Grantee Research Project Results
Final Report: Pore Scale Determination of the Rate of NAPL Dissolution or Volatilization
EPA Grant Number: R826268Title: Pore Scale Determination of the Rate of NAPL Dissolution or Volatilization
Investigators: Keller, Arturo A.
Institution: University of California - Santa Barbara
EPA Project Officer: Aja, Hayley
Project Period: October 1, 1997 through September 30, 1999
Project Amount: $133,179
RFA: Exploratory Research - Physics (1997) RFA Text | Recipients Lists
Research Category: Water , Air , Safer Chemicals , Land and Waste Management
Objective:
Our objective in this study was to theoretically predict and experimentally observe the dissolution and volatilization processes at the pore scale as a function of saturation and pore space geometry for various Non-Aqueous Phase Liquids (NAPLs), and then to determine how the mass transfer coefficient can be derived from pore scale processes, which can then be scaled to field dimensions, and to understand the functional relationship between mass transfer and hydrologic and physicochemical properties and processes. We also sought to determine the relationship between NAPL saturation, Sn, and the effective interfacial contact area between, app1. and how well this correlates with. Our hypothesis was that the relationship between app1 and Sn varies with time, is non-linear, but can be expressed as a power function. Our specific objectives are to:
- Develop a conceptual and mathematical model of mass transfer based on pore scale processes;
- Provide a methodology for scaling up our pore scale model to the field scale;
- Confirm our model using pore scale observations of the mass transfer process
Summary/Accomplishments (Outputs/Outcomes):
Non-Aqueous Phase Liquids (NAPLs), such as chlorinated solvents and hydrocarbon fuels, have been used extensively in private industry, military installations and DOE facilities. NAPL spills during transport and leaks from underground storage tanks have inevitably occurred and represent a major risk to water supply, since even a small amount of NAPLs can contaminate large volumes of groundwater. NAPL ganglia (blobs) trapped in the porous soil or rock matrix at ?residual? saturation (Dawson, 1992; Kueper et al., 1993) are a continuous source of contamination to the aquifer or the soil vapor, through dissolution or vaporization (Garg and Rixey, 1999; Rixey, 1996; Smith et al., 1996).
To design a remediation scheme, it is important to understand at a basic level the physicochemical processes that govern inter-phase mass transfer of NAPLs in the subsurface, both in the unsaturated and the water-saturated regions. The rate at which NAPLs dissolve or volatilize in large part determines the time required to clean up a contaminated site, whether via Soil Vapor Extraction (SVE) (Falta et al., 1993; Gomez-Lahoz et al., 1994b; Lingineni and Dhir, 1997; Rathfelder et al., 1996; Wilson et al., 1994), bioremediation and its many variants (bioventing, bioslurping, etc.) (Ghoshal and Luthy, 1998; Ghoshal et al., 1996; Mukherji and Weber, 1998; Seagren et al., 1993; Sekelsky and Shreve, 1999; Yang et al., 1995) and pump-and-treat (Gomez- Lahoz et al., 1994a). Non-equilibrium conditions are most important during active remediation, when the rate of inter-phase mass transfer can significantly limit the effectiveness of any treatment scheme, but may also play a role during natural attenuation (Grathwohl, 1998; Libelo et al., 1998; Reichert et al., 1998; Stauffer et al., 1998).
Modeling and experimental work (Armstrong et al., 1994; Geller, 1990; Geller and Hunt, 1993; Grathwohl et al., 1990; Hunt et al., ; Imhoff et al., 1990; Imhoff et al., 1993; Imhoff and Miller, 1996; Imhoff et al., 1996; Mayer and Miller, 1992; McKay and Gillham, 1993; Wilkins et al., 1995) indicates that inter-phase mass transfer may be a limiting factor when NAPL saturations are low, the aqueous or vapor phase velocities are large or the NAPL is only slightly soluble, conditions which are typical during soil remediation of many common organic pollutants. Many remediation proposals consider that mass-transfer is not rate limiting; up to recently, few numerical models have included this important consideration in their simulations, with the corresponding potential error in determining the required clean-up time and/or risk to drinking water supplies or human/ecological exposure. Some of the models that do consider rate-limited mass transfer include the work by (Abriola et al., 1993; Abriola and Pinder, 1985; Forsyth and Sudicky, 1998; Forsyth et al., 1998; Rathfelder et al., 1996; Rubin et al., 1997; Wilkins et al., 1995).
Conceptual Model of Mass Transfer from the Pore Scale
Three important processes need to be considered in a phenomenologically based model: (1) molecular diffusion from the blob to upstream and downstream water; (2) dissolution into pore water trapped in the crevices which is then transported advectively downgradient; and (3) mixing of waters with different concentrations of dissolved NAPL constituents. Consider the pore space presented in Figure E1. As soon as organic molecules transfer from the NAPL blob to the surrounding water phase, they begin to diffuse by Brownian motion towards upstream and downstream regions, which we will indicate with subscripts u (upstream) and d (downstream). Consider an ?inlet? location far enough upgradient that upstream diffusion is insignificant. The concentration of solute c in water at that point is . The concentration at a point just upstream of the NAPL blob,
, may be different from
due to molecular diffusion. Using a mass balance around this upstream point, we obtain:
where , which is the Peclet number, i.e. the ratio between advective and diffusive processes; ? is the porosity of the medium; Sw is the saturation of water; qw is the specific discharge; Lu is the distance between the NAPL blob and Point U; and
is the molecular diffusion coefficient for solute c in water.. The exact same derivation can be used to derive the formulation for the downstream point D. For very low flowrates (Pe << 1), the concentration around the pore blob approaches the equilibrium concentration,
. For very large flowrates (Pe >> 1),
, since solute c is being swept away as fast or faster than it can diffuse from the NAPL blob.
Transport of solute c out of the crevice by water creeping past the NAPL blob is governed by the advective flow of water past the NAPL. We can assume that the water is flowing slowly enough for complete mixing of the water in the crevice such that the concentration everywhere within this region is . The change in concentration from Point U to Point D due to this mass flux is then:
where is the conductance of water through the crevice [L4 T M-1]; Lud is the length between Point U and Point D, [L]; Ap is the cross-section of the pore body, [L2]; and Pu, Pd are the pressures of water at Point U and Point D. At very low flowrates (Pe << 1), the pressure differential between Point U and Point D is quite small, and given that the conductance through this path is much lower than through ?NAPL-free? pore bodies, the change in mass from Point U to Point D due to transport through the crevice is quite small. In the limit, as
Water from different pathways mixes again after the NAPL blob. Water at mixes with water at
to form water at the next point below Point i, to produce water at a concentration
. The change in concentration from i to i + 1 is then:
the crevice, [-], Lp is the distance between Point i and Point i + 1, [L], approximately the length of a pore body.
Using the definition of the Sherwood number, Sh, we show that:
where we make the assumption that d50 ? Lp, since these two dimensions are of the same order of magnitude. We have thus derived an expression for Sh (and thus the mass transfer coefficient) that depends on Pe and the parameters included M, which combine molecular diffusion, transport of solute out of the crevices and mixing of pore waters after passing the NAPL blob. Through M, we have also captured a dependence of Sh on Sn (= 1 - Sw), thus accounting for the change in the rate of mass transfer with decreasing Sn.
By looking at the two extreme conditions, we can see how the model behaves. When Pe << 1, M is nearly 1, since the most important process is molecular diffusion and the concentration around the NAPL blob is essentially the same, nearly . In this case, Sh is directly proportional to Pe. When Pe >> 1, M can be shown to be proportional to Pe-1/2, and thus Sh is proportional to Pe1/2.
How does this model compare with the empirical observations? For the Miller and Mayer (1990) and Powers et al. (1994) empirical correlations, Sh appears to depend on Pe to a power of from 0.6 to 0.75. The power law model derived from a consideration of pore scale processes varies from 0.5 to 1 as Pe decreases, which is consistent with the empirical observations, which have typically been performed at Pe ranging from 10 to 1000. The observed variability in the exponent of the empirical correlations may be explained by the fact that the underlying processes for mass transfer are changing as Pe varies.
Experimental Observation of Mass Transfer at the Pore Scale
To study in detail the dynamic dissolution and volatilization processes at the pore scale and to be able to quantitatively measure NAPL saturation and interfacial area requires a setup that non-intrusively permits the observation and recording of pore scale processes. In this project, we developed a micromodel setup, which we had previously successfully applied to observe pore scale displacement of NAPL during two- and three-phase flow conditions (Keller et al., 1997).
At the beginning of the sequence in Figure E2, we observe a NAPL blob which spans over two pore bodies, since the connecting pore throat has dimensions which are of similar size as one of the pore bodies. The effective interfacial contact area is mostly at the pore throats, which represents a much smaller area than the total surface area of the NAPL blob. After 99 seconds, the NAPL blob has shrunk in size and has reconfigured itself, withdrawing to a single pore body. Further rearrangements occur throughout the volatilization process as the NAPL blob finds positions of lower energy. Note for example how the NAPL blob adopts a spherical shape (circular in this quasi- two-dimensional porous media) once it does not have enough volume to occupy the entire pore body. The rate of mass transfer at the pore scale is strongly dependent on the diffusion of mass in the upstream and downstream directions. Molecules leaving the NAPL blob must diffuse out through some very small pore throats (on the order of 3 to 10 µm). Once past the pore throats, these molecules are swept away by the flowing gas phase, so the rate-controlling step is molecular diffusion through these thin throats.
We have captured these observations of the dissolution and volatilization processes in a schematic description of the sequence of rearrangements in our simplified quasi-rectangular pore body, connected to other pores through the corners, as shown in Figure E3. Initially the pore is highly saturated with NAPL and the only contact with the flowing fluid is through at the exposed interface in the pore throats. Once mass begins to transfer to the flowing fluid (via molecular diffusion through the pore throats), app1 increases, since the flowing fluid is now exposed to more of the NAPL surface area. Active flushing of the free pore space may occur, although in general fluid flow through this region is expected to be slow, given that there is not a significant pressure gradient across the pore body. As the mass transfer process proceeds and the NAPL blob slowly recedes, app1 remains generally constant, until the point where the NAPL blob reconfigures to a spherical shape. At that point, there is a sudden increase in the effective interfacial contact area, as more of the flowing fluid can contact the NAPL blob. However, this increase is short-lived, as app1 begins to sharply decrease as the NAPL sphere becomes smaller and smaller. Thus, although the behavior of app1 with respect to Sn is highly non-linear, it can be understood and generalized.
Conclusions:
We present a mathematical model that phenomenologically describes mass transfer (whether volatilization or dissolution) at the pore scale. The model can be scaled up to the dimensions of a laboratory core or a field site. The model predicts that Sh depends on Pe to a power of one-half when the flowrate of the flowing fluid (air or water) is very slow (Pe << 1). As Pe increases, the model predicts that the dependence of Sh on Pe increases, until at very large flowrates (Pe >> 1), Sh is directly proportional to Pe. Since Pe is the ratio between advective and diffusive processes, this indicates that at the higher flowrates typical of laboratory core experiments it would be likely to observe a dependence greater than 0.5. In fact, most recent experiments (Miller and Mayer, 1990) and (Powers et al., 1994) indicate that the dependence of Sh on Pe in laboratory experiments ranges from 0.6 to 0.75, well in line with the prediction of the phenomenological model. One would expect that for field studies of the rate of mass transfer, the correct exponent would be closer to 0.5, since most field sites operate at low Pe values. Under these conditions, the assumption that local equilibrium (LEA) exists seems to be justified. However, for active remediation (e.g. Soil Vapor Extraction, vigorous pump and treat, or soil flushing with surfactants which raise the solubility limit), LEA may not be valid, and Sh is likely to depend more on Pe (i.e. a higher exponent would be warranted, closer to the laboratory experiments).
In addition to the dependence of Sh on Pe, it also depends on Sn, via the molecular diffusion process. The dependence of Sh on Sn is not as easily explained, given that the underlying relationship is between Sh and anw, which up to now had been difficult to characterize. However, an evaluation of the functional relationship indicates that as Sn becomes smaller, the rate of mass transfer (and thus Sh) increases, so there is an inverse relationship between Sh and Sn. It is also readily seen that as Sn becomes very small, the flowing fluid can more easily flow past the NAPL blob, and a larger interfacial area is contacted, also increasing the rate of mass transfer.
In Section 5 we present experimental evidence that the phenomenological model described in Section 3 is correct. Even at the low Pe of these micromodel experiments, advective transport through the crevices is small relative to the diffusive transport, since the cross-sectional area for the flowing fluid is very small, in particular when the cross-section is a complex mixture of geometries. We observe how the mass transfer process follows essentially a power law behavior, with an increasing exponent as the end of the mass transfer occurs. This acceleration in mass transfer can be explained by the fact that once the NAPL blob begins to shrink, it allows more crevice transport, the surface area contacted by the flowing fluid increases, and overall mass transfer grows.
We present a model for the dependence of anw on Sn, which is based on our observations and generalization of the mass transfer process. This model predicts that, at the pore scale, initially as Sn decreases, anw begins to increase, since the NAPL blob shrinks slightly and exposes more interfacial contact area. Once the NAPL blob has shrunk enough to become spherical (thereby reducing its surface energy), anw sharply increases, only to decrease as the spherical blob rapidly losses size. Thus in large part explains the observed acceleration of mass transfer at the end of the mass transfer process for an individual NAPL blob. This behavior appears to be generalizable to any fluid/fluid pair (NAPL/gas, NAPL/water, and probably air/water), and can be easily codified for further analysis in a network model.
One important consideration is that the aggregate behavior of a combination of NAPL blobs is more complex, even though it is only the sum of simple processes. This macroscopic complexity is responsible for the empirical approach undertaken to date to explain mass transfer, and also the variance between different experimentalists. The use of numerical modeling tools (e.g. network models) based on these observations at the pore scale can serve to explain the macroscopic behavior, and elucidate the relative importance of the various processes and the degree of NAPL saturation.
This project has served to establish a fundamental understanding of mass transfer, which is of relevance under many natural and engineered systems, including site remediation through a variety of methods. The phenomenological model serves to underpin the empirical models, and explain their variance. The experimental observations from this project will serve to develop more complete numerical models that can be used for prediction of the rate of mass transfer under a variety of conditions, including different soils, fluid pairs, saturation and factors that affect the physicochemical properties of the compounds of interest.
Supplemental Keywords:
Mass transfer, air, water, porous media, remediation, network model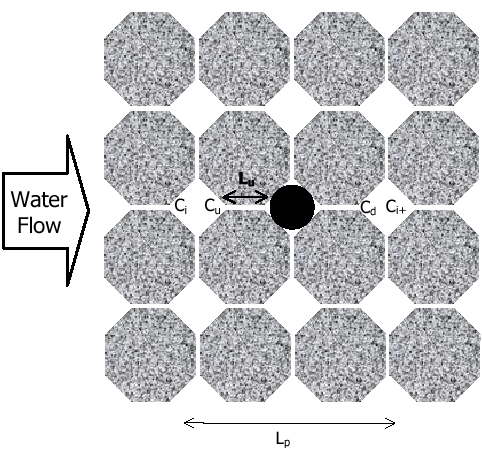
Figure E1. Residual NAPL blob disolving in flowing water.

Figure E2. Volatilization of NAPL to Air.
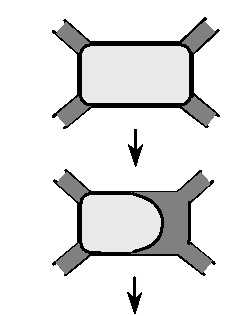
Figure E2. Generalized sequence of NAPL
rearrangements during mass transfer.
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
