Grantee Research Project Results
Final Report: Mechanisms of In Utero BPA Exposure on Fetal Gonad Development
EPA Grant Number: R834593C003Subproject: this is subproject number 003 , established and managed by the Center Director under grant R834593
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Water Innovation Network for Sustainable Small Systems
Center Director: Reckhow, David A.
Title: Mechanisms of In Utero BPA Exposure on Fetal Gonad Development
Investigators: Flaws, Jodi
Institution: University of Illinois Urbana-Champaign
EPA Project Officer: Hahn, Intaek
Project Period: February 15, 2010 through February 14, 2014
RFA: Children's Environmental Health and Disease Prevention Research Centers: Formative Centers (with NIEHS) (2009) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
Project 3 was designed to test the hypothesis that in utero exposure to bisphenol A (BPA) causes gonadal defects via estrogen receptor alpha (ERα). To test this hypothesis, we proposed the following specific aims: 1) determine whether loss of ERα renders developing gonads insensitive to deleterious effects of BPA and 2) determine if overexpression of ERα increases the susceptibility of embryos to BPA. We did not change the hypothesis or specific aims and completed the proposed work as detailed below.
Summary/Accomplishments (Outputs/Outcomes):
Data Obtained During Completion of Specific Aim 1
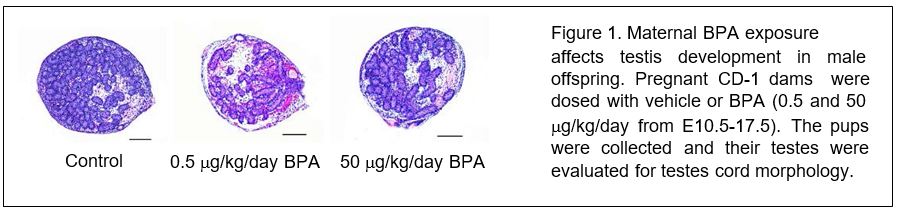
Embryonic BPA exposure adversely affects the development of the testes and ovary in the offspring of mice. Before we could properly design experiments to determine if maternal BPA exposure exerts its toxicity through ERα pathways in gonads of offspring, we needed to determine whether maternal exposure to BPA affects testis or ovarian development in mice. Pregnant CD-1 dams were dosed orally with tocopherol-stripped corn oil (vehicle) or BPA from E10.5-E17.5. This time period covers sex determination, testis cord organization/expansion and ovarian formation. Pups were collected via Caesarean section at E19.0. All pups were counted, weighed, and subjected to measurements of anogenital distance (AGD). The gonads were subjected to histological evaluations.
In utero exposure to BPA did not alter litter size or AGD compared to controls. Histological analysis revealed abnormal testis morphology in 80% of testes exposed to 0.5 µg/kg/day of BPA and 50% of testes in the 50 µg/kg/day BPA treatment groups (Figure 1). Dysgenic BPA-exposed testes contained fewer testis cord cross-sections and increased interstitial area compared to vehicle controls. Underdevelopment of the testis cords was particularly evident near the rete testes, where uncoiled lengths of testis cord appeared as finger-like projections. Interestingly, this histological abnormality is reminiscent of that observed during testicular dysgenesis syndrome. Collectively, these data indicate that maternal BPA exposure alters testis development in the offspring, and that the effects of BPA are likely non-monotonic because the low dose of BPA exerts a more dramatic effect than the high dose of BPA.
Ovaries from BPA exposed mice were less cellular with more dense and congested vascularization than ovaries from vehicle exposed mice (Figure 2). The percentage of abnormal primordial follicles in the BPA exposed ovaries was significantly increased compared to the vehicle control (vehicle control = 16.19 ± 1.99 %; BPA 0.5 µg/kg/day = 36 ± 3.36%; BPA 50 µg/kg/day = 27.55 ± 1.65%, n = 3, p < 0.05). Collectively, these data indicate that prenatal BPA exposure reduces healthy primordial follicle numbers. This is of concern because reduced numbers of healthy primordial follicles can lead to subfertility and/or premature reproductive senescence.
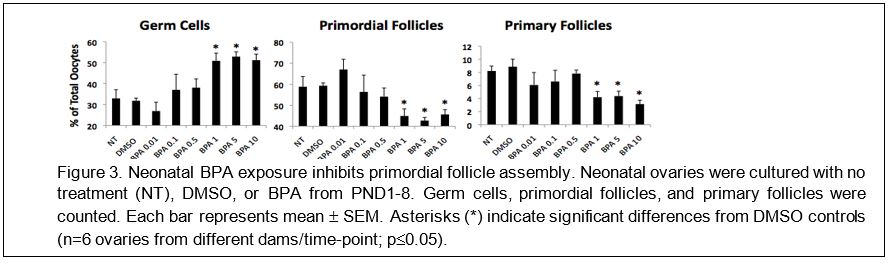
Neonatal BPA exposure adversely affects the development of the ovary. Next, we examined whether neonatal BPA exposure alters the developing ovary. This was important to do because the mouse ovary continues to form primordial follicles during the early neonatal period (PND1-8). Neonatal ovaries were collected, individually cultured in media ± dimethylsulfoxide (DMSO; vehicle) or BPA at 0.01-10 µg/ml for 8 days, and morphologically examined. This system best allows us to test the direct effects of BPA on two critical processes in ovarian development: germ cell nest breakdown and primordial follicle formation. Ovaries treated with BPA (1-10 µg/ml) had more germ cells in nests, and fewer primordial and primary follicles compared to control (Figure 3). This suggests that neonatal BPA exposure interferes with germ cell nest breakdown and inhibits primordial follicle assembly. This is of concern because inhibition of primordial follicle assembly can lead to subfertility.
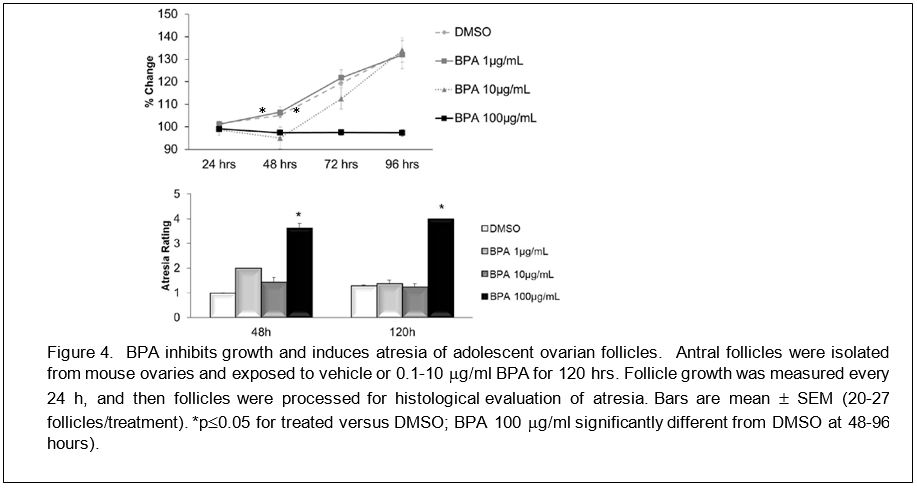
Adolescent BPA exposure adversely affects developing antral follicles in the ovary. We used a validated follicle culture system to test whether BPA exposure directly causes toxicity to adolescent ovarian follicles. Antral follicles (10-15 follicles/ovary) from adolescent ovaries were cultured in media containing DMSO or BPA for 96-120 hours. Every 24 hours, we measured follicular growth. After 96-120 hours, follicles were processed for histological evaluation of atresia. Antral follicles treated with vehicle remained viable and grew for the entire culture period. Antral follicles treated with BPA (100 µg/ml) did not grow, and instead had an increased incidence of atresia (Figure 4), suggesting that BPA inhibits growth and induces atresia of adolescent antral follicles.
After the cultures, media were subjected to enzyme-linked immunosorbent assays for hormones in the estradiol biosynthesis pathway, and follicles were processed for quantitative real-time polymerase chain reaction of steroidogenic enzymes. The results indicate that BPA (100 µg/ml) inhibits follicle growth and that pregnenolone co-treatment was unable to restore/maintain growth. Further, BPA 10 and 100 mg/ml inhibit progesterone, dehydroepiandrosterone, androstenedione, estrone, testosterone, and estradiol production. Pregnenolone co-treatment was able to increase production of pregnenolone, progesterone, and dehydroepiandrosterone and maintain androstenedione and estrone levels in BPA treated follicles compared to DMSO controls, but was unable to protect testosterone or estradiol levels. Further, pregnenolone was unable to protect follicles from BPA (10-100 µg/ml) induced inhibition of steroidogenic enzymes compared to the DMSO control. Collectively, these data show that BPA targets the estradiol biosynthesis pathway in the ovary.
ERα antagonism does not make gonads insensitive to BPA (Specific Aim 1 from P20 grant). To determine whether loss of ERα renders gonads insensitive to BPA during adolescence, we co-treated adolescent antral follicles with BPA and estradiol (E2) to examine if E2 binding to ERs blocks BPA-induced growth inhibition. Exposure to BPA (100 µg/ml) decreased follicle growth compared to DMSO and E2 controls (Figure 5A). E2 co-treatment did not protect antral follicles from BPA-induced inhibition of growth (Figure 5A). Further, we pre-treated antral follicles with the ER antagonist ICI to examine if ICI blocks BPA-induced growth inhibition. Exposure to BPA (100 µg/ml) decreased follicle growth compared to DMSO and ICI controls (Figure 5B). Pre-treatment with ICI did not protect antral follicles from BPA-induced inhibition of growth (Figure 5B). These data suggest that BPA may not exert toxicity in ovaries through ER pathways.
Overexpression of ERs does not make gonads more sensitive to BPA (Specific Aim 2 from P20 grant). We tested the hypothesis that ERα overexpression increases the susceptibility of pups to BPA. Pregnant control and ERα overexpressing dams were orally dosed with tocopherol-stripped corn oil (vehicle) or BPA from E10.5 to birth. On PND0, pup weight was decreased in the pups from BPA 0.5 µg/kg/day treated ERα overexpressing dams compared to BPA 0.5 µg/kg/day treated control dams. AGDs of males from BPA 50 µg/kg/day treated ERα overexpressing dams were smaller than pups from BPA 50 µg/kg/day treated control dams. In female pups from control mice, BPA 50 µg/kg/day significantly increased the numbers of dying germ cells compared to vehicle controls. In female pups from ERα overexpressing dams, BPA did not affect germ cells and primordial follicles compared to vehicle. BPA did not alter testes cord formation in pups from ERα overexpressing dams. These data suggest that ERα overexpressors may be more sensitive than control mice to BPA-induced changes in pup weight and AGD, but not in gonadal parameters.
To further determine if ERα overexpression makes the ovary more sensitive to BPA, we treated ERα overexpressing and control antral follicles from adolescent mice with vehicle or BPA. BPA (100 µg/ml) decreased follicle growth compared to DMSO in control and ERα overexpressing follicles from 72-120 hrs. BPA (1-10 µg/ml) did not affect growth in either control or ERα overexpressing follicles. Collectively, these data suggest that overexpression of ERα does not render antral follicles more sensitive to BPA than controls.
Journal Articles:
No journal articles submitted with this report: View all 3 publications for this subprojectSupplemental Keywords:
Health, RFA, Scientific Discipline, Health Risk Assessment, Risk Assessments, Environmental Chemistry, Children's Health, Biochemistry, biological markers, children's vulnerablity, human health riskProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R834593 Water Innovation Network for Sustainable Small Systems Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834593C001 Prenatal Exposure to BPA/Phthalates: Infant Physical and Behavioral Development
R834593C002 Adolescent Exposure to BPA/Phthalates Cognitive and Behavioral Development
R834593C003 Mechanisms of In Utero BPA Exposure on Fetal Gonad Development
R834593C004 Effects of Bisphenol A on the Developing Cortex
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
3 journal articles for this subproject
Main Center: R834593
35 publications for this center
9 journal articles for this center