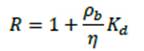Grantee Research Project Results
Final Report: Use of Bone Char for the Removal of Arsenic and Uranium from Groundwater at the Pine Ridge Reservation
EPA Grant Number: SU834713Title: Use of Bone Char for the Removal of Arsenic and Uranium from Groundwater at the Pine Ridge Reservation
Investigators: Werth, Charles J , Llewellyn, Alex , Parker, Kimberly M , Salvatore, Michelle , Becraft, Jacob , Genchanok, Yana , Dam, Emily Van , Freeck, Jason , Miller, Adam , Wang, Hanting , Nell, Marika , Feeney, Connor , Morton, Jeremy , Steege, Eden , Nguyen, Tien-Hung
Institution: University of Illinois Urbana-Champaign , Oglala Lakota College
EPA Project Officer: Page, Angela
Phase: I
Project Period: August 15, 2010 through August 14, 2011
Project Amount: $10,000
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet (2010) RFA Text | Recipients Lists
Research Category: Pollution Prevention/Sustainable Development , P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources , P3 Challenge Area - Chemical Safety , Sustainable and Healthy Communities
Objective:
The Pine Ridge Reservation in South Dakota is the poorest reservation in the country. While some homes on the reservation receive treated municipal water, many residents rely on private wells. A U. S. Geological Survey (USGS) report in the 1990s revealed that much of the groundwater on the reservation contains arsenic (As) and uranium (U) above Environmental Protection Agency (EPA) maximum contaminant limits (MCLs).i Acting on this information, members of the Oglala Lakota College (OLC) contacted The Center of Advanced Materials for the Purification of Water with Systems (WaterCAMPWS) at the University of Illinois Urbana-Champaign (UIUC). An undergraduate student team was subsequently formed to address the problem and verified this concern during a trip to the Reservation in August 2009. A variety of methods are available to remove these metals from drinking water, but most are relatively expensive, require an expert to maintain, or do not make use of indigenous materials. Absorbent biomaterials, such as bone char, present a novel and sustainable approach to remove As and U from drinking water.
A UIUC team arrived at the reservation in November 2010 for a second assessment in order to collect a larger quantity of the contaminated water for testing and to expand the set of sampled sources. To date, 35% of the private wells sampled contain As above the EPA maximum contaminant level (MCL) of 10 parts per billion (ppb), and 6% contain U above the EPA MCL of 30 ppb. This data, in conjunction with the USGS report, indicate that As and U contamination is a widespread and reoccurring problem in the Pine Ridge Reservation.
Figure 1: (Top) An OLC student, (Left) a UIUC student, and (Right) a local resident at sampled sources
The objective of this work was to develop a bone char filter that can be used to efficiently remove As and U from groundwater at the Pine Ridge Reservation. While bone char has been shown to efficiently adsorb As and U from water, the efficiency of removal depends on local water conditions and bone char characteristics. For Phase I, the goals of the group were to 1) evaluate As and U contamination in the Pine Ridge Reservation, 2) characterize bone char, 3) assess U and As removal kinetics using actual bone char, and 4) develop a bone char filtration device. These initial steps are required prior to implementation. This report presents research in all four areas of interest, and begins to assess quantitatively and qualitatively the advantages and disadvantages of implementing a bone char filtration device.
Summary/Accomplishments (Outputs/Outcomes):
Bone Char Characterization
Effect of Particle Size on Removal Kinetics
Bone char samples were made at five charring conditions: 500°C for 7 hours, 21 hours and 63 hours, and 700°C for 7 hours and 21 hours. Each char was sieved to two separate samples of particle sizes of <300 μm and 300-2000 μm, and tested on both actual groundwater and synthetic water to determine which particle size achieved optimum removal kinetics. Results indicated that there was no significant difference in U removal kinetics with respect to particle size. Removal kinetics were slightly faster for the <300 μm particle size in the synthetic As(V) solution, but the 300-2000 μm bone char is preferable due to a smaller pressure drop across the filter.
Surface Area Measurements
The BET surface area was measured using a Micrometric ASAP 2010 instrument for each char with particle sizes of 300-2000. It was observed that increased charring results in decreased surface area. Bone char is reported in literature to have a surface area of 76 m2/g, which is consistent with both the 500°C 7-hour and 21-hour data of 80 and 76 m2/g, respectively.
Effect of Charring Conditions on Removal Kinetics
Each char was tested in the Pine Ridge Reservation groundwater to see if a trend was established for removal kinetics, and to determine an optimum char to be used as an absorbent for the proposed filter. Optimum results for U removal were achieved with the bone charred at 500°C for 7 hours and 21 hours with 98% and 99% removal, respectively, after 24 hours. No As removal was measured in the Pine Ridge Reservation groundwater, suggesting that the primary As contaminant is the As(III) species. Therefore, removal kinetics were measured using synthetic As(V) solution. It will be necessary to oxidize As(III) in the Pine Ridge Reservation Groundwater. The bone charred at 700ºC for 21 hours achieved the best results at 10 hours with 87% As(V) removal, contrary to trend for U removal.
Oxidation of As(III) to As(V)
Preliminary testing is underway to determine the optimum method for oxidizing As(III) to As(V) in the Pine Ridge Reservation groundwater. The first study involved adding 1 mg/L calcium hypochlorite to the groundwater and testing the removal kinetics using bone charred at 500°C for 7 hours. After 13 hours, 17% As was removed. Removal is measurably better than the groundwater without calcium hypochlorite, but it still not as effective as the removal of 26% from the synthetic As(V) solution using the same bone char. Further experimentation is required to investigate methods to increase removal capacity and kinetics.
Calculation of Retardation Factor for As and U Removal
In order to determine the theoretical design specifications, the retardation factor, or "R-value", had to be defined. This value is used calculate the theoretical amount of water a system can treat. For a filtration system, this is defined as:
Where η is the porosity, ρb is the bulk density of bone char, and Kd is the volume of water that can be treated per unit mass of bone char. R-values were calculated for As and U to be 870 and 2270, respectively.
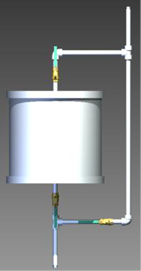
Preliminary U Filter Design
The design of a filter was primarily based on three criteria: average drinking water usage, bone char removal kinetics and capacity, and the availability of components for design. For the preliminary filter design, it was decided that 12 inch diameter by 12 inch length PVC piping would be used to house the bone char (see Figure 2). Theoretically, 87% of U is removed from the water which is sufficient to bring the contaminant level below the MCL. Using the calculated retardation factor of 2270, the length of time before bone char reaches its capacity is 318 days.
Figure 2: Preliminary bone char filter design
Life Cycle Analysis
In order to quantitatively analyze the environmental impact the filter has, a life cycle analysis (LCA) was completed on the preliminary bone char filter design relative to a reverse osmosis (RO) filter. Though initially the RO filter has a lower impact in several categories, the need to frequently replace the membrane for the RO filter negatively impacts its sustainability in the long run. After 5 years, the LCA suggests that the bone char filter proves to be the more responsible choice in the majority of categories. This trend continues more dramatically in studies of 10 and 15 years.
Conclusions:
The team investigated and reported novel research on bone char removal kinetics of As and U in the parts per billion range with the aid of an ICP-MS. Although research has been conducted on U and As removal using bone char, past experiments were performed at U and As concentrations far exceeding those commonly measured in groundwater, i.e. 50-600. In addition, the particle size, surface area, and charring conditions of bone char were characterized based on removal kinetics of both As and U. Finally, all experiments were conducted with both synthetic water and actual groundwater obtained from the Pine Ridge Reservation. To the team’s knowledge, this is the first research to ever do such tests on actual groundwater.
The activities for Phase I were completed in order to successfully achieve the four objectives identified earlier. With these goals completed, the next phase of the project can focus on optimization, implementation, business development, and education.
References:
Heakin, A.J. Water Quality of Springs and Public-Supply Well, Pine Ridge Indian Reservation, South Dakota, 1992-1997. U. S. Geological Survey
Walker, G. M., and L. R. Weatherley. "Adsorption of Dyes from Aqueous Solution — the Effect of Adsorbent Pore Size Distribution and Dye Aggregation." Chemical Engineering Journal83 (2001): 201-06. Print.
Chen, Y., Chai, L., Shu, Y. (2008). Study of arsenic(V) adsorption on bone char from aqueous solution. Journal of Hazardous Materials, 160, 168-172.
Jeanjean, J., Rouchaud, J.C., Tran, L., et al. (1995). Sorption of uranium and other heavy metals on hydroapatite. Journal of Radioanalytical and Nuclear Chemistry, 201 (6), 529-539.
Gaioli, Marisa, Daniel E. Gonzalez, and Diego Amoedo. "Hidroarsenisismo Cronico Regional Endemico: Un Desafio Diagnostico Y De Prevencion." Archivos Argentinos De Pediatría 107.5 (2009): 467-73
Schwartz,, Stephanie M. "Life and Conditions on the Pine Ridge Oglala Lakota (Sioux) Reservation of South Dakota." Link Center Foundation. 2009. Web. 13 Mar. 2011.
Druwe, Ingrid L., and Richard R. Vaillancourt. "Influence of Arsenate and Arsenite on Signal Transduction Pathways: an Update." Archives of Toxicology 84.8 (2010): 585-96.
Skipton, Sharon O., Bruce I. Dvorak, Fred Baumert, and Wayne Woldt. "Drinking Water: Uranium." Neb Guide: University of Nebraska Lincoln (2008)
Soghoian, MD, Samara. "Toxicity, Heavy Metals." Natural Russia. 28 June 2006. Web. 10 Mar. 2011.
Journal Articles:
No journal articles submitted with this report: View all 4 publications for this projectSupplemental Keywords:
bone char, hydroxylapatite, hydroxyapatite, arsenic, uranium, human health, Oglala Lakota, Pine Ridge ReservationP3 Phase II:
Use of Bone Char for the Removal of Arsenic and Uranium from Groundwater at the Pine Ridge Reservation | 2012 Progress Report | 2013 Progress Report | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.