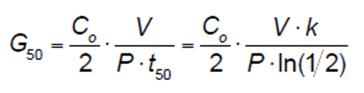Grantee Research Project Results
2014 Progress Report: Integration of Filtration and Advanced Oxidation: Development of a Membrane Liquid-Phase Plasma Reactor
EPA Grant Number: R835332Title: Integration of Filtration and Advanced Oxidation: Development of a Membrane Liquid-Phase Plasma Reactor
Investigators: Bellona, Christopher , Holsen, Thomas M. , Mededovic Thagard, Selma , Dickenson, Eric
Current Investigators: Bellona, Christopher , Holsen, Thomas M. , Dickenson, Eric , Mededovic Thagard, Selma
Institution: Clarkson University
Current Institution: Clarkson University , Southern Nevada Water Authority
EPA Project Officer: Packard, Benjamin H
Project Period: August 16, 2012 through August 15, 2016 (Extended to August 15, 2017)
Project Period Covered by this Report: August 16, 2013 through August 15,2014
Project Amount: $499,779
RFA: Research and Demonstration of Innovative Drinking Water Treatment Technologies in Small Systems (2011) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
Engineer, develop and demonstrate an integrated process comprised of membrane technology and electrical discharge plasma generated via a novel reticulated vitreous carbon (RVC) electrode material. The successful development of this process will result in a technology that is scalable, robust, requires minimal chemical input, has a small foot-print, and achieves a finished water quality better than treatment systems that require multiple technologies.
Progress Summary:
During the project period, the research team completed most of Phase II of the project (Fundamental Bench-scale Plasma/Membrane Reactor). The team has completed the work necessary for the design and construction of a demonstration-scale plasma reactor.
Phase I: Phase I of the current project was completed last year and a summary of the work was provided in the first progress report.
Phase II: The objective of Phase II is to conduct bench-scale studies to optimize the plasma process for contaminant destruction, elucidate reaction mechanisms, and evaluate system water quality. Several different sets of experiments have been performed to complete Phase II and a brief summary of findings are presented below.
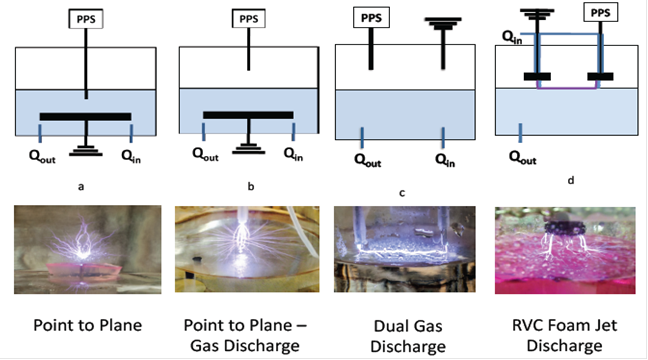
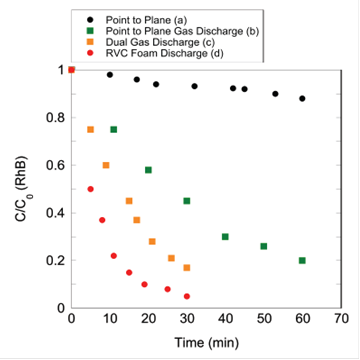
Identification of Most Effective Reactor Design: A variety of reactors (around 12 total) were built and used in contaminant degradation experiments with rhodamine-B (RhB), bisphenol-A, naproxen and phenytoin to screen for the most effective reactor configuration. The main difference between reactor types included whether or not plasma was formed in the gas phase or liquid phase, the type of highvoltage electrode, and how the water flowed into the reactor. The major findings from these experiments were that contaminant degradation is highest for gas discharge plasma, maximizing the contact time between the liquid phase and plasma increased contaminant degradation, and formation of plasma with a multiple point high-voltage significantly increased contaminant degradation. A schematic of four different reactor types is presented in Figure 1 and the degradation of RhB for the different reactors in Figure 2. RhB degradation was significantly enhanced by forming plasma in the gas phase and introducing the feed water solution through the reticulated vitreous carbon foam electrode.
Figure 1. Schematic of several plasma reactors with pictures of the formed plasma. Note – PPS is plasma power supply, and RVC is reticulated vitreous carbon foam.
Figure 2. Degradation of RhB by plasma for the reactors shown in Figure 1.
Influence of Reactor Parameters on Degradation: A series of experiments were conducted to identify the major reactor parameters that influence contaminant removal. The parameters investigated included the reactor diameter, the grounded electrode area, the recirculation rate, the initial contaminant concentration, air or argon sparging, and the frequency and input power of power supply pulses. Reactor efficiency was quantified using the G50 (g/kWh) value, which was calculated using the following formula:
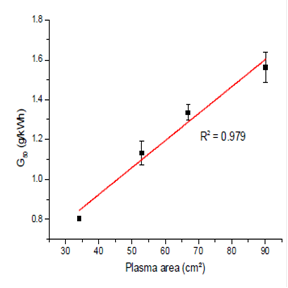
Where Co is the initial RhB concentration, V is the solution volume, t50 is the time required to reduce the initial concentration of the dye by 50%, P is the average input power and k is the pseudo-first order reaction rate constant (the experimental data confirmed the pseudo-first order assumption). Along with the initial contaminant concentration, the reactor diameter and grounded electrode area had a significant impact upon contaminant degradation. A summary of the impact of the grounded electrode area on the G50 value for RhB is provided in Figure 3. A larger grounded electrode increases the plasma area, which increases contact between the plasma and the contaminant. Thus, a plasma reactor is being designed that maximizes the plasma area relative to the liquid volume at any point in the system.
Figure 3. The impact of plasma area on the G50 value for RhB.
Additional Relevant Work: Along with running experiments to investigate the degradation of RhB, bisphenol-A, naproxen and phenytoin, work has commenced to identify byproducts of plasma degradation using liquid chromatography-time of flight (TOF) mass spectrometry. Preliminary work indicates that plasma partially mineralizes contaminants or converts them to hydroxylated intermediates. Further work will be conducted to better understand the formation of byproducts during treatment by plasma.
The plasma process produces considerable amount of hydrogen peroxide, which is not completely utilized during treatment. The research team performed a considerable amount of work to develop methods to convert the hydrogen peroxide to peroxide radicals that degrade organic contaminants. One such method investigated was Fenton’s reaction, which utilizes the reaction between Fe (II) to produce hydroxide radicals. It was determined that using an iron coated grounded electrode significantly improved the degradation of bisphenol-A and phenytoin due to Fenton’s reaction. We are evaluating whether or not such a process could be feasible as an integrated component of the plasma-membrane reactor.
We have finished Phase II and are now commencing with Phase III of the project. Because the ceramic membrane system was built during year one, the research is progressing on schedule.
Future Activities:
We are currently running experiments with a suite of organic contaminants (Table 1) at environmentally relevant concentrations to evaluate the two most effective reactors identified thus far. These experiments will be conducted over the next 2 weeks. Samples will be sent to Southern Nevada Water Authority for analysis. The research group currently has three manuscripts in preparation, which, will be submitted over the next 2 months. The research team is currently building a demonstration-scale plasma reactor that will be tested during Phase IV of the project.
Table 1. Contaminates for plasma degradation experiments.
|
Compound name |
Target Concentration |
| Acetaminophen | 500 ng/L |
| Atenolol | 500 ng/L |
| Caffeine | 500 ng/L |
| Carbamazepine | 500 ng/L |
| DEET (N,N-Diethyl-meta-toulamide) | 500 ng/L |
| Dibromoacetic acid | 50 µg/L |
| Fluoxetine | 500 ng/L |
| Gemfibrozil | 500 ng/L |
| Ibuprofen | 500 ng/L |
| Ketoprofen | 500 ng/L |
| Meprobamate | 500 ng/L |
| Monochloroacetic acid | 50 µg/L |
| N-Nitrosodimethylamine (NDMA) | 500 ng/L |
| Naproxen | 500 ng/L |
| Perfluorobutyric acid (PFBA) | 500 ng/L |
| Perfluorooctanoic acid (PFOA) | 500 ng/L |
| Perfluorooctane sulfonate (PFOS) | 500 ng/L |
| Primidone | 500 ng/L |
| Sucralose | 500 ng/L |
| Sulfamethoxazole | 500 ng/L |
| TCEP (Tris(2-chloroethyl)phosphate) | 500 ng/L |
| Triclocarban | 500 ng/L |
| Triclosan | 500 ng/L |
| Trimethoprim | 500 ng/L |
| Trichloroacetic acid | 50 µg/L |
Journal Articles:
No journal articles submitted with this report: View all 22 publications for this projectSupplemental Keywords:
Drinking water, chemicals, engineering, innovative technology, membrane filtration, advanced oxidation, plasma reactorProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.