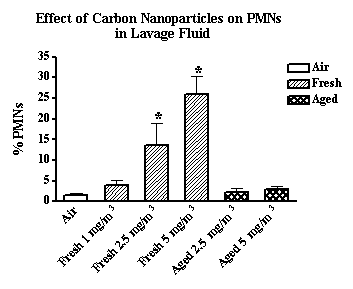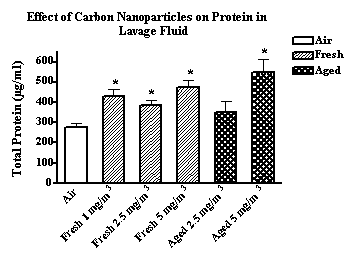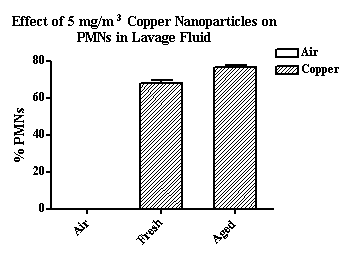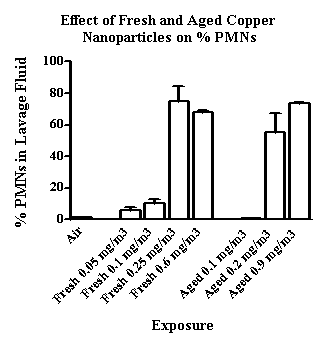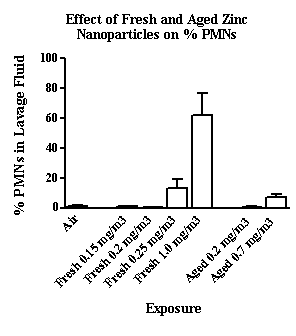Grantee Research Project Results
Final Report: Role of Particle Agglomeration in Nanoparticle Toxicity
EPA Grant Number: R832528Title: Role of Particle Agglomeration in Nanoparticle Toxicity
Investigators: Gordon, Terry , Chen, Lung Chi , Cohen, Beverly S.
Institution: New York University Medical Center Allied Hlth Ed
EPA Project Officer: Hahn, Intaek
Project Period: October 1, 2005 through September 30, 2008
Project Amount: $375,000
RFA: Exploratory Research: Nanotechnology Research Grants Investigating Environmental and Human Health Effects of Manufactured Nanomaterials: A Joint Research Solicitation - EPA, NSF, NIOSH (2005) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
The objective of this study was to determine whether agglomeration of nanoparticles affected their physicochemcial characteristics and therefore their toxicity. We hypothesized that that there would be a difference in the toxicity of freshly generated vs. aged nanoparticles. In testing this hypothesis, we: 1) measured the agglomeration rate of several types of nanoparticles; 2) identified whether agglomeration and associated toxicity was affected by differing exposure conditions including temperature, humidity, and particle charge; and 3) compared the toxicity of singlet vs. agglomerated particles in mice exposed via the inhalation route.
Summary/Accomplishments (Outputs/Outcomes):
Aim I: To measure the agglomeration rate of carbon nanoparticles under dynamic exposure conditions. Carbon and metal nanoparticles will be generated with an arc furnace and the agglomeration rate will be established for each particle type.
Aim I was completed. Carbon nanoparticles freshly generated by the arc method were approximately 10 to 15 nm in count median diameter (CMD) whereas aging of these particles resulted in a count median diameter that varied between 150 to 250 nm in different experiments. Not surprisingly, nanoparticles which were freshly generated from pure zinc or copper electrodes had a CMD similar to carbon nanoparticles.
Aim II. To identify the extent to which agglomeration is affected by altering physical exposure conditions such as humidity and particle charge.
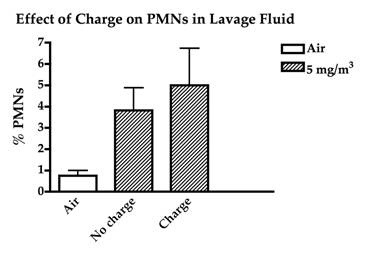
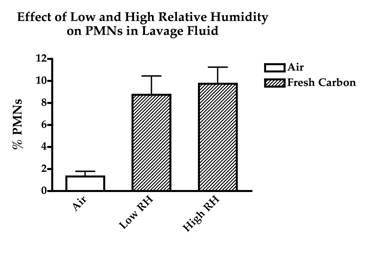
To examine the effect of particle charge on nanoparticle toxicity, carbon nanoparticles were generated at 5 mg/m3 and mice were exposed to the non-modified nanoparticles or nanoparticles which were passed through a Americium particle neutralizer. The particle neutralizer had no effect on particle toxicity as seen by equal inflammation which was observed in mice exposed to the non-modified and the neutralized nanoparticles (Figure 1). To examine whether relative humidity affected either particle agglomeration or toxicity, additional groups of animals were exposed to fresh carbon nanoparticles that were delivered to the nose-only exposure chamber under conditions of either 15% or 85% relative humidity. Neither particle size nor toxicity was different between these 2 extremes of exposure chamber humidity (Figure 2).
Figure 1. The effect of charged and neutral carbon nanoparticles on lung inflammation as assessed by the influx of inflammatory cells (polymorphonuclear neutrophils – PMNs) in lung lavage fluid.
Figure 2. The effect of high (85%) and low (15%) relative humidity on lung inflammation produced by inhalation of freshly generated carbon nanoparticles as assessed by the influx of PMNs in lung lavage fluid.
Aim III. To compare the toxicity of ‘singlet’ vs. agglomerated particles in mice exposed via the inhalation route. Acute lung injury and inflammation will be studied in mice exposed to fresh (predominantly ‘singlet’) or aged (predominantly agglomerated) carbon nanoparticles. Conditions studied in Aims I and II will be varied to examine the influence of particle composition, humidity, and charge on carbon nanoparticle agglomeration and toxicity.
Aims I and II demonstrated that neither charge nor relative humidity extremes affected the toxicity of freshly generated carbon nanoparticles. Therefore, the studies proposed in Aim III were conducted using the particle charge state which naturally occurred in particles generated by the arc method and a relative humidity of 30-50%.
Inbred BALB/c mice were exposed to fresh and aged carbon, zinc, and copper nanoparticles generated by the arc method. Freshly generated carbon nanoparticles (11-60 nm CMD range) produced a significantly greater increase in neutrophils in lavage fluid compared to carbon nanoparticles aged approximately 3 minutes (190-250 nm) (see 2.5 and 5 mg/m3 data in Figure 3). There was little difference, however, in total protein concentrations in the lavage fluid of mice exposed to fresh vs. aged carbon nanoparticles (Figure 4). Thus, freshly generated carbon nanoparticles were present predominantly as singlets and produced greater inflammation (increased neutrophils (PMNs)) but not lung injury (total protein concentration).
Figure 3. The effect of fresh and aged carbon nanoparticles (1 to 5 mgm3) on the influx of PMNs in lung lavage fluid.
Figure 4. The effect of fresh and aged carbon nanoparticles (1 to 5 mgm3) on the influx of PMNs in lung lavage fluid.
Using the identical generation and exposure system, copper and zinc nanoparticles produced far greater lung injury and inflammation than did the carbon nanoparticles. As seen in Figure 5, 5 mg/m3 copper nanoparticles resulted in 70 to 80% PMNs (of total cells recovered) compared to approximately 25% PMNs after exposure to carbon nanoparticles. This difference in nanoparticle toxicity was more apparent at lower exposure concentrations. Significant increases in PMNs were observed at 0.2 mg/m3 and above for both fresh and aged copper nanoparticles (Figure 6). Zinc nanoparticles were slightly less toxic than the copper nanoparticles and a 60% increase in PMNs was seen after exposure to 1 mg/m3 fresh but not aged zinc nanoparticles (Figure 7). Thus, we observed a difference in nanoparticle toxicity for fresh vs. aged nanoparticles although the difference appeared to be greater for the carbon compared to the metal nanoparticles.
Figure 5. The effect of fresh and aged copper nanoparticles (5 mgm3) on the influx of PMNs in lung lavage fluid.
Figure 6. The effect of fresh and aged copper nanoparticles (0.05 to 0.9 mgm3) on the influx of PMNs in lavage fluid.
Figure 7. The effect of fresh and aged zinc nanoparticles (0.15 to 1 mgm3) on the influx of PMNs in lavage fluid.
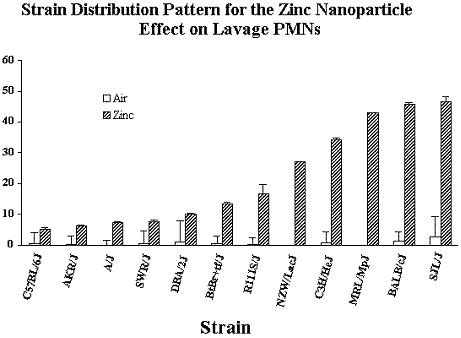
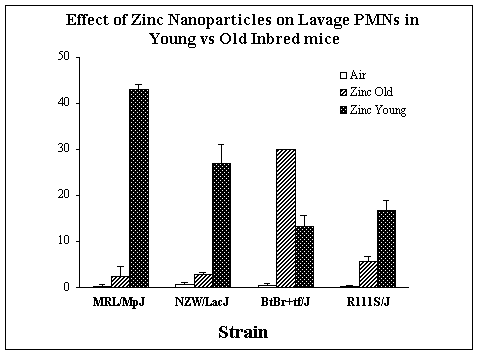
Upon completion of the 3 Specific Aims, we asked the additional questions whether host factors, such as genetics or age, would modify the response to inhaled nanoparticles. To address these issues, young and old adult mice (9 inbred strains) were exposed for 2 hrs to 0.7 mg/m3 fresh zinc nanoparticles. As we have observed with other inhaled particles and gases, significant inter-strain differences in response to inhaled zinc nanoparticles occurred. Surprisingly, the effect of inhaled nanoparticle on young vs. old mice varied significantly depending on the inbred strain of mice studied. Whereas the old (8 – 12 months old) mice were more sensitive than the young (2 - 3 months) adult mice in 3 inbred strains, the opposite effect (young were more sensitive than the old adult mice) occurred in the BtBr inbred strain.
Figure 8. The effect of fresh zinc nanoparticles (0.7 mgm3) on the influx of PMNs in lavage fluid in 12 inbred strains of mice.
Figure 9. The effect of fresh zinc nanoparticles (0.7 mgm3) on the influx of PMNs in lavage fluid in 4 inbred strains of young and old adult mice.
Conclusions:
Several important findings were observed in this research project. First, it appeared that some surface characteristics of nanoparticles do not affect particle agglomeration or toxicity. We found that the forces that give a particle its electrical charge did not influence the toxicity of carbon or metal nanoparticles. Additionally, relative humidity did not affect particle toxicity and or agglomeration in our mouse bioassay. Both findings were not expected, because certain nanoparticle surface characteris 2 or 3 times, such as chemical moities, are known to modify particle toxicity and one would predict that this affect would be even more dramatic for nanoparticles which have a greater surface to mass ratio.
As has been demonstrated for larger particles, the composition of a nano-size particle has a major influence on its toxic properties. Carbon nanoparticles were relatively inert compared to zinc and copper, the 2 metal nanoparticle types tested in this project. This suggests that risk assessment for nanoparticles must consider particle composition as well as size. Importantly, our data demonstrate that aged and agglomerated copper nanoparticles (approximately 200 nm in diameter) produced greater lung inflammation than did freshly generated (11-60 nm) carbon nanoparticles. Thus, particle composition appeared to be a more important risk factor than was the agglomeration state of the nanoparticles. A greater variety of nanoparticles would need to be tested to validate this observation.
Finally, additional experiments examined the role of genetics and age on the response of the mammalian lung to inhaled nanoparticles. Because inbred strains of mice are nearly genetically identical within a strain but less so amongst inbred strains, our observation of greatly varying responses among 11 inbred strains of mice exposed to freshly generated zinc nanoparticles suggests that genetic host factors influence the toxicity of nanoparticles. This observation would need validation studies in cross-breeding experiments that would establish whether the difference in sensitivity to inhaled nanoparticles can be inherited. Our experimental data also suggests that age can play a significant role in the toxicity of inhaled nanoparticles, although the increased responsiveness of older mice in some inbred strains was reversed in one inbred strain. Thus, there may be an interaction between genetics and the aging mammalian lung’s response to inhaled nanoparticles.
Key Personnel: There were no changes in key personnel.
Journal Articles:
No journal articles submitted with this report: View all 4 publications for this projectSupplemental Keywords:
nanoparticles, particle charge, age, mice, genetic susceptibility, Health, Scientific Discipline, ENVIRONMENTAL MANAGEMENT, Health Risk Assessment, Risk Assessments, Biochemistry, Risk Assessment, lung injury, nanochemistry, animal model, bioavailability, carbon fullerene, nanotechnology, nanomaterials, animal bioassays, nanoparticle toxicity, analysis of chemical exposureProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.