Grantee Research Project Results
2015 Progress Report: Novel Methods to Assess the Effects of Chemicals on Child Development
EPA Grant Number: R835434Center: Water Innovation Network for Sustainable Small Systems
Center Director: Reckhow, David A.
Title: Novel Methods to Assess the Effects of Chemicals on Child Development
Investigators: Schantz, Susan L.
Institution: University of Illinois Urbana-Champaign
EPA Project Officer: Hahn, Intaek
Project Period: June 1, 2013 through May 31, 2018 (Extended to May 31, 2019)
Project Period Covered by this Report: June 1, 2015 through May 31,2016
Project Amount: $3,962,727
RFA: Children's Environmental Health and Disease Prevention Research Centers (with NIEHS) (2012) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
Project 1: Joint Effects of Endocrine Disruptors, Diet, and BMI on Child Development
The major goals of this project are to: 1) assess sources of exposure to phthalates, bisphenol A (BPA), and other endocrine disrupting chemicals (EDCs) during the prenatal and adolescent periods; 2) examine the association of prenatal or adolescent exposure to phthalates, BPA, and other EDCs (and interactions among these exposures) with physical, behavioral and cognitive development in infants and adolescents; 3) assess the potential for a high fat diet (HFD)/obesity during two critical periods—prenatal or adolescent—to interact with chemical exposure to influence physical, behavioral, and cognitive development; and 4) investigate the association of prenatal exposure to phthalates, BPA, and other EDCs with markers of oxidative stress or inflammation in maternal and cord blood. In addition, we hypothesize that associations of exposure with these outcomes will vary by child sex.
Project 2: Endocrine Disrupting Chemicals, Diet, and Gonadal Toxicity
The proposed studies were designed to test the hypothesis that BPA, phthalate, and high fat diet exposure interact to increase oxidative stress in developing and adolescent gonads, leading to infertility, early reproductive senescence, and transgenerational effects on fertility in the offspring. To test this hypothesis, the following specific aims will be completed: 1) determine if high fat diet and BPA/phthalate/phthalate mixture exposure increase oxidative stress in the gonads of female and male mice, 2) determine if high fat diet and BPA/phthalate/phthalate mixture exposure destroy germ cells and cause epigenetic changes known to reduce germ cell quality in the gonads of female and male mice, and 3) determine if high fat diet and BPA/phthalate/phthalate mixture exposure cause infertility and early reproductive senescence in the first and subsequent generations in mice.
Project 3: Endocrine Disruptors and Diet: Effects on the Developing Cortex
Endocrine disruptors are ubiquitous in the environment, as are high fat diets, and both of these environmental factors could disturb the normal development of the nervous system during early development and during adolescence. This will be directly examined in a rodent model so that the cellular and molecular mechanisms of the effects can be elucidated. The environmental disruptors that are being separately investigated are bisphenol A (BPA) and a phthalate mixture in the presence of either a high or low fat diet. The disruptor and diet are imposed at two developmental time points—perinatally and during adolescence—in separate experiments. Endpoints include inflammation markers at the end of exposure and social and cognitive behaviors in adulthood.
Community Outreach and Translation Core (COTC)
Aim 1. To develop a strong Community Outreach and Translation Core (COTC) that is informed by the Community Advisory Board (CAB) and Dr. Susan Korrick, the Center’s Pediatric Health Specialist, and that works bi-directionally with the PIs of the Center. The CAB and PIs will work together in an iterative manner using feedback from stakeholders in developing dissemination materials.
Aim 2. Through stakeholder partnerships formed in the CAB, disseminate emerging knowledge about the effects of chemical exposure and high fat diets during the prenatal period, on the developing young child, and during adolescence.
Progress Summary:
Project 1: Joint Effects of Endocrine Disruptors, Diet, and BMI on Child Development
For the prospective birth cohort component of this project, the third reporting period has been devoted to five things: (1) continued recruitment of pregnant women into the study, (2) tracking of enrolled women throughout pregnancy, (3) cognitive assessments of infants born to study participants, (4) conducting 4-year cognitive assessments of children from our P20 pilot cohort, and (5) preparation of the dataset collected during the earlier P20 Formative Center stage of our research for statistical analysis and publication. As of August 15, 2016, 411 pregnant women had been enrolled in the study. Of those 411, 15 became ineligible during pregnancy and 27 withdrew from the study during pregnancy. Another 26 women either withdrew, could not be contacted, or became ineligible after their infants were born. Thus, as of August 15, 2016, there were 343 women actively enrolled in the new phase of the study. A total of 277 of study participants had given birth and 251 of those infants were still actively enrolled in the study. Earlier, we developed and implemented new state-of-the-art procedures for assessing cognition of infants during the first year of life. We currently are assessing cognitive functions including recognition memory, attention, and information processing speed at 1-5 weeks of age, 4-5 months of age, and 7-8 months of age in infants born to women in this study. All assessments are computer automated and the 4-5 and 7-8 month assessments make use of infrared eye tracking technology to track the infant’s looking behavior. We also recently began follow-up assessments of children recruited during the initial Formative Center phase of our research as they reached 46-48 months of age. The test battery includes assessments of working memory, attention, inhibition, cognitive flexibility, numbers and counting, and language development.
For the adolescent component of this project, our research to date has been devoted to completion of data collection. We have leveraged data from an ongoing prospective birth cohort, the New Bedford Cohort (NBC), in combination with work performed as part of our previous Children's Formative Center. For the current project, a key component of this leveraged work was collection of urine samples (for EDC exposure measurements) on 200 NBC adolescents. We successfully completed adolescent urine sample collection in 2014, and now have samples from 205 NBC participants in keeping with our project goal of 200 urine samples. Eighty-one percent of adolescents examined during the data collection provided at least one urine sample and 144 (70%) of these provided two urine samples, collected approximately 1 week apart. As part of the parent study assessments, we have completed prospectively collected neurobehavioral assessments and home assessments, as well as height, weight, diet, medical, demographic, lifestyle, and exposure information available on these 205 children, which are key data for this project's analyses. During the current reporting period, the urine samples were analyzed for ECDs at the Centers for Disease Control and Prevention (CDC) where levels of 11 common phthalate metabolites and 8 phenols (BPA, triclosan, butyl paraben, methyl paraben, propyl paraben, benzophenone-3, 2,4-dichlorophenol, and 2,5-dichlorophenol) were measured.
Project 2: Endocrine Disrupting Chemicals, Diet, and Gonadal Toxicity
In the current funding period, we tested the hypothesis that BPA exposure directly inhibits germ cell nest breakdown by inhibiting oxidative stress and/or apoptotic pathways. Our results indicate that low doses of BPA exposure significantly inhibit germ cell nest breakdown by inhibiting the expression of key ovarian apoptotic genes, but not by interfering with the oxidative stress pathway. This work was published (Zhou, et al., 2015; please see publication list).
In the current funding period, we also tested the hypothesis that prenatal exposure to BPA has transgenerational effects on female fertility. The results indicate that BPA exposure affects reproductive function in female mice and that some effects may be transgenerational in nature. This work was published (Ziv-Gal, et al., 2015; please see publication list).
We also expanded our work to examine the mechanisms by which BPA causes transgenerational effects on female fertility. Specifically, we tested the hypothesis that prenatal BPA exposure causes transgenerational effects on germ cell numbers and ovarian gene expression. Our results indicate that BPA exposure does not alter the percentage of germ cells or numbers of primordial follicles in F2 and F3 generations compared to control. Our results also indicate that in utero BPA exposure significantly alters the expression of antioxidant related genes in postnatal day 4 mouse ovaries, but the effects are different in the F1 and F3 generations. This work was published (Berger, et al., 2016; please see publication list).
We also focused on testing the hypothesis that prenatal exposure to BPA adversely impacts testicular development in mice. The results showed that serum testosterone levels of the mice prenatally exposed to BPA were significantly lower compared to control mice, but none of the BPA groups showed differences in fertility, birth rate, litter size, or female/male pup ratios compared to control.
In the previous funding period, we also conducted experiments on phthalates. Specifically, we tested the hypothesis that prenatal DEHP exposure affects female reproduction. The results indicate that prenatal Diethylhexyl-phthalate (DEHP) exposure alters F1 sex ratio, increases preantral follicle numbers, and causes some breeding abnormalities compared to control. This work was published (Niermann, et al., 2015).
We expanded the work above by testing the hypothesis that prenatal exposure to DEHP impairs the fertility of female mice in the F1, F2, and F3 generations. The data suggest that prenatal DEHP exposure may have transgenerational effects on fertility outcomes such as percentage of time spent in estrus and diestrus, sex ratio, litter size, and occurrence of dead pups.
Further, we tested the hypothesis that prenatal DEHP exposure affects gonadal development in male offspring. The results indicate that the highest dose (750 mg/kg/day) significantly reduced fertility, with a maximum defect shown at 19 months of age. The results also suggest that the reduced fertility is due to defects in sperm and low testosterone levels.
In addition, we conducted experiments to test the hypothesis that exposure to an environmentally relevant phthalate mixture alters folliculogenesis and induces oxidative stress in the ovary. The results indicate that the phthalate mixture significantly decreases the numbers of germ cells, primordial follicles, and primary follicles, and it interferes with the expression of key antioxidant enzymes compared to control.
Finally, we tested the hypothesis that prenatal phthalate mixture exposure affects gonadal development in male offspring. At 4 months, phthalate-mixture treated males exhibited a normal range of fertility without any significant difference in their litter sizes or female-to-male ratio. This is consistent with our results from BPA and DEHP experiments in which we saw no impact on fertility in young adult life, but did observe defects later in adulthood (>12 months). Therefore, we will repeat the fertility test at the ages 12 months and older during the next funding period.
Project 3: Endocrine Disruptors and Diet: Effects on the Developing Cortex
In the previous P20 grant, we found that pre- plus post-natal exposure to a 400 μg/kg dose of BPA resulted in more neurons and glia in the adult prefrontal cortex of male, but not female, rats. The intriguing parallel with findings from autistic human males, has led us to start our investigations with perinatal BPA and the high fat diet of Aim 1. The design is large: 2 diet groups (high and low fat) x 3 doses of BPA (0, 40, and 400 mg/kg/day) x 2 sexes = 12 groups. Managing this study design has been accomplished with consecutive cohorts of matings that produce litters for each group so that behavioral analysis and sacrifice for neuroanatomical histology are not unwieldy. We recently have finished the behavioral tasks with these animals and are starting the neuroanatomical analysis.
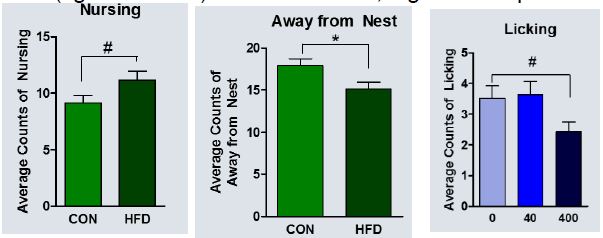
Maternal behavior. Dams fed the high fat diet nursed their pups more and spent less time away from the nest (Figure 1). In other words, high fat diet promoted maternal behavior. On the other hand, the 400 μg/kg dose of BPA resulted in less maternal licking of pups. A manuscript currently is in preparation with these results.
Figure 1: High fat diet promoted maternal behavior
Social play. We reported in last year's report that early BPA exposure (both doses) resulted in less social play when adolescents. This result continues with the addition of the last two cohorts of animals.
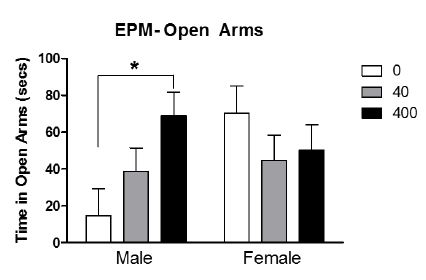
Elevated plus maze. We found effects of BPA in the groups given the control diets, not high fat. There was an interaction between sex by BPA exposure such that males exposed to the highest dose of BPA spent more time in the open arms (i.e., were less fearful) than controls. Females were not affected (Figure 2).
Figure 2: Interaction between sex by BPA exposure
Other behavioral tasks. No group differences were found in performance in the social recognition or intra/extra dimensional shift tasks in the adults exposed to BPA and high fat diet perinatally.
Gene expression. In collaboration with Dr. Pan’s laboratory, we have examined gene expression of 15 genes within the medial prefrontal cortex at P10. Two genes had significant interactions between BPA exposure and diet. Also, there were two genes that were affected by BPA in both diet groups and both sexes: ERα expression was increased by both doses of BPA relative to controls, as was the antioxidant SOD1.
Phthalates
A similar design currently is being run to investigate the effects of the phthalates during perinatal development. Dams consume either 0, 100, or 1000 μg/kg/day of a mixture of phthalates that is based on the ratio of compounds found in maternal urine in Project 1 (the Champaign/Urbana mixture). These doses also are crossed with low or high fat diets. Again, like the BPA study, this results in 12 groups that need to be bred and later run in cohorts. At this time, we are testing the last cohort of rats on the array of behavioral tasks. We have not yet decoded the results from the behavioral tasks. We do know that males from phthalate exposed groups had lower body weight at P10 (end of exposure) and P25 (post-weaning).
Transgenerational phthalate effects
In collaboration with the Flaws lab, we are looking at transgenerational effects of several doses (20 μg, 200 μg, 500 mg, and 750 mg/kg) of the phthalate DEHP on cognitive and anxiety-like behaviors in mice (CD-1). In a cohort that is three generations removed from maternal treatment (F3 generation), we have found a significant increase in time spent in the open arms of an elevated plus maze in phthalate-treated adult females. There were no significant effects on activity level in an open field maze, or on long-term memory in an object recognition task. Studies involving males currently are being analyzed, and additionally, gene expression will be analyzed in several brain areas relevant to the tasks.
Community Outreach and Translation Core (COTC)
Community Advisory Board (CAB). The CAB for the Illinois COTC includes leaders in early care and education, parenting, child advocacy, and public health in Illinois. In addition, a national leader in online Extension education is on the Board. The CAB was instrumental in reviewing materials for the Spring into Action workshop, the redesign of our messaging campaign for child care providers, and provided input on developing a webinar for child care providers. Interviews with CAB members following the annual meeting suggested that they are pleased with the overall involvement with the board; they encouraged the COTC to develop a dissemination plan and offered suggestions for adding new members to the board that would represent a community-based perspective. These names are being considered for the next meeting.
Evaluation of Spring into Action Conference. On April 21, 2015, the Illinois COTC co-hosted a 2-day conference in Springfield, Illinois to address environmental health in child care. The co-host was Illinois Action for Children, the child care licensing agency for Chicago and Cook County and preeminent statewide advocacy organization to increase access to quality child care among lower income families. The keynote speaker for the conference was Dr. Susan Buchanan, MD, Illinois COTC CAB member and Director, Region 5 PEHSU. Her talk was followed by breakout sessions, in which child care providers participated in semi-structured discussions about current resources and beliefs on protecting children’s environmental health in child care settings. Additionally, there was an opportunity for conference attendees to participate in an interactive learning activity that focused on strategies to reduce exposures based on the Eco-Friendly Child Care checklist. The keynote was extremely well received and generated considerable interest. Ninety percent of those surveyed (N = 100) agreed or strongly agreed that Dr. Buchanan helped them better understand that environmental exposures can affect children’s development and health. Ninety-three percent of those surveyed agreed or strongly agreed that Dr. Buchanan helped them learn about contaminants in the child care environment that can effect children’s development and health.
In the follow up workshops, we learned that the base knowledge of child care providers, advocates, and state administrators about children’s environmental health is very low. Therefore, we have restructured our messaging campaign to provide more basic overviews of what constitutes environmental health and provide examples of good practices rather than focus solely on endocrine disruptors. The child care providers also wanted practical steps that they could implement in their homes and child care sites.
Midwest Children’s Environmental Health Symposium. We organized a collaborative symposium with University of Michigan’s Children’s Environmental Health Center to present research, translation, and community outreach on how environmental exposures affect children’s health and development. The symposium was attended by approximately 40 faculty, graduate students, and staff from the University of Illinois and Champaign-Urbana community. The attendees represented a variety of disciplines including comparative biosciences, human development and family studies, medicine, nutritional sciences, neurosciences, and psychology. Dr. Fiese was interviewed by the local National Public Radio (NPR) station about the event, which was aired across the state and a network of agricultural programming across the country.
APHA Symposia. We participated in the symposia along with three other Children Environmental Health Center COTCs. The presentation was titled “Raising Awareness of the Role of Childcare Providers in Insuring Children’s Environmental Health.” It described preliminary results from a survey of 79 child care providers across the state. We also presented evaluation results from our Spring into Action Conference and impressions from child care providers as to the importance of knowledge gain about general understanding of the broad issues relevant to children’s environmental health and healthy growth and development.
Future Activities:
Project 1: Joint Effects of Endocrine Disruptors, Diet, and BMI on Child Development
Over the coming year, we will continue to recruit pregnant women into the cohort, and will assess infants at 1-5 weeks, 4-5 months, and 7-8 months. We also will begin 27-29 month follow ups of the children in September 2016. We will continue our assessments of 46-48 month-old children recruited during the earlier Formative phase (to be completed in October 2016), we will analyze data collected during the Formative phase, and submit abstracts for presentation and papers for publication. EDC data now are available on the adolescent cohort; thus, the focus in the coming year will be on data analyses to address study aims, as well as preparation of abstracts for presentation at scholarly meetings and manuscripts for publication.
Project 2: Endocrine Disrupting Chemicals, Diet, and Gonadal Toxicity
During the next funding period, we plan to complete additional experiments on the transgenerational effects of DEHP on female reproduction. Specifically, we plan to conduct detailed histological evaluations of follicle numbers in ovaries collected from the F1, F2, and F3 generations. We also plan to examine sex steroid hormone levels as well as gonadotropin levels in the females from the F1, F2, and F3 generations. In addition, we plan to expand our work on the effects of the phthalate mixture on female reproduction. Specifically, we will examine the effects of prenatal phthalate mixture on female fertility in the F1, F2, and F3 generations.
Lastly, we plan to complete experiments on the transgenerational effects of BPA phthalates on male reproduction. Specifically, we will measure serum steroid and pituitary hormone levels, perform semen analyses in aging mice, investigate epigenetic modification by BPA and DEHP with tissue samples collected at PND 20 and 60, and conduct histological examinations of tissues collected from aged mice.
Project 3: Endocrine Disruptors and Diet: Effects on the Developing Cortex
Activities for the coming year include:
BPA
1. Submit two papers on the behavioral data from the perinatal BPA exposure groups.
2. Finish the quantification of the number of neurons and glia in the medial prefrontal cortex.
3. Start the quantification of synapses in this tissue.
4. Perform the genetic expression (qPCR) and epigenetic analysis on the P90 tissue with Dr. Pan’s laboratory.
Phthalates
1. Finish the behavioral analysis and submit the results in one to two papers.
2. Perform the genetic and epigenetic analysis on both P10 and P90 tissue with Dr. Pan’s laboratory.
3. Start the neuroanatomical quantification of the number of neurons, glia, and synapses in the medial prefrontal cortex in the tissue.
Transgenerational effects
1. Perform qPCR on tissue from the F3 phthalate DEHP exposed animals.
Community Outreach and Translation Core (COTC)
1. Continue CAB meetings—The CAB continues to be engaged in the COTC and remains eager to support the project. The CAB members have welcomed opportunities to broaden their own education and knowledge on children’s environmental health. They will work with us to identify other key collaborators, professional conferences to present at, as well as appropriate targets for our messaging and outreach.
2. Revise Child Care Survey—Revise the survey to focus on broad-based knowledge of environmental health and distribute the survey statewide with a focus on home-based child care providers.
3. Develop Dissemination Materials for Child Care Providers—Using the findings from the formative research, we will develop outreach materials for child care providers that we will disseminate through our CAB partnerships and represented organizations. All materials also will be available on the project website. We will work with our Communication Specialist to develop a dissemination plan.
4. Webinars—We are in the process of scheduling webinars focused on children’s environmental health with the Illinois Governor’s Office of Early Childhood Development and with eExtension. These webinars will feature Great Lakes PEHSU Director Dr. Susan Buchanan and Dr. Barbara Fiese.
5. Continue to Collaborate with Other COTCs and CEHN—We will continue to participate in monthly CEHC COTC calls. We are working to develop a stronger partnership with the Michigan COTC. We also will continue our nascent partnership with the CEHN Eco-Friendly Child Care.
6. Develop Additional PSA Video—We will develop and produce an additional video that will be distributed as PSAs as well as made available on a variety of platforms (e.g., website, YouTube).
7. Just in Time Parenting—We will develop materials and messaging and disseminate them through Just in Time Parenting (represented on our CAB).
8. Statewide Working Group on Children’s Environmental Health—We will initiate the formation of this group at a time advised by our CAB.
References:
Ziv-Gal A, Wang W, Zhou C, Flaws JA. The effects of in utero bisphenol A exposure on reproductive capacity in several generations of mice. Toxicology and Applied Pharmacology 2015;284:354-362. PMID: 25771130.
Niermann S, Rattan S, Brehm E, Flaws JA. Prenatal exposure to di-(2-ethylhexyl) phthalate (DEHP) affects reproductive outcomes in female mice. Reproductive Toxicology 2015;53:23-32. PMID: 25765777.
Zhou C, Wang W, Peretz J, Flaws JA. Bisphenol A exposure inhibits germ cell nest breakdown by reducing apoptosis in cultured neonatal mouse ovaries. Reproductive Toxicology 2015;57:87-99. PMID: 26049153.
Berger A, Ziv-Gal A, Cudiamat J, Wang W, Zhou C, Flaws JA. The effects of in utero bisphenol A exposure on the ovaries in multiple generations of mice. Reproductive Toxicology 2016;60:39-52. PMID 26746108.
Journal Articles: 31 Displayed | Download in RIS Format
| Other center views: | All 68 publications | 31 publications in selected types | All 31 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Barakat R, Lin P-CP, Rattan S, Brehm ES, Canisso IF, Abosalum ME, Flaws JA, Hess R, Ko C. Prenatal exposure to DEHP induces premature reproductive senescence in male mice. Toxicological Sciences 2017;156(1):96-108. |
R835434 (2016) R835434 (2017) |
Exit Exit Exit |
|
|
Berger A, Ziv-Gal A, Cudiamat J, Wang W, Zhou C, Flaws JA. The effects of in utero bisphenol A exposure on the ovaries in multiple generations of mice. Reproductive Toxicology 2016;60:39-52. |
R835434 (2015) |
Exit Exit Exit |
|
|
Brehm E, Rattan S, Gao L, Flaws JA. Prenatal exposure to di(2-ethylhexyl) phthalate causes long-term transgenerational effects on female reproduction in mice. Endocrinology 2018;159(2):795-809. |
R835434 (2017) |
Exit Exit |
|
|
Drobna Z, Henriksen AD, Wolstenholme JT, Montiel C, Lambeth PS, Shang S, Harris EP, Zhou C, Flaws JA, Adli M, Rissman EF. Transgenerational effects of bisphenol A on gene expression and DNA methylation of imprinted genes in brain. Endocrinology 2018;159(1):132-144. |
R835434 (2017) |
Exit Exit |
|
|
Eckstrum KS, Edwards W, Banerjee A, Wang W, Flaws JA, Katzenellenbogen JA, Kim SH, Raetzman LT. Effects of exposure to the endocrine-disrupting chemical bisphenol A during critical windows of murine pituitary development. Endocrinology 2018;159(1):119-131. |
R835434 (2017) |
Exit Exit |
|
|
Gal A, Lin P-C, Barger AM, MacNeill AL, Ko C. Vaginal fold histology reduces the variability introduced by vaginal exfoliative cytology in the classification of mouse estrous cycle stages. Toxicologic Pathology 2014;42(8):1212-1220. |
R835434 (2013) |
Exit Exit |
|
|
Kiester B, Sloane S, Fujimoto E, Fiese B, Su L. What Do Childcare Providers Know about Environmental Influences on Children's Health? Implications for Environmental Health Literacy Efforts. INTERNATIONAL JOURNAL OF ENVIRONMENTAL RESEARCH AND PUBLIC HEALTH 2021;18(10):5489. |
R835434 (Final) |
Exit Exit |
|
|
Kougias DG, Cortes LR, Moody L, Rhoads S, Pan Y-X, Juraska JM. Effects of perinatal exposure to phthalates and a high-fat diet on maternal behavior and pup development and social play. Endocrinology 2018;159(2):1088-1105. |
R835434 (2017) |
Exit Exit |
|
|
Kougias DG, Sellinger EP, Willing J, Juraska JM. Perinatal exposure to an environmentally relevant mixture of phthalates results in a lower number of neurons and synapses in the medial prefrontal cortex and decreased cognitive flexibility in adult male and female rats. Journal of Neuroscience 2018;38(31):6864-6872. |
R835434 (2017) |
Exit Exit |
|
|
Li Q, Davila J, Kannan A, Flaws JA, Bagchi MK, Bagchi IC. Chronic exposure to bisphenol A affects uterine function during early pregnancy in mice. Endocrinology 2016;157(5):1764-1774. |
R835434 (2016) |
Exit Exit |
|
|
Li Q, Lawrence CR, Nowak RA, Flaws JA, Bagchi MK, Bagchi IC. Bisphenol A and phthalates modulate peritoneal macrophage function in female mice involving SYMD2-H3K36 dimethylation. Endocrinology 2018;159(5):2216-2228. |
R835434 (2017) |
Exit Exit |
|
|
Niermann S, Rattan S, Brehm E, Flaws JA. Prenatal exposure to di-(2-ethylhexyl) phthalate (DEHP) affects reproductive outcomes in female mice. Reproductive Toxicology 2015;53:23-32. |
R835434 (2014) R835434 (2015) |
Exit Exit Exit |
|
|
Oakley OR, Kim KJ, Lin PC, Barakat R, Cacioppo JA, Li Z, Whitaker A, Chung KC, Mei W, Ko C. Estradiol synthesis in gut-associated lymphoid tissue: leukocyte regulation by a sexually monomorphic system. Endocrinology 2016;157(12):4579-4587. |
R835434 (2016) |
Exit Exit |
|
|
Peretz J, Vrooman L, Ricke WA, Hunt PA, Ehrlich S, Hauser R, Padmanabhan V, Taylor HS, Swan SH, VandeVoort CA, Flaws JA. Bisphenol A and reproductive health: update of experimental and human evidence, 2007-2013. Environmental Health Perspectives 2014;122(8):775-786. |
R835434 (2013) R835434 (2014) R834593C001 (Final) R835436 (2014) R835436 (2015) R835436 (2017) |
|
|
|
Rattan S, Zhou C, Chiang C, Mahalingam S, Brehm E, Flaws JA. Exposure to endocrine disrupting chemicals during adulthood: consequences for female fertility. Journal of Endocrinology 2017;233(3):R109-R129. |
R835434 (2017) |
Exit Exit Exit |
|
|
Rattan S, Brehm E, Gao L, Niermann S, Flaws JA. Prenatal exposure to di(2-ethylhexyl) phthalate disrupts ovarian function in a transgenerational manner in female mice. Biology of Reproduction 2018;98(1):130-145. |
R835434 (2017) |
Exit Exit |
|
|
Rattan S, Brehm E, Gao L, Flaws JA. Di(2-ethylhexyl) phthalate exposure during prenatal development causes adverse transgenerational effects on female fertility in mice. Toxicological Sciences 2018;163(2):420-429. |
R835434 (2017) |
Exit Exit |
|
|
Richardson KA, Hannon PR, Johnson-Walker YJ, Myint MS, Flaws JA, Nowak RA. Di(2-ethylhexyl) phthalate (DEHP) alters proliferation and uterine gland numbers in the uteri of adult exposed mice. Reproductive Toxicology 2018;77:70-79. |
R835434 (2017) |
Exit Exit Exit |
|
|
Sellinger E, Kougias D, Drzewiecki C, Juraska J. Behavioral effects in adult rats exposed to low doses of a phthalate mixture during the perinatal or adolescent period. NEUROTOXICOLOGY AND TERATOLOGY 2020;79(106886). |
R835434 (Final) |
Exit Exit |
|
|
Wang W, Hafner KS, Flaws JA. In utero bisphenol A exposure disrupts germ cell nest breakdown and reduces fertility with age in the mouse. Toxicology and Applied Pharmacology 2014;276(2):157-164. |
R835434 (2013) R835434 (2014) |
Exit Exit Exit |
|
|
Wise LM, Sadowski RN, Kim T, Willing J, Juraska JM. Long-term effects of adolescent exposure to Bisphenol A on neuron and glia number in the rat prefrontal cortex: differences between the sexes and cell type. Neurotoxicology 2016;53:186-192. |
R835434 (2014) R835434 (2015) |
Exit Exit Exit |
|
|
Wise LM, Hernández-Saavedra D, Boas SM, Pan YX, Juraska JM. Perinatal high-fat diet and bisphenol A:effects on behavior and gene expression in the medial prefrontal cortex. Developmental Neuroscience 2018;21:1-16. |
R835434 (Final) |
Exit Exit |
|
|
Yazdy MM, Coull BA, Gardiner JC, Aguiar A, Calafat AM, Ye X, Schantz SL, Korrick SA. A possible approach to improving the reproducibility of urinary concentrations of phthalate metabolites and phenols during pregnancy. Journal of Exposure Science & Environmental Epidemiology 2018;28(5):448-460. |
R835434 (2017) R835434 (Final) |
Exit Exit |
|
|
Zhou C, Wang W, Peretz J, Flaws JA. Bisphenol A exposure inhibits germ cell nest breakdown by reducing apoptosis in cultured neonatal mouse ovaries. Reproductive Toxicology 2015;57:87-99. |
R835434 (2014) R835434 (2015) |
Exit Exit Exit |
|
|
Zhou C, Gao L, Flaws JA. Exposure to an environmentally relevant phthalate mixture causes transgenerational effects on female reproduction in mice. Endocrinology 2017;158(6):1739-1754. |
R835434 (2016) |
Exit Exit |
|
|
Zhou C, Flaws JA. Effects of an environmentally relevant phthalate mixture on cultured mouse antral follicles. Toxicological Sciences 2017;156(1):217-229. |
R835434 (2016) |
Exit Exit |
|
|
Zhou C, Gao L, Flaws JA. Prenatal exposure to an environmentally relevant phthalate mixture disrupts reproduction in the F1 female mice. Toxicology and Applied Pharmacology 2017;318:49-57. |
R835434 (2016) |
Exit Exit Exit |
|
|
Ziv-Gal A, Wang W, Zhou C, Flaws JA. The effects of in utero bisphenol A exposure on reproductive capacity in several generations of mice. Toxicology and Applied Pharmacology 2015;284(3):354-362. |
R835434 (2014) R835434 (2015) |
Exit Exit Exit |
|
|
Ziv-Gal A, Flaws JA. Evidence for bisphenol A-induced female infertility: a review (2007-2016). Fertility and Sterility 2016;106(4):827-856. |
R835434 (2016) |
Exit Exit Exit |
|
|
Strakovsky RS, Schantz SL. Impacts of bisphenol A (BPA) and phthalate exposures on epigenetic outcomes in the human placenta. Environmental Epigenetics 2018;4(3):dvy022 (18 pp.). |
R835434 (2017) |
Exit Exit Exit |
|
|
Strakovsky RS, Schantz SL. Using experimental models to assess effects of bisphenol A (BPA) and phthalates on the placenta:challenges and perspectives. Toxicological Sciences 2018 |
R835434 (2017) |
Exit |
Supplemental Keywords:
adolescent health, bisphenol A, BPA, children's health, cognition, endocrine disruptors, EDCs, epidemiology, growth, neurobehavior, phenols, phthalates, prenatal exposure, ovary, testis, gonads, oxidative stress, high fat diet, diet, social behavior, maternal behavior, genetic expressionRelevant Websites:
Children's Environmental Health Research Center at Illinois Exit
Children's Environmental Health Center's Community Outreach and Translation Core Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2017 Progress Report
- 2016 Progress Report
- 2014 Progress Report
- 2013 Progress Report
- Original Abstract
31 journal articles for this center