Grantee Research Project Results
Final Report: Perinatal Exposures, Epigenetics, Child Obesity & Sexual Maturation
EPA Grant Number: R834800Center: Center for Research on Early Childhood Exposure and Development in Puerto Rico
Center Director: Alshawabkeh, Akram
Title: Perinatal Exposures, Epigenetics, Child Obesity & Sexual Maturation
Investigators: Peterson, Karen E. , Dolinoy, Dana , Meeker, John D.
Institution: University of Michigan
EPA Project Officer: Hahn, Intaek
Project Period: August 9, 2010 through August 8, 2014
Project Amount: $1,335,311
RFA: Children's Environmental Health and Disease Prevention Research Centers: Formative Centers (with NIEHS) (2009) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
Center Objectives
Overweight and obesity in children has increased significantly over the past few decades. Many attempts have been made to reverse this trend through school food reforms, clinical counseling of at-risk youth, camps and group clinics that focus on lifestyle and behavior change. However, the Formative Children’s Environmental Health and Disease Prevention Center (UM-CEHC) research offers a new perspective on the childhood obesity dilemma. Recent environmental health research suggests that early-life exposure to environmental toxins, such as lead, bisphenol A and phthalates, which we are studying at the UM-CEHC, may also affect child growth trajectories, body mass index (BMI) and the tempo of sexual maturation.
If an association between in utero or early-life exposure to lead, bisphenol A or phthalates with child growth, development and/or sexual maturation is demonstrated, this information can be used to inform future policies for decreasing exposure to substances related to obesity and the multitude of associated co-morbidities. Knowledge of environmental exposures that are associated with adverse health effects can also be used by physicians, public health workers and community activists to educate the general public about ways to avoid these exposures.
The specific aims of this Center are to:
- Characterize the effect of lead, bisphenol A and phthalates on weight gain and status (BMI) and the development of obesity in early/mid childhood and on pubertal timing assessed by Tanner staging and hormonal biomarkers.
- Utilizing lead as a model toxicant, examine the epigenetic mechanisms (e.g., DNA methylation of four key growth-regulating genes) associated with perinatal exposure and potential modification of the development of obesity and pubertal timing.
- Enhance career development by assisting new investigators and trainees in the advancement of their research skills and knowledge in translational and children’s environmental health research.
- Involve key stakeholders in Michigan and Mexico via a Community Engagement Plan aimed to educate and inform study participants, policy makers and public health decision makers on children’s environmental health exposures of concern.
- Apply research findings, infrastructure and partnerships gained through the Formative P20 Center to the development of a full P01 Children’s Environmental Health Center at the University of Michigan.
Research Project Objectives
Cross-sectional studies suggest that low-level lead exposure is associated with physician-observed onset of puberty in boys and girls, delayed age of self-reported menarche in girls, and marginally reduced testicular volume in boys. Prenatal lead exposure among rats has been shown to cause a decrease in several puberty-related hormones. Lead is consistently associated with decreased birth and infant length that continues into early childhood as stature decrement; however, the association between lead exposure and child weight status is less consistent. This project is primed to address both the question of lead’s effect on child weight status and tempo of sexual maturation. Preliminary studies suggest that maternal lead measures are inversely associated with umbilical cord DNA methylation. Epigenetic fetal programming via DNA methylation may provide a pathway for lead exposure to influence disease susceptibility. The analysis of umbilical cord DNA methylation in Research Project subjects will provide valuable insight into a possible association between prenatal lead exposure and childhood weight gain and weight status.
The specific aims of this Research Project are to:
- Describe the relationship of prenatal lead exposure to sexual maturation.
- Describe the association of prenatal lead exposure with childhood weight gain and status.
- Examine the relationship of prenatal lead exposure to cord blood DNA methylation levels in four genes associated with early growth.
Pilot Project 1 Objectives
Using lead and BPA as representative environmental toxicants, a mouse model of dose-dependent epigenetic and life-course physiological assessment will strengthen children’s health risk assessment and shape diagnostic and therapeutic strategies. First, the identification of epigenetic exposure biomarkers will be useful in the identification of at-risk individuals prior to disease onset. Second, unlike genetic mutations, epigenetic profiles are potentially reversible. Therefore, epigenetic approaches for prevention and treatment, such as nutritional supplementation or pharmaceutical therapy, may be developed to counteract negative epigenomic profiles. For example, if we determine that lead exposure results in hypomethylation and increased incidence of yellow Avy/a offspring, it would be important to determine if nutritional supplements (e.g., folic acid, genistein) not only during development but also later in life can protect against these negative effects on the epigenome. Therefore, the future of epigenomics holds tremendous potential not only for individualized children’s health, but also for population-wide disease diagnostic, screening and prevention strategies.
The specific aims of this Pilot Project are to:
- Develop a mouse model of dose-dependent maternal lead effects on the fetal epigenome.
- Examine the relationship of prenatal lead exposure to physiological parameters and hormonal status throughout the life-course.
Pilot Project 2 Objectives
Human exposure to phthalates and bisphenol A are widespread, so the public health significance for Pilot Project 2 is potentially huge. The health effects of this exposure may be amplified by the sensitive developmental windows being assessed in the study. In the meantime, the analysis being conducted using NHANES and other data sources could also have large impacts in helping us understand human exposure to these compounds and their related health effects.
The specific aims of Pilot Project 2 are to:
- Compare urinary concentrations of BPA and phthalate metabolites from pregnant women and adolescents in Mexico City with those measured in the U.S. general population.
- Explore the association between BPA and phthalate exposure and birth outcomes.
- Investigate the relationship between early-life (in utero) exposure to BPA and phthalates and measures of weight, body mass index, growth in infancy and early childhood.
- Explore the association between exposure to BPA and phthalates in early life (in utero) and in adolescence with stages of sexual maturation and relevant endocrine markers in Mexican youth.
- Determine predictors of BPA and phthalate exposure among adolescents in Mexico City.
Summary/Accomplishments (Outputs/Outcomes):
Center Summary
Career Development
- Our institutional commitment to Career Development is reflected in the Center leadership and organizational structure: both Pilot Project Principal Investigators (Dr. Dolinoy, Dr. Meeker) and the Leader of the Environmental Biostatistics Core (Dr. Sanchez) were new investigators at the time the P20 CEHC was initiated in 2010 and have been highly successful in obtaining National Institutes of Health R21 and R01 funding and in publishing in peer-reviewed journals. These new investigators were all Assistant Professors at the time the grant began and have all been promoted to Associate Professors with tenure.
- Doctoral students affiliated with the P20 CEHC completed dissertation research, publications and/or presentations, and three have received Ph.D. degrees: Olivia Anderson, M.P.H., R.D., Ph.D. in Environmental Health Sciences, 2013 (Mentor: Dr. Dolinoy); Kelly Ferguson, Ph.D. in Environmental Health Sciences, 2014 (Mentor: Dr. Meeker); and Tiffany C. Yang, M.P.H., R.D., Ph.D. in Nutritional Sciences, 2014 (Mentor: Dr. Peterson).
- Two postdoctoral fellows also participated and presented their findings at bi-monthly meetings and mentored doctoral students. Jaclyn Goodrich, Ph.D., is affiliated with P20 CEHC research and was appointed to Center Scientist of the U-M P30 NIEHS Environmental Health Science Core Center in 2014. Dr. Goodrich’s background is in gene/environment interactions, with a specific focus on metals and endocrine active compounds. Postdoctoral Fellow Christopher Faulk, Ph.D., who oversees Pilot Project 1, was awarded a K99/R00 grant from NIEHS. Center faculty, postdoctoral fellows and doctoral students participated in the following seminars and symposia during Year 3: Annual UM EHS Toxicology Symposium, and Annual Michigan Metabolomics and Obesity Symposium
Community Engagement
- Partnership with Head Start for Kent County, the Kent County Health Department and the Healthy Homes Coalition to identify collaborative projects and topics to improve children’s environmental health. This collaborative base was essential to continue projects for the Community Outreach and Translation Core of the P01 CEHC. Our partners have a long-standing relationship around lead abatement, and they have identified asthma as a prevalent and persistent public health problem in Kent County. We worked to develop a spatial mapping project with our partners and with the West Michigan Environmental Action Council and the Asthma Network of West Michigan. Through a collaborative process and a partnership with Grand Valley State University, we will be working to produce maps illustrating the prevalence of asthma that will be used to educate the community and create a stronger platform for addressing childhood asthma in Kent County, Michigan. This mapping project will strengthen the base for addressing children’s environmental health in Kent County and can be adapted for other relevant exposures and child health outcomes during the P01 CEHC project period.
- In 2012, we began collaborating with the Children’s Environmental Health Network (CEHN), a national nonprofit that works to protect children from environmental health hazards. Through in-person meetings (October 2013, in conjunction with CEHC national meetings) and conference calls with Nse Witherspoon and Carol Stroebel of CEHN, we were able to identify the Eco-Healthy Child Care Program as a project for collaboration. We plan to continue to this work in the P01 CEHC, where we will work to bring a state-wide training of the Eco-Healthy Child Care Program to Michigan. The UM-CEHC contributed to a White Paper that will be used for the CEHN’s Wingspread Retreat in Fall 2014.
- In 2013, Dr. Alejandra Cantoral, who is affiliated with the P20 CEHC, presented findings linking consumption of sugar-sweetened beverages with overweight and obesity in children in the ELEMENT cohort at the 15th Annual Congreso de Investigación en Salud Pública in Mexico. Through our ongoing partnership with the Instituto de Salud Pública de Mexico (INSP), this and ongoing P01 research on environmental and dietary determinants of child obesity and metabolic outcomes will educate and inform public health researchers nationwide in Mexico.
- In June 2014, our partners at INSP were successful in launching a new initiative with the Blacksmith Institute and Local Ministry of Health in the State of Morelos (where INSP is located) to remove lead from the ceramics in Morelos, supported by a wide coalition of artisans, hotels and restaurants: http://www.insp.mx/centros/evaluacion-y-encuestas/prensa/3314-morelos-libre-plomo.html Exit. Research funded by the NIEHS and EPA, including the P20, that has documented the effects of Pb on children’s health has been an essential part of the evidence base supporting the implementation of this environmental health policy in the state of Morelos, Mexico.
Administrative Core
- In addition to bi-monthly meetings of Center investigators, regular modes of communication, including conference calls between investigators at different locations (e.g., INSP), enhanced the efficiency of data collection, maintenance of protocols for protection of human subjects, and integrity of biospecimens and data management.
- Creation of a bio-repository of historical samples from the parent longitudinal cohort, ELEMENT, has progressed.
Environmental Biostatistics Core
- An updated, streamlined data recording and transferring system was created by study statisticians in Mexico and Michigan to enhance data sharing and support human Project data analyses.
- Sample collection and questionnaire protocols for human Projects were updated, and standardization of study staff for anthropometric measurements and Tanner staging will occur shortly.
- Expanding on the collaborations, Drs. Sanchez and Song, together with Ph.D. student Meihua Wu, submitted a manuscript where approaches for sample size determination for high dimensional classification studies were developed and software was implemented to make those approaches accessible to the broader community.
- Dr. Sanchez and biostatistics colleagues also submitted a manuscript where they developed statistical approaches to examine the impact of correlated exposures (e.g., multiple biomarkers) on growth trajectories. The study focused also on how to improve estimation accuracy for effect modification of genetic or epigenetic marks on the association between multiple pollutant exposures and growth.
- Dr. Peterson continued to offer her advanced doctoral seminar, EHS 796 Special Topics in Environmental Health Sciences: “Lifecycle periods for development of obesity.” The course provides in-depth examination of conceptual frameworks, research design and methods in human and animal studies related to environmental and nutrition exposures and health outcomes in the context of students’ dissertation research and also supports the development of scientific presentation skills.
Research Project Summary
Summary of Findings (Outputs/Outcomes):
We surpassed our initial target of 200 participants and recruited 250 participants, 118 males and 132 females. Participants aged approximately 7 to 15 years provided biological specimens (blood and urine) and completed the food frequency, physical activity and exposure assessment questionnaires; anthropometry (height, weight, waist circumference and skinfolds); and a physical exam by a physician, including Tanner staging and a self-report Tanner staging questionnaire.
Statistical analysis for Aim 1, examining the effect of Pb exposure on hormones in conjunction with observed and self-reported Tanner staging, continued in Year 3. The manuscript “Environmental lead exposure and odds of delayed pubertal onset” (Afeiche, et al.) was submitted in February 2012 to the journal Pediatrics. Utilizing data from the ELEMENT cohorts, this study examined the effect of cumulative prenatal lead exposure (maternal patella lead) on children’s pubertal onset, assessed by self-reported Tanner stage for breast, pubic hair and genitalia. Maternal patella lead was not associated with odds of earlier or later puberty onset in boys or girls, although a 1 μg/g increase in maternal patella lead was associated with non-significant odds of earlier breast development in girls. This manuscript was revised and resubmitted during the No-Cost Extension period to the Journal of Perinatal and Pediatric Epidemiology and currently is under review. In addition, we completed statistical analysis for our validation of self-reported sexual maturation (proxy-assisted) and these findings were accepted for a poster being presented in June 2014 at the Society for Pediatric Epidemiology Research (SPER).
Abstract:
Validation of self-assessed sexual maturation using the method of triads (Chavarro, et al.)
Background: Self-assessed sexual maturation is often required in large-scale epidemiologic studies, and generally validated against physician assessments. However, physician assessments are highly dependent on specialized training and experience.
Methods: We adapted a self-assessment instrument that 248 Mexican children and adolescents, age 8–15 years, completed with parental assistance. They were examined by a single, trained pediatric endocrinologist and provided fasting blood samples for measurement of levels of reproductive hormones (T, E2, SHBG, Inhibin B) and other hormones known to increase during adolescence (C-peptide, IGF-1, DHEA-S). Spearman correlations and 95 percent confidence intervals were calculated among the average rank of all hormones, self- and physician-assessed Tanner stage. The method of triads, a technique developed to validate dietary assessment tools, was used to estimate the correlation between self-assessed sexual maturation and true, but unobservable, sexual maturation based on all available data. Ninety-five percent confidence intervals were constructed using bootstrap sampling.
Results: Validity of self-reported genitalia staging for boys was modest (r[95%CI] = 0.50[0.31–0.65]). Results were similar when testicular volume was used instead of physician’s Tanner staging. Breast stage was well reported (0.89[0.79–0.97]) and slightly superior to physician assessment (0.80[0.70–0.89]). Pubic hair stage was well reported by boys (0.91[0.79–0.99]) and girls (0.99[0.96–1.00]) and superior to physician assessment (0.79[0.57–0.97] and 0.91[0.83–0.97], respectively). Correlations between self and physician assessments were comparable to those previously observed in traditional validation studies.
Conclusions: Self-assessment can be validly used in epidemiologic studies for the evaluation of sexual maturation in girls and pubic hair development in boys. Physician assessment may be necessary to differentiate genitalia from pubic hair development in boys.
Also related to Aim 1, our analysis relating Pb exposure to hormones associated with secondary sexual characteristics in boys and girls was accepted for oral presentation at ISEE 2014 (Seattle).
Abstract:
Association of life stage lead exposures with pubertal onset and hormones related to secondary sexual characteristics in a Mexican birth cohort (Peterson, et al.)
Few studies have considered the impact of lead exposures at multiple developmental stages on pubertal onset or their relationships with hormones influencing secondary sexual characteristics. We examined prenatal, early childhood and concurrent lead exposure in relation to pubertal onset and fasting serum hormone concentrations at ages 8–14 in a Mexican birth cohort. We estimated associations of maternal bone lead at 1-month postpartum and blood lead levels (umbilical cord, cumulative 1–4 years of age, concurrent) with odds of physician-assessed Tanner stage >2 in boys (N = 117; genitalia [G] and pubic hair [P]) and in girls (N = 131; breast [B], pubic hair, menarche) and with log-transformed concentrations of dehydroepiandrosterone sulfate (DHEA-S), estradiol (E2), inhibin B, sex-hormone binding globulin (SHBG) and testosterone (T) with multivariable logistic and linear regression, respectively, adjusting for age, BMI-Z score and maternal education. Lead levels were: patella, 8.9 ± 10.2 μg/g bone; tibia, 7.4 ± 9.7 μg/g; loge-umbilical cord blood, 3.7 ± 2.1 μg/dL; 1–4 year loge-blood (cumulative), 14.4 ± 1.4 μg/dL; and loge-current blood, 3.3 ± 2.8 μg/dL. In girls, a loge-μg/dL increase in current blood lead was associated with 0.42 lower odds (95% CI: 0.21–0.83) of ≥ P2, and patella lead was negatively associated with inhibin B (b = −0.014, p = 0.02). In boys, cord blood and tibia lead were positively associated with DHEA-S (b = 0.323 p = 0.004; b = 0.021, p = 0.002, respectively), a loge-dL increase in cumulative blood lead from 1–4 years was associated with a −1.141 decrease in T (p = 0.007), and patella lead was positively associated with SHBG (b = 0.011, p = 0.01). Findings suggest early-life lead exposures may be negatively associated with pubertal onset in girls and blood levels of specific sex hormones in boys and girls. Specifically, prenatal lead exposure was positively associated with hormones influencing adrenarche in boys, while concurrent exposure may be related to delayed pubarche in girls. This analysis also suggests that hormonal biomarkers may be important outcomes to augment Tanner staging that may provide insight into mechanisms by which Pb and is related to pubertal onset and maturational tempo.
Relevant to Aim 2, the manuscript, “Prenatal lead exposure and weight of 0 to 5 year-old children in Mexico City” (Afeiche, Peterson, et al., 2011) was published in Environmental Health Perspectives. The article utilized previous ELEMENT cohort data, with study subjects overlapping with those targeted for recruitment for the UM-CEHC Research Project and Pilot Project 2. This was the first longitudinal study to investigate the association of cumulative prenatal lead exposure on children’s weight trajectory between birth and 5 years of age. Analysis suggested that prenatal lead exposure, assessed by maternal patella lead, measured 1 month postpartum, is associated with a significantly lower weight in female children by age 5. Male children with prenatal lead exposure were found to have nonsignificantly increased weight at 5 years of age.
In addition, the manuscript “Windows of lead exposure sensitivity, attained height, and BMI at 48 months” (Afeiche, et al.) was published in the Journal of Pediatrics. Analysis constructed each child’s lead exposure history for three developmental periods: prenatal (maternal patella lead), infancy and early childhood (mean child blood lead between birth and 24 months and 30 to 48 months, respectively). Children with higher than median blood lead levels during infancy were significantly shorter at 48 months than children with blood lead levels below the median, suggesting infancy as the window of greatest sensitivity to lead exposure. Increased lead exposure trended to lower BMI, but these results were not significant.
Building on this Aim 2 work and findings presented at ISEE in 2013 showing persistent declines in physical growth at 7–15 years related to early Pb exposure, we next examined the relationship of Pb in different sensitive periods (in utero, adiposity rebound and peripuberty) to metabolic outcomes in youth 7–15 years. These pilot data for our P01 application were presented at ISEE 2013 in Basel, Switzerland (Peterson, et al.): “Association of in utero and peripubertal EDC exposures with metabolic syndrome risk.” Although we did not see a hypothesized increase in adiposity in the pubertal transition in our earlier work, we found that Pb was associated with measures of elevated cardio-metabolic risk. Among boys, Pb at 4 years was related to higher fasting glucose, whereas concurrent Pb was associated with lower high density lipoprotein (HDL) cholesterol in girls. These analyses currently are being redone using the full P20 sample size (n = 248), and we plan to submit a manuscript by October 2014.
Table 1. Association of lead exposures with glucose and lipids (mg/dL) at 8-17 yr
| Exposure Timing | Overall a=233) | Girls (a=113) | Boys(a=120) | |||
|---|---|---|---|---|---|---|
| Beta | p-value | Beta | p-value | Beta | p-value | |
| Glucose | ||||||
| Blood lead, 4r | 0.69 | 0.03 | 1.75 | NS | 6.04 | 0.053 |
| HDL Cholesterol | ||||||
| Blood lead, 8-17 yr | -3.12 | 0.02 | -3.69 | 0.03 | -2.92 | NS |
| Tibea bone lead, 1mo postpartum | 0.70 | NS | 1.75 | NS | 0.36 | 0.03 |
| Triglycerides | ||||||
| Cord blood lead | 0.16 | 0.006 | 0.25 | 0.01 | 1.10 | NS |
Adjusted for age, maternal, schooling, 54.5 possessions, cohort
To generate pilot data for our P01 application for Project 2, submitted in July 2012, we also expanded our analysis to consider the role of BPA and exposure in cardio-metabolic risk in NHANES data from 2003–2009, work lead by Dr. Joyce Lee and Dr. Vasanantha Padmanabhan (who became Co-PI of the UM P01 CEHC) and their fellow, Dr. Donna Eng. This analysis evaluated cross-sectional associations between urinary BPA and multiple measures of adiposity, cholesterol, insulin and glucose for children aged 6 to 18 years, adjusting for relevant covariates (e.g., demographics, urine creatinine, tobacco exposure, soda consumption). We found a higher odds of obesity (BMI ≥ 95th percentile) with increasing quartiles of BPA for quartiles 2 versus 1 (odds ratio [OR] 1.74, 95% confidence interval [CI] 1.17–2.60, P = .008), 3 versus 1 (OR 1.64, 95% CI 1.09–2.47, P = .02), and 4 versus 1 (OR 2.01, 95% CI 1.36–2.98, P = .001). We also found a higher odds of having an abnormal waist circumference-to-height ratio (quartiles 2 vs. 1 [OR 1.37, 95% CI 0.98–1.93, P = .07], 3 vs. 1 [OR 1.41, 95% CI 1.07–1.87, P = .02], and 4 vs. 1 [OR 1.55, 95% CI 1.12–2.15, P = .01]). Although higher levels of urinary BPA were associated with a higher odds of obesity (BMI > 95%) and abnormal waist circumference-to-height ratio, longitudinal analyses are needed to disentangle temporal relationships between BPA exposure at different sensitive periods and the development of obesity and chronic disease risk factors in children.
Thus, to take advantage of the longitudinal birth cohort data from ELEMENT, we then explored the relationship of BPA and phthalates measured in third trimester urines on development of cardio-metabolic outcomes. At ISEE 2013 (Basel), as shown below, we presented pilot data showing that BPA in utero was negatively associated with leptin levels in both boys and girls. Although few longitudinal studies exist, these finding are consistent with limited data from human and animal studies that suggest BPA may be associated with a lean rather than an obese phenotype, since leptin levels will in part reflect adipose reserves. Analysis and development of a manuscript based on these data in the full P20 sample is being conducted from July to November 2014.
Table 2. Association of BPA exposure with cholesterol and leptin (mg/dL) at 8-14 yr
| Exposure timing | Overall (a=72) | Girls (a=33) | Boys (a=33) | |||
|---|---|---|---|---|---|---|
| Beta | p-value | Beta | p-value | Beta | p-value | |
| Total Cholesterol | ||||||
| 3rd trimester | 0.04 | NS | 13.1 | 0.047 | -7.3 | NS |
| 8-14 yr | -1.47 | NS | 13.1 | 0.017 | -7.8 | NS |
| LDL Cholesterol | ||||||
| 3rd Trimester | 1.78 | NS | 9.48 | (0.067) | -8.6 | NS |
| 8-13 yr | 0.04 | NS | 15.1 | 0.034 | -5.5 | NS |
| Leptin | ||||||
| 3rd trimester | -0.30 | 0.044 | -0.55 | 0.006 | -0.37 | (0.076) |
| 8-14 yr | 0.86 | 0.49 | -0.15 | 0.47 | 0.10 | 0.61 |
In addition, concurrent BPA was related with elevated total and LDL cholesterol, as shown below, suggesting greater cardio-metabolic risk in adolescent girls but not boys. In contrast, concurrent (but not in utero) levels of some phthalates were associated with lower total and LDL cholesterol in boys only. We currently are preparing manuscripts based on these pilot data to submit for publication in 2014.
Table 3. Association of phthalate exposures with cholesteron (mg/dL) at 8-14yr
| Phthalate Metabolite | Overall (n=72) | Boys (n=39) | ||
|---|---|---|---|---|
| h | p-value | h | p-value | |
| Total Cholesterol | ||||
| MEHP | 6.25 | 0.059 | -4.88 | NS |
| MEOHP | -6.47 | 0.14 | -14.75 | 0.052 |
| MEP | 2.99 | 0.18 | -8.88 | 0.02 |
| LDL Cholesterol | ||||
| MEHP | -6.11 | 0.45 | -15.2 | 0.007 |
| MEOHP | -6.89 | .087 | -14.5 | 0.038 |
| MEP | -2.67 | 0.20 | -8.60 | 0.02 |
| MBP | -5.05 | 0.13 | -9.95 | 0.03 |
Finally, combined cohort data from ELEMENT were utilized to examine other predictors of obesity in adolescence in a population undergoing the nutrition transition (e.g., dietary quality) and birth outcomes related to low birth weight (premature birth). These analyses are highly relevant to the aims of our PO1 application that examines toxicant × diet interactions in relationship to obesity and metabolic outcomes. First, to understand the role of diet during sensitive periods on children’s weight status, we used principal components analysis to construct dietary patterns and then related these to BMI at 4 years and in adolescence. We examined the relationship of diet at 4 years of age with weight status cross-sectionally and at 7–15 among 575 children in the ELEMENT cohorts. Principal components analysis of 100 FFQ items defined three dietary patterns: vegetable/fish; fruits/grains; meats/dairy/sweets. We used quantile regression to model the association of each preschool dietary factor with BMI at 10th, 25th, 50th, 75th, and 90th percentiles of the analytic sample, adjusting for child’s sex, age, birth weight, energy intake, and maternal BMI and schooling. Mean (SD) child age at follow-up was 10.0 (2.4) years and BMI 19.5 (3.9); birth weight 3.1 (0.4) kg, mother’s BMI 28.1 (4.6), and schooling 10.6 (2.9) years. The vegetable/fish pattern was associated with lower BMI at 10th (−0.51, p ≤ .0001) and 25th (−0.41, p = .003) quantiles; the meat/dairy/sweets pattern (higher in fat, protein and calcium) was associated with lower BMI at 10th (−0.33, p = .005), 25th (−0.42, p = .0004) and 50th (−0.42, p = .01) quantiles. The fruit/grains pattern (higher in carbohydrates) was related to higher adolescent BMI only at the 90th quantile (0.46, p = .049). After adjusting for energy intake, food patterns during preschool years may have differential impacts across the BMI distribution during pre-adolescence and adolescence. We presented these findings at the American Society of Nutrition and the Obesity Society meetings (Peterson, et al.) and plan to submit a manuscript for review in 2014. Further research through our P01 Project 2 will allow us to examine how changes in dietary patterns and macronutrient distribution during sensitive periods may impact development of obesity and measures of fat distribution during the pubertal transition directly and in interaction with toxicants (Pb, Cd, BPA, phthalates).
Within a developmental origins framework, we also examined the association of preterm birth (< 37 weeks gestational age) with BMI, obesity and waist circumference in adolescence. Results showed no association between preterm birth and obesity, but sex-stratified models were suggestive for increased odds of obesity in females, but decreased odds in males. Additionally, mother’s obesity was positively and significantly associated with increased odds of adolescent obesity, waist circumference and BMI. These results were presented at The Obesity Society meeting in 2012 in San Antonio, Texas (Yang, et al.). A manuscript based on these data analyses was drafted and has been submitted for review at the Journal of Developmental Origins of Health and Disease.
To address Specific Aim 3, all laboratory analyses for two studies investigating the associations between prenatal Pb exposure and cord blood DNA methylation levels were completed. In the first study, DNA isolated from 248 archived cord blood leukocyte samples from early participants of ELEMENT (“Cohort I”) was bisulfite converted. Percent methylation was quantified via pyrosequencing at repetitive elements (LINE-1) and three growth-related genes (IGF2, H19, HSD11B2). Statistical analyses relating methylation levels to three biomarkers of prenatal Pb exposure (maternal tibia and patella Pb, cord blood Pb) revealed borderline statistically significant (p < 0.1) associations between patella Pb and hypermethylation of HSD11B2 and IGF2 (Goodrich, et al.).
The second study addressing Aim 3 involves examining prenatal and early childhood Pb exposures and their impact on DNA methylation levels at birth, at a peri-adolescent time point, and on the methylation change (drift) between the two time points in the same individuals. DNA was isolated and bisulfite converted from blood leukocytes collected from 247 ELEMENT children attending a follow-up visit between the ages of 8 and 15 years. Archived cord blood leukocyte DNA was available for 79 of these children who originated from ELEMENT Cohorts 2 and 3. The pyrosequencing platform was used to quantify percent methylation at the same candidate regions selected in the first study (LINE-1, H19 and HSD11B2) on all peri-adolescent DNA samples and matching cord blood DNA samples. The Sequenom EpiTYPER platform was used to quantify methylation at IGF2.
Cross-sectional studies suggest that low-level lead exposure is associated with physician-observed onset of puberty in boys and girls, delayed age of self-reported menarche in girls, and marginally reduced testicular volume in boys. Prenatal lead exposure among rats has been shown to cause a decrease in several puberty-related hormones. Lead is consistently associated with decreased birth and infant length, which continues into early childhood as stature decrement; however, the association between lead exposure and child weight status is less consistent. This project is primed to address both the question of lead’s effect on child weight status and tempo of sexual maturation. Preliminary studies suggest that maternal lead measures are inversely associated with umbilical cord DNA methylation. Epigenetic fetal programming via DNA methylation may provide a pathway for lead exposure to influence disease susceptibility. The analysis of umbilical cord DNA methylation in Research Project subjects will provide valuable insight into a possible association between prenatal lead exposure and childhood weight gain and weight status.
Pilot Project 1 Summary
Specific Aim 1: Develop a mouse model of dose-dependent maternal lead effects on the fetal epigenome. We conducted maternal lead (Pb) exposure via water in control (0 ppm lead) and three exposure groups (2.1 ppm, 16 ppm or 32 ppm). Prior to study inception, animal water used for control animals and to make the stock Pb solutions was sent to NSF international (Ann Arbor, MI) for verification of Pb-free water. Additionally, stock Pb solutions were sent to NSF for verification of expected levels.
Animal colony expansion for this study was initiated in late 2010, and perinatal Pb exposure began in Summer 2011 and ended in Summer 2012. At 3 weeks of age, wean weight was recorded and tail tips for DNA isolation and epigenetic analyses were collected from all mice. A summary of these results is below, and we published the following article supported by Aim 1 (trainees mentored by Dr. Dolinoy are underlined):
Faulk C., Barks A., Liu K., Goodrich JM, Dolinoy DC. Early life lead exposure results in dose and sex-specific effects on weight and epigenetic gene regulation in weanling mice. Epigenomics. 5(5):487-500, 2013.
Abstract:
Aims: Epidemiological and animal data suggest that development of adult chronic conditions is influenced by early-life, exposure-induced changes to the epigenome. This study investigates the effects of perinatal lead (Pb) exposure on DNA methylation and body weight in weanling mice. Materials and Methods: Viable yellow agouti (Avy) mouse dams were exposed to 0, 2.1, 16 and 32 ppm lead acetate before conception through weaning. Epigenetic effects were evaluated by scoring coat color of Avy/a offspring and quantitative bisulfite sequencing of two retrotransposon-driven (Avy and CabpIAP) and two imprinted (Igf2 and Igf2r) loci in tail DNA. Results: Maternal blood lead levels were below the limit of detection in controls and 4.1, 25.1 and 32.1 μg/dL for each dose, respectively. Lead exposure was associated with a trend of increased wean body weight in males (p = 0.03) and altered coat color in Avy/a offspring. DNA methylation at Avy and CabpIAP was significantly different from controls following a cubic trend (p = 0.04, p = 0.01), with male-specific effects at the Avy locus. Imprinted genes did not shift in methylation across exposures. Conclusions: Dose- and sex-specific responses in body weight and DNA methylation indicate that lead acts on the epigenome in a locus-specific fashion, dependent on the genomic feature hosting the CpG site of interest, and that sex is a factor in epigenetic response.
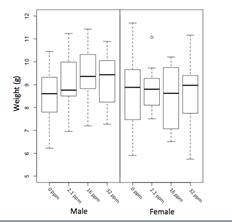
Figure 1. Day 22 weight in a/a animals across exposure groups. A linear increase in wean weight with increasing lead exposure was found in males (p = 0.04) but not females (p = 0.83) in a/a animals (n = 144). Among males, the mean weight for 0 ppm was 8.55 g (1.10 SD), the 2.1 ppm group measured 9.14 g (1.15 SD), the 16 ppm group measured 9.51 g (1.22 SD), and the 32 ppm group measured 9.22 g (1.16 SD). Among females, the mean weight for 0 ppm was 8.60 g (1.48 SD), the 2.1 ppm group measured 8.74 g (0.99 SD), the 16 ppm group measured 8.35 g (1.37 SD), and the 32 ppm group measured 8.63 g (1.45 SD).
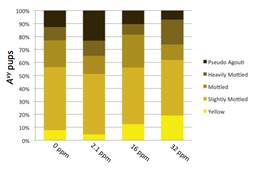
Figure 2. Coat color distribution by exposure. The percentage of animals in each coat color across 5 categories for all Avy/a animals (n = 170) separated by Pb group is shown. A significant increase in yellow offspring between control and 32 ppm exposure (p = 0.01) reflects a decrease of methylation at the Avy allele. At the 2.1 ppm concentration, the percentage of pseudoagouti mice increased from 12% to 23%, indicating an increased frequency of highly methylated Avy offspring in this group and is suggestive of a non-monotonic effect at this locus with low lead exposure.
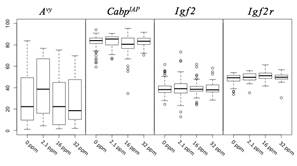
Figure 3. DNA methylation levels at metastable and imprinted loci. D22 tail tissue methylation assayed via pyrosequencing reveals a cubic trend of methylation in Avy (p = 0.04, all Avy animals) and CabpIAP (p = 0.01, all Avy/a and a/a animals) loci, resulting in an increased methylation level at the 2.1 ppm level and decreased methylation at the higher levels. The imprinted loci, Igf2 and Igf2r, do not show a significant shift in methylation levels across dosage in either linear or cubic trends for any subgroup of animals, suggesting that methylation response is dependent on the genetic features at each locus. Avy naturally exhibits a wide range of variation from near 0% to 85% in this population, and the range is slightly reduced as dosage increases. In contrast, CabpIAP has a smaller range and higher median methylation.
This work was featured on Epigenie.com, a high-readership blog about epigenetics: October 2, 2013, Epigenie, “Get the Lead Out of Your Epigenome,” http://epigenie.com/get-the-lead-out-of-your-epigenome/ Exit. As individuals age, the epigenome undergoes “epigenetic drift,” a phenomenon of increasingly divergent DNA methylation marks over the life course. Studies of aging and genetic dysregulation (e.g., cancer) support a decrease in global genomic DNA methylation (hypomethylation) concurrent with locus-specific increases in DNA methylation (hypermethylation). Consequently, epigenetic drift has emerged as a biomarker for aging, with increased DNA methylation targeting developmental genes. The preponderance of evidence to date, however, has been measured in cross-sectional samples, thereby lacking a longitudinal component evaluating DNA methylation in paired samples over time. This publication is the second in a series using a humanized mouse model to evaluate the effects of perinatal lead (Pb) exposure on life-course health outcomes.
Faulk C., Liu K., Barks A., Goodrich J.M., Dolinoy D.C. Longitudinal epigenetic drift in mice perinatally exposed to lead. Epigenetics. 9(7):934-41.
We initially found that two types of loci often used to detect environmental perturbations, transposon insertions and imprinted genes, react differently to Pb exposure, suggesting that locus-specific effects may serve as a signature of historical exposures (Epigenomics, 2013). Now, we show DNA methylation from unexposed mice increases over time at the imprinted Igf2 and Igf2r loci, but not at the imprinted H19 locus or the CabpIAP metastable epiallele. Pb exposure, however, accelerates DNA hypermethylation in the CabpIAP metastable epiallele and moderates hypermethylation in Igf2r. Thus, the presence and magnitude of epigenetic drift is locus-dependent, and enhancement of drift was mediated by perinatal Pb exposure, in some, but not all, loci. Lead is a persistent toxicant with significant effects even at blood levels consistent with the lowest recommended action level. This work indicates that epigenetic effects established in early development may persist beyond the initial exposure, and predispose individuals for disease risk. Parallel efforts in humans are evaluating whether maternal Pb exposure induces oxidative stress and adverse health outcomes via altered epigenetic programming.
Specific Aim 2: Examine the relationship of prenatal lead exposure to physiological parameters and hormonal status throughout the life-course. Of the litters generated, ~ 1 male and 1 female (a/a genotype only) were followed until 10 months of age and phenotyped at the Michigan Metabolomics and Obesity Center Animal Phenotyping Core for life-course evaluation of body composition (lean mass, fat mass and fluid), spontaneous activity, energy expenditure and food intake. These evaluations occurred at 3 time points throughout life (3, 6 and 9 months). At the 9 month visit, animals were assessed for fasting insulin and glucose levels. Only a/a offspring are followed for life-course animal phenotyping to exclude bias from metabolic disturbance induced by the Avy allele in Avy/a offspring, described elsewhere (Miltenberger, et al., 1997). These results have been written up and are currently under re-review at Environmental Health Perspectives (trainees mentored by Drs. Dolinoy, Peterson, and Sanchez are underlined):
Faulk C., Barks A., Sánchez B., Zhang Z, Anderson OS, Peterson KE, Dolinoy DC. Perinatal lead (Pb) exposure results in sex-specific effects on weight, food intake, fat, and insulin response across the murine life-course. PLoS One 2014.
Abstract:
Background: Developmental lead (Pb) exposure has been associated with lower body weight in human infants and late onset obesity in mice. Objective: We determined the association of perinatal Pb exposure in mice with changes in obesity-related phenotypes into adulthood. Methods: Mice underwent exposure via maternal drinking water supplemented with 0 (control), 2.1 (low), 16 (medium), or 32 (high) ppm Pb acetate 2 weeks prior to mating through lactation. Offspring were phenotyped at ages 3, 6 and 9 months for energy expenditure, spontaneous activity, food intake, body weight, body composition, and—at age 10 months—for glucose tolerance. Data analyses were stratified by sex and adjusted for litter effects. Results: Exposed females and males exhibited increased energy expenditure as compared to controls (p < 0.0001 for both). In females, horizontal activity differed significantly from controls (p = 0.02) over the life-course. Overall, food intake increased in exposed females and males (p < 0.0008 and p < 0.0001, respectively) with significant linear trends at 9 months in females (p = 0.01) and 6 months in males (p < 0.01). Body weight was significantly increased in males at the medium and high exposures (p = 0.004 and p = 0.029). Total body fat differed among exposed females and males (p < 0.0001 and p < 0.0001, respectively). Insulin response was significantly increased in medium exposure males (p < 0.05). Conclusions: Perinatal Pb exposure at blood lead levels between 4.1 μg/dL and 32 μg/dL is associated with increased food intake, body weight, total body fat, energy expenditure, activity and insulin response in mice. Physiological effects of developmental Pb exposure persist and vary according to sex and age.
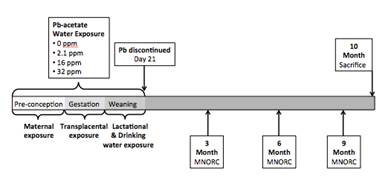
Figure 1. Lead exposure timeline. Dams were exposed 2 weeks prior to mating and throughout pregnancy. Pups continued exposure post-natally during lactation and via direct exposure in drinking water until weaning at day 22. After discontinuation of Pb exposure, mice were assayed for physiological parameters at 3, 6 and 9 months of age at the Michigan Nutrition Obesity Research Center (MNORC), and sacrificed at 10 months of age.
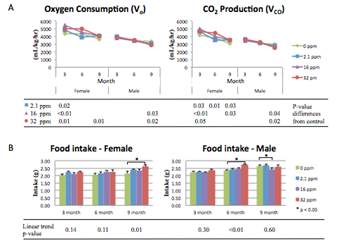
Figure 2. Energy expenditure and food intake. Energy expenditure (ml/kg/hr) measured over a 72-hr period by indirect calorimetry. (A) Oxygen consumption and carbon dioxide (CO2) production decline with age across both sexes and all exposure groups. P-values represent differences from control. (B) Food intake across the life-course, stratified by sex. Stars indicate p-values < 0.05 as compared to control. Linear trend p-values represent exposure-dependent trend significance.
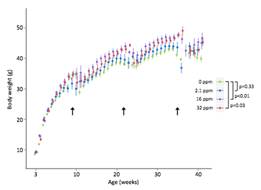
Figure 3. Weekly body weight measurements. Male offspring across all exposure groups (grams) were weighed. Both medium and high exposure groups are significantly increased in weight across the life course as compared to control mice (in green). Arrows indicate weeks when mice were transferred for physiological parameter testing resulting in stress-induced weight loss. Bars represent 95% confidence intervals. P-values were calculated using a linear mixed model with weight as a fixed effect.
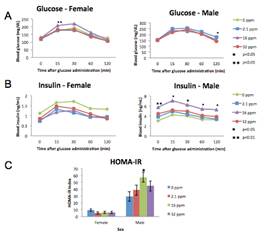
Figure 4. Blood insulin and glucose (9 months). (A) Insulin (ng/ml) is increased in males at the medium exposure and (B) glucose (mg/dl) measured by a fasting oral glucose tolerance test at 0 (baseline), 15, 30, 60 and 120 minutes shows an increase only at the medium exposure level in females and low exposure level in males at single time points. (C) Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) calculated from baseline blood insulin and glucose levels indicates an index of insulin resistance. Males at the medium exposure level show a significant increase in HOMA-IR over control. Single stars indicate p-values < 0.05, double stars indicate p-values < 0.01 when compared to controls. Bars represent standard error.
Instead of terminating the mouse BPA study at post-natal day 22, we were able to continue life-course evaluation of physiological parameters. This life course analysis was published in the following article (trainees mentored by Drs. Dolinoy, Peterson, and Sanchez are underlined):
Anderson OS., Peterson KE, Sanchez BN, Zhang Z. Mancuso PM, Dolinoy, DC. Perinatal bisphenol A exposure promotes hyperactivity, lean body composition, and hormonal responses across the murine life-course. The FASEB Journal. 27(4):1784-1792, 2013. PMCID: 3606526.
In the course of evaluating life-course effects of BPA exposure on metabolic homeostasis and hormone levels, we also observed a dose-dependent increase in liver tumors. This work was published in 2014 in Environmental Health Perspectives (trainees mentored by Dr. Dolinoy are underlined):
Weinhouse C, Anderson OS, Bergin IL, Vandenbergh DJ, Gyekis JP, Dingman MA, Yang J, Dolinoy DC. Dose-dependent incidence of hepatic tumors following perinatal bisphenol A exposure. Environmental Health Perspectives. 2014.
Significance
Using Pb and BPA as representative environmental toxicants, these mouse models of dose-dependent and human-relevant exposure levels combined with epigenetic and life-course physiological assessment significantly strengthen children’s health risk assessment and shape diagnostic and therapeutic strategies. First, the identification of epigenetic exposure biomarkers will be useful in the identification of at-risk individuals prior to disease onset. Second, unlike genetic mutations, epigenetic profiles are potentially reversible. Therefore, epigenetic approaches for prevention and treatment, such as nutritional supplementation or pharmaceutical therapy, may be developed to counteract negative epigenomic profiles. For example, if we fully determine that Pb exposure results in hypomethylation at candidate genes concomitant with changes in life-course physiological parameters, it would be important to determine if nutritional supplements (e.g., folic acid and genistein) not only during development but also later in life can protect against these negative effects on the epigenome and subsequent phenotype. Likewise, it will be important to determine if diet, including high fat diets, exacerbate the effects of perinatal chemical exposures on the epigenome and life course metabolic homeostasis. Therefore, the future of epigenomics holds tremendous potential not only for individualized children’s health, but also for population-wide disease diagnostic, screening and prevention strategies.
Pilot Project 2 Summary
Pilot Project 2 was highly successful in addressing our aims, with 16 published peer-reviewed articles or book chapters and at least 4 more in review or in preparation. We were also able to accomplish something that is likely quite rare in environmental epidemiology studies: with the leadership of the Research Project and the Mexico field team, we were actually able to recruit and obtain necessary data on more participants (N = 250) than originally proposed (N = 200). This represents a large sample size for a pilot study, which should serve us well as we continue to submit manuscripts for publication.
Addressing Specific Aim 5, we published a paper on “Predictors of urinary bisphenol A and phthalate metabolite concentrations in Mexican children” (Lewis, et al., 2013; Chemosphere, 93:2390-98). In this analysis we found that self-reported use of a number of personal care products in the 48 hours prior to urine sample collection was associated with exposure to multiple phthalates among children in the study. In boys, cologne use was associated with multiple phthalates, while in girls, the use of colored cosmetics (make-up), deodorant, conditioner and other hair care products were important for exposure. Among girls, the number of personal care products used associated with significantly increased MEP concentrations. Since exposure to phthalates in children may be associated with endocrine-related effects, reduced or delayed use of certain personal care products (e.g., cologne/perfume, colored cosmetics) in children may be warranted.
This paper also addressed Specific Aim 1, where we reported geometric mean urinary concentrations of the analytes at third trimester were comparable to those in urine samples from adults 20 years of age and older in U.S. NHANES 1999–2004, except for MBP (P20: 65 ng/mL; US: 17–22 ng/mL) and MEHP (P20: 6.7–7.0 ng/ml; US: 2.2–4.2 ng/mL), which were higher in this study, and MBZP (P20: 3.5–3.6 ng/mL; US: 8.2–9.2 ng/mL), which was lower in this study. Geometric mean urinary concentrations of the analytes at 8–13 years of age were comparable to those in urine samples from 6–11 year-old boys and girls in U.S. NHANES 2009–2010, except for MEP (P20: 70–95 ng/mL; US: 35 ng/mL), MBP (P20: 92–101 ng/mL; US: 22 ng/mL), MEHP (P20: 6.1–7.6 ng/mL; US: 1.6 ng/mL), MEHHP (P20: 45–50 ng/mL; US: 15.0 ng/mL), MEOHP (P20: 19–22 ng/mL; US: 9.8 ng/mL), and MECPP (P20: 68–77 ng/mL; US: 27.7 ng/mL), which were higher in this study, and MBZP (P20: 5.1–5.6 ng/mL; US: 11.6 ng/mL), which was lower in this study. Concentration ranges presented here for the P20 data represent geometric mean concentrations for male and female child participants.
Related to Specific Aim 3, associations between archived maternal third trimester urinary measures of BPA and nine phthalate metabolites with peripubertal (aged 8–14 years) BMI z-score, waist circumference and triceps skinfold were analyzed with continuous and quartile measures of metabolites. A cross-sectional study of adolescent urinary concentrations of BPA and phthalate metabolites with these outcome measures was also assessed. Results indicated sexually dimorphic non-monotonic relationships between both maternal and peripubertal urinary metabolites with peripubertal measures. Additionally, direction of association with outcomes differed by timing of urinary measures and significant findings were negative and occurred with second or third quartiles of metabolites. These results were presented at the International Society for Environmental Epidemiology 2013 in Basel, Switzerland (Yang, et al.). The manuscript based on these data, entitled “Bisphenol A and phthalates urinary concentrations in utero and in adolescence: associations with weight status and adiposity differ by sex” (Yang, et al.) has been completed and is being reviewed at the journal Environmental Health Perspectives.
Also related to Aim 3, we examined the association of BPA and phthalates with BMI and height trajectories from early childhood to adolescence in the P20 sample. Using fractional age polynomials and mixed effects models, BMI and height trajectories were fit separately for males and females for each tertile of maternal third trimester urinary measures (BPA and nine phthalate metabolites). Findings showed that growth trajectories of boys and girls were associated with different metabolites. The highest predicted BMI and height trajectories for females occurred with the third tertile of phthalate metabolites whereas the highest predicted trajectories occurred in males with the first or second tertiles of phthalate metabolites. Findings for BPA were inverted when compared to phthalate metabolites, with highest predicted height trajectory occurring with the first tertile of BPA in females and third tertile in males. The manuscript “Differences in BMI and height trajectories of males and females associated with in utero urinary concentrations of bisphenol A and phthalate metabolites” (Yang, et al.) has been completed and will be submitted to American Journal of Epidemiology in July 2014 following sign-off by all co-authors.
Relevant to Specific Aim 4, presented in August 2013 (Basel Switzerland) and highlighted in the December 2013 EPA/NIEHS Children’s Center Webinar series, we also have some very compelling new data from our analysis of associations between in utero or peripubertal exposure to EDCs and sexual maturation. Among boys (See Tables 1 and 2 for selected results), we found that in utero exposure to certain phthalates was associated with reduced odds of pubertal onset (pubarche and gonadarche), and increased levels of serum SHBG. While childhood exposure to phthalates was not associated with pubertal onset, DEHP metabolites were associated with decreased free T (and increased SHBG). Overall, findings appear to be consistent with anti-androgenic properties of multiple phthalates in males reported in animal and very limited human studies to date. This work has been described in a paper, “Prenatal and peripubertal phthalates and bisphenol A in relation to sex hormones and puberty in boys” (Ferguson, et al., 2014), published in June 2014 in the journal Reproductive Toxicology.
Table 1. Adjusted odds ratios for pubertal onset among 115 boys for an interquartile range (IQR) increase in
Journal Articles: 28 Displayed | Download in RIS Format
| Other center views: | All 72 publications | 33 publications in selected types | All 28 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Afeiche M, Peterson KE, Sanchez BN, Cantonwine D, Lamadrid-Figueroa H, Schnaas L, Ettinger AS, Hernandez-Avila M, Hu H, Tellez-Rojo MM. Prenatal lead exposure and weight of 0- to 5-year-old children in Mexico City. Environmental Health Perspectives 2011;119(10):1436-1441. |
R834800 (2011) R834800 (2012) |
Exit |
|
|
Afeiche M, Peterson KE, Sanchez BN, Schnaas L, Cantonwine D, Ettinger AS, Solano-Gonzalez M, Hernandez-Avila M, Hu H, Tellez-Rojo MM. Windows of lead exposure sensitivity, attained height, and body mass index at 48 months. The Journal of Pediatrics 2012;160(6):1044-1049. |
R834800 (2012) R834800 (2013) R831725 (Final) |
Exit |
|
|
Anderson OS, Sant KE, Dolinoy DC. Nutrition and epigenetics:an interplay of dietary methyl donors, one-carbon metabolism and DNA methylation. The Journal of Nutritional Biochemistry 2012;23(8):853-859. |
R834800 (2012) R834800 (2013) |
Exit Exit Exit |
|
|
Anderson OS, Nahar MS, Faulk C, Jones T, Liao C, Kannan K, Weinhouse C, Rozek RS, Dolinoy DC. Epigenetic responses following maternal dietary exposure to physiologically relevant levels of bisphenol A. Environmental and Molecular Mutagenesis 2012;53(5):334-342. |
R834800 (2012) |
Exit Exit |
|
|
Anderson OS, Peterson KE, Sanchez BN, Zhang Z, Mancuso P, Dolinoy DC. Perinatal bisphenol A exposure promotes hyperactivity, lean body composition, and hormonal responses across the murine life course. The FASEB Journal 2013;27(4):1784-1792. |
R834800 (2012) R834800 (2013) |
Exit Exit Exit |
|
|
Cantonwine D, Meeker JD, Hu H, Sanchez BN, Lamadrid-Figueroa H, Mercado-Garcia A, Fortenberry GZ, Calafat AM, Tellez-Rojo MM. Bisphenol A exposure in Mexico City and risk of prematurity:a pilot nested case control study. Environmental Health 2010;9:62. |
R834800 (2011) R834800 (2012) R831725 (Final) |
Exit Exit Exit |
|
|
Dolinoy DC, Faulk C. Introduction: the use of animals models to advance epigenetic science. ILAR Journal 2012;53(3-4):227-231. |
R834800 (2013) |
Exit Exit |
|
|
Eng DS, Lee JM, Gebremariam A, Meeker JD, Peterson K, Padmanabhan V. Bisphenol A and chronic disease risk factors in US children. Pediatrics 2013;132(3):e637-e645. |
R834800 (2013) |
Exit Exit |
|
|
Faulk C, Dolinoy DC. Timing is everything: the when and how of environmentally induced changes in the epigenome of animals. Epigenetics 2011;6(7):791-797. |
R834800 (2011) R834800 (2012) R834800 (2013) |
Exit Exit Exit |
|
|
Faulk C, Barks A, Dolinoy DC. Phylogenetic and DNA methylation analysis reveal novel regions of variable methylation in the mouse IAP class of transposons. BMC Genomics 2013;14:48. |
R834800 (2013) |
Exit Exit Exit |
|
|
Faulk C, Barks A, Liu K, Goodrich JM, Dolinoy DC. Early-life lead exposure results in dose- and sex-specific effects on weight and epigenetic gene regulation in weanling mice. Epigenomics 2013;5(5):487-500. |
R834800 (2013) |
Exit |
|
|
Faulk C, Barks A, Sanchez BN, Zhang Z, Anderson OS, Peterson KE, Dolinoy DC. Perinatal lead (Pb) exposure results in sex-specific effects on food intake, fat, weight, and insulin response across the murine life-course. PLoS One 2014;9(8):e104273. |
R834800 (2013) |
Exit Exit Exit |
|
|
Ferguson KK, Loch-Caruso R, Meeker JD. Urinary phthalate metabolites in relation to biomarkers of inflammation and oxidative stress: NHANES 1999-2006. Environmental Research 2011;111(5):718-726. |
R834800 (2011) R834800 (2012) |
Exit Exit Exit |
|
|
Ferguson KK, Loch-Caruso R, Meeker JD. Exploration of oxidative stress and inflammatory markers in relation to urinary phthalate metabolites: NHANES 1999-2006. Environmental Science & Technology 2012; 46(1):477-485. |
R834800 (2011) R834800 (2012) |
Exit Exit |
|
|
Ferguson KK, Hauser R, Altshul L, Meeker JD. Serum concentrations of p,p’-DDE, HCB, PCBs and reproductive hormones among men of reproductive age. Reproductive Toxicology 2012;34(3):429-435. |
R834800 (2013) |
Exit Exit |
|
|
Fortenberry GZ, Hu H, Turyk M, Barr DB, Meeker JD. Association between urinary 3,5,6-trichloro-2-pyridinol, a metabolite of chlorpyrifos and chlorpyrifos-methyl, and serum T4 and TSH in NHANES 1999-2002. Science of the Total Environment 2012;424:351-355. |
R834800 (2013) |
Exit Exit |
|
|
Fortenberry GZ, Meeker JD, Sanchez BN, Barr DB, Panuwet P, Bellinger D, Schnaas L, Solano-Gonzalez M, Ettinger AS, Hernandez-Avila M, Hu H, Tellez-Rojo MM. Urinary 3,5,6-trichloro-2-pyridinol (TCPY) in pregnant women from Mexico City:distribution, temporal variability, and relationship with child attention and hyperactivity. International Journal of Hygiene and Environmental Health 2014; 217(2-3):405-412. |
R834800 (2013) R835436 (2014) R835436 (2015) R835436 (2017) |
Exit Exit Exit |
|
|
Kordas K, Ettinger AS, Bellinger DC, Schnaas L, Téllez Rojo MM, Hernández-Avila M, Hu H, Wright RO. A dopamine receptor (DRD2) but not dopamine transporter (DAT1) gene polymorphism is associated with neurocognitive development of Mexican preschool children with lead exposure. The Journal of Pediatrics 2011;159(4):638-643. |
R834800 (2011) R834800 (2012) |
Exit Exit Exit |
|
|
Lamadrid-Figueroa H, Téllez-Rojo MM, Angeles G, Hernández-Ávila M, Hu H. Bias correction by use of errors-in-variables regression models in studies with K-X-ray fluorescence bone lead measurements.Environmental Research 2011;111(1):17-20. |
R834800 (2011) R834800 (2012) |
Exit Exit Exit |
|
|
Lewis RC, Meeker JD, Peterson KE, Lee JM, Pace GG, Cantoral A, Tellez-Rojo MM. Predictors of urinary bisphenol A and phthalate metabolite concentrations in Mexican children. Chemosphere 2013;93(10):2390-2398. |
R834800 (2013) |
Exit Exit |
|
|
Meeker JD, Ferguson KK. Relationship between urinary phthalate and bisphenol A concentrations and serum thyroid measures in U.S. adults and adolescents from National Health and Nutrition Examination Survey (NHANES) 2007-2008. Environmental Health Perspectives 2011;119(10):1396-1402. |
R834800 (2011) R834800 (2012) |
Exit |
|
|
Meeker JD, Calafat AM, Hauser R. Urinary phthalate metabolites and their biotransformation products: predictors and temporal variability among men and women. Journal of Exposure Science and Environmental Epidemiology 2012; 22(4):376-386. |
R834800 (2012) |
Exit |
|
|
Meeker JD. Exposure to environmental endocrine disruptors and child development. Archives of Pediatrics and Adolescent Medicine 2012;166(10):952-958. |
R834800 (2012) R834800 (2013) |
Exit Exit Exit |
|
|
Sánchez BN, Hu H, Litman HJ, Téllez-Rojo MM. Statistical methods to study timing of vulnerability with sparsely sampled data on environmental toxicants. Environmental Health Perspectives 2011;119(3):409-415. |
R834800 (2011) R834800 (2012) |
Exit |
|
|
Tellez-Rojo MM, Cantoral A, Cantonwine DE, Schnaas L, Peterson K, Hu H, Meeker JD. Prenatal urinary phthalate metabolites levels and neurodevelopment in children at two and three years of age. Science of the Total Environment 2013;461-462:386-390. |
R834800 (2013) |
Exit Exit |
|
|
Weinhouse C, Anderson OS, Bergin IL, Vandenbergh DJ, Gyekis JP, Dingman MA, Yang J, Dolinoy DC. Dose-dependent incidence of hepatic tumors in adult mice following perinatal exposure to bisphenol A. Environmental Health Perspectives 2014;122(5):485-491. |
R834800 (2013) |
|
|
|
Wickerham EL, Lozoff B, Shao J, Kaciroti N, Xia Y, Meeker JD. Reduced birth weight in relation to pesticide mixtures detected in cord blood of full-term infants. Environment International 2012;47:80-85. |
R834800 (2012) |
Exit Exit Exit |
|
|
Zhang A, Hu H, Sánchez BN, Ettinger AS, Park SK, Cantonwine D, Schnaas L, Wright RO, Lamadrid-Figueroa H, Tellez-Rojo MM. Association between prenatal lead exposure and blood pressure in children. Environmental Health Perspectives 2011;120(3):445-450. |
R834800 (2011) R834800 (2012) |
Exit |
Supplemental Keywords:
Children’s health, human health, health effects, children’s environmental health, prenatal exposure, environmental risks, exposure assessment, epigenetics, maternal exposure, Mexican, lead, bisphenol a, phthalates, animal exposure, agouti mouse, animal phenotyping, child weight, BMI, sexual maturation, Health, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, HUMAN HEALTH, Environmental Policy, Biology, Exposure, Children's Health, Biochemistry, dietary exposure, childhood obesity, epigenetics, age-related differences, environmental risks, perinatal exposure, abnormal sexual maturation, biological response, dietary factors, genetic susceptibility, assessment of exposure, developmental disordersRelevant Websites:
University of Michigan Children's Environmental Health and Disease Prevention Center Exit
Progress and Final Reports:
Original Abstract Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834800C001 In Utero Lead Exposure: Fetal Epigenetics and Life-Course Physiologic Effects
R834800C002 Impacts of Life-stage Exposures to BPA and Phthalates on Growth and Development
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.