. Little (2016). "A reference method for measuring emissions of SVOCs in small chambers." Building and Environment 95: 126-132.
Wu, Y., C. M. A. Eichler, J. Cao, J. L. Benning, A. Olson, S. Chen, C. Liu, E. P. Vejerano, L. C. Marr and J. C. Little (2018). "Particle/gas partitioning of phthalates to organic and inorganic airborne particles in the indoor environment." Environmental Science & Technology 52(6): 3583-3590.
Wu, Y., C. M. A. Eichler, S. Chen and J. C. Little (2016). "Simple Method To Measure the Vapor Pressure of Phthalates and Their Alternatives." Environmental Science & Technology 50: 10082-10088.
Wu, Y., C. M. A. Eichler, W. Leng, S. S. Cox, L. C. Marr and J. C. Little (2017). "Adsorption of Phthalates on Impervious Indoor Surfaces." Environmental Science & Technology 51(5): 2907-2913.
Wu, Y., Z. Liu, J. M. Herrera-Alsonso, E. Marand and J. C. Little (2016). "A new method to predict the effective diffusion coefficient of gases and vapors in polyurethane/clay nanocomposite membranes." Journal of Membrane Science 510: 201-208.
Wu, Y., M. Xie, S. S. Cox, L. C. Marr and J. C. Little (2015). "Simple method to measure the gas-phase SVOC concentration adjacent to a material surface." Indoor Air 26(6): 903-912.
Xie, M., Y. Wu, J. C. Little and L. C. Marr (2015). "Phthalates and alternative plasticizers and potential for contact exposure from children's backpacks and toys." Journal of Exposure Science & Environmental Epidemiology 26: 119-124.
Journal Articles on this Report : 4 Displayed | Download in RIS Format
| Other project views: | All 35 publications | 11 publications in selected types | All 11 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Cao J, Eichler C, WU Y, Little J. Dynamic method to measure partition coefficient and mass accommodation coefficient for gas-particle interaction of phthalates. Absolute Science and Technology 2019;1-14. |
R835606 (Final) |
Exit Exit |
|
|
Eichler C, Cao J, Isaacman-VanWertz G, Little J. Modeling the formation and growth of organic films on indoor surfaces. INDOOR AIR 2019;29(1):17-29 |
R835606 (Final) |
Exit |
|
|
Manuja A, Ritchie J, Buch K, Wu Y, Eichler CMA, Little JC, Marr LC. Total surface area in indoor environments. Environmental Science-Processes & Impacts 2019;21(8):1384-1392. |
R835606 (Final) |
Exit |
|
|
Wu Y, Eichler CMA, Cao J, Benning JL, Olson A, Chen S, Liu C, Vejerano E, Marr LC, Little JC. Particle/gas partitioning of phthalates to organic and inorganic airborne particles in the indoor environment. Environmental Science & Technology 2018; 52(6):3583-3590. |
R835606 (Final) |
Exit Exit |
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.