Grantee Research Project Results
Final Report: Novel Reactor Design for Biodiesel Production
EPA Grant Number: SU834015Title: Novel Reactor Design for Biodiesel Production
Investigators: Cairncross, Richard A. , Cernansky, Nicholas P.
Institution: Drexel University
EPA Project Officer: Page, Angela
Phase: II
Project Period: August 15, 2008 through August 14, 2010
Project Amount: $74,960
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet - Phase 2 (2008) Recipients Lists
Research Category: Pollution Prevention/Sustainable Development , P3 Challenge Area - Air Quality , P3 Challenge Area - Chemical Safety , P3 Awards , Sustainable and Healthy Communities
Objective:
This project is closely allied to the People, Prosperity, and Planet (P3) principles proposed by the EPA. Rather than converting high-value food crops to biofuels, the reactor developed in this project converts low-value waste oils into biodiesel – so it benefits the Planet by reducing a waste stream and by avoiding land-use changes that have been associated with biofuels. The reactor is robust for a variety of feedstocks and should be applicable at both the small-scale and large-scale – so it benefits People by enabling more efficient conversion of locally available waste greases and plant oils into biofuels. Feasibility studies conducted during this project show that the reactor design will compete economically with traditional biodiesel production and be less sensitive to changes in feedstock prices – so it benefits Prosperity.
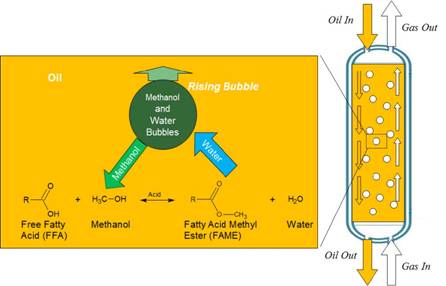
SU834015 – Schematic of a Bubble Column Reactor with insert showing the primary chemical reaction and mechanism by which the bubbles supply methanol and remove by-product water.
Biodiesel is an alternative fuel that can be produced from a wide variety of plant oils, animal oils and waste oils. The reactor studied in this project is especially suited for low-value feedstock oils that contain high concentrations of free fatty acids (FFA), in contrast to refined vegetable oils that are nearly pure triglycerides. Many waste oils and alternative un-refined (crude) plant oils have high FFA content, which leads to excessive soap production and low conversion to biodiesel using the conventional base-catalyzed reactions. The novel reactor in this project (shown schematically in Figure 1) uses acid catalysts that do not produce soaps and runs at temperatures above the boiling point of methanol – so methanol bubbles rise through a column of oil and react with the oil to produce biodiesel. The bubbling methanol provides agitation and removes the by-product water, enabling higher overall conversion to biodiesel.
Objectives and Strategies:
The following is the list of project objectives along with a brief evaluation of the achievements related to each objective:
1. Use prototype reactor to measure reaction rate constants and mass transfer coefficients for water and methanol. Reaction rates were measured for the bubble column reactor under a variety of conditions demonstrating the robustness of the reactor for a variety of feedstocks. Mass transfer coefficients were estimated using the mathematical model and showed that under normal reaction conditions, the reaction kinetics dominate over mass transfer resistances.
2. Develop a detailed mathematical model of the bubble reactor. The mathematical model accurately represents conversion versus time data from the bubble column reactor. A single set of kinetic and mass transfer constants can predict the conversion data for experiments with varying amounts of water in the methanol feed (from 0-20% water by volume).
3. Conduct feasibility studies using mathematical model and additional process data for large-scale and small-scale production. Several Chemical Engineering senior design teams conducted process feasibility studies to evaluate the economic profitability of the bubble column reactor in comparison to other technologies for converting waste oils to biodiesel. In addition, sustainability metrics were used to evaluate environmental impacts. In these studies the bubble column reactor was moderately better than the other technologies for both economic and environmental impacts.
4. Use bubble reactor model to determine appropriate scaling parameters for bubble column reactor and construct and test a pilot-scale reactor. While a truly “pilot-scale” reactor was not constructed, laboratory reactors were constructed with a range of sizes and with the ability to perform both batch and continuous reactions. The reaction experiments identified key features relevant for scale-up of the reaction. For example, the study of the effect of flowrate on conversion demonstrated a key trade-off between reaction time (which relates to required reactor size) and excess methanol fed to reactor (which relates to amount of methanol required).
5. Incorporate biodiesel production projects into coursework at various levels. This project motivated topics that were used regularly for senior and freshman design projects. Results from this project were used as examples in senior design lectures and in specialized renewable energy courses.
6. Increase awareness of issues surrounding biofuel production. Over 30 undergraduate students have been involved in doing research or design work for this project and numerous presentations about biodiesel and biofuels have been given on campus and at professional meetings based on this research. This project is well-known on the Drexel campus and students who are interested in renewable energy and biofuels often consult with Dr. Cairncross for advice. Yearly demonstration experiments have been conducted with visiting middle school and high school students.
Summary/Accomplishments (Outputs/Outcomes):
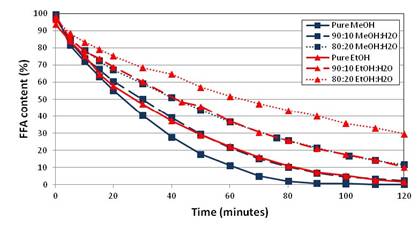
Figure 2 – Conversion of FFA to biodiesel in a Bubble Column Reactor performed with 0.1% sulfuric acid catalyst and 0.75 mL/min feed rate of liquid alcohol-water mixture. Oleic acid content of the reactor is tracked over time by titration until 120 minutes.
The bubble column reactor is highly robust for converting free fatty acids (FFA) to biodiesel. Figure 2 displays the conversion of oleic acid (a model FFA) to biodiesel in a bubble column reactor operating at 120°C where the feed alcohol contains varying composition of water in methanol and ethanol. In most biodiesel reactors, water is detrimental to conversion because the esterification reaction (in Figure 1) is reversible and equilibrium-limited. The rising methanol bubbles in the bubble column reactor strip water produced by the reactions and remove it from the reactor, enabling the high conversions shown in Figure 2. In this project, the performance of the bubble column was demonstrated under a variety of conditions: catalyst type and concentration, feed alcohol flowrate, FFA content, temperature, and reactor configuration. Testing of biodiesel product from the reactor met all ASTM specifications for biodiesel. Mixed oil feedstocks that contain both FFA and triglycerides also were used in the bubble column reactor – the bubble column reactor effectively converts all of the FFA in mixed feedstocks into biodiesel but achieves low conversion of triglycerides to biodiesel. Therefore, a second reaction step is necessary to convert triglycerides to biodiesel using a base catalyst; we have demonstrated the effectiveness of this two-step procedure using a bubble column reactor and mixed feedstocks containing 10-100% FFA.
Process feasibility studies using a mathematical model of the bubble column reactor developed in this project have evaluated the economic and environmental feasibility of the bubble column reactor in comparison to supercritical reactors and high-pressure reactors that currently are used to convert trap grease to biodiesel. Although all three processes are economically feasible and significantly reduce greenhouse gas emissions in comparison to petroleum diesel, the bubble column process was predicted to have somewhat better performance by both economic and environmental metrics.
Conclusions:
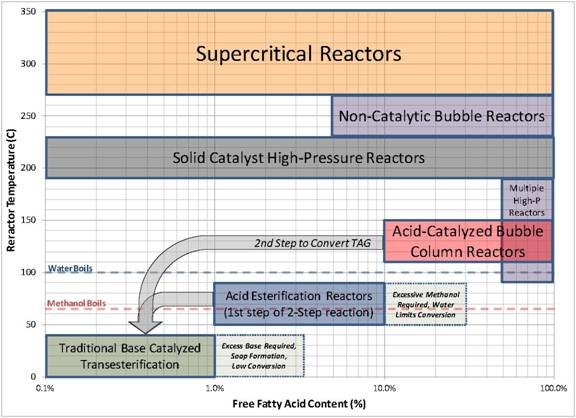
Figure 3: A map of process conditions for different biodiesel reactor technologies. The horizontal axis represents the initial FFA content of the oil in the reactor and the vertical axis represents the typical range of temperatures used in the reactor. All ranges in the figure are approximate.
The bubble column biodiesel reactor is robust for converting low-value and impure feedstocks into biodiesel. Figure 3 displays approximate ranges of applicability of different reactor technologies for biodiesel production and how the bubble column reactor fills a critical gap in enabling processing of a wide variety of oil feedstocks into biodiesel. The traditional biodiesel process uses base catalysts for transesterification and is effective for feedstocks containing less than 1% FFA. For feedstocks in the range of 1-10% FFA, two-step acid esterification/base transesterification is effective. However, for feedstocks containing between 10% to 100% FFA, the options are limited. Supercritical reactors, high pressure reactors, and solid catalysts are effective but are technologically demanding and require higher temperatures and pressures. The bubble column reactor fills the gap and enables complete conversion of FFA and triglycerides to biodiesel in a two-step process using the bubble column as the first step.
Also the bubble column has been demonstrated to be robust for feeds containing water. For both ethanol and methanol as the alcohol feed, high conversions are achieved when the alcohol contains up to 20% water. A reaction model has been developed that predicts the reaction kinetics, equilibrium limitations and mass transfer resistances within the bubble column reactor. Using model parameters based on results with pure methanol, the model accurately predicts changes in conversion when the feed contains up to 20% water. The bubble reactor has been demonstrated to convert FFA in mixed feedstocks and trap grease into biodiesel with high conversion in less than two hours.
This EPA P3 project has been a valuable educational program enrolling more than 30 undergraduate students directly in research or design projects, involving approximately 150 middle-school and high-school students in biodiesel demonstration experiments, and exposing more than 300 students in courses to biofuels processes examples of applying engineering principles.
Journal Articles:
No journal articles submitted with this report: View all 10 publications for this projectSupplemental Keywords:
biodiesel, biofuels, waste to energy, renewable energy, free fatty acids, esterification, RFA, Scientific Discipline, Sustainable Industry/Business, POLLUTION PREVENTION, Sustainable Environment, Energy, Environmental Chemistry, Technology for Sustainable Environment, Environmental Engineering, sustainable development, environmental sustainability, alternative materials, biomass, alternative fuel, biodiesel fuel, energy efficiency, energy technology, alternative energy sourceRelevant Websites:
Project Title: Extraction of Lipids from Wastewater to Produce Biofuels Exit
Progress and Final Reports:
Original AbstractP3 Phase I:
Novel Reactor Design for Biodiesel Production | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.