Grantee Research Project Results
Final Report: Eco-Friendly Solvent Free Photodecarbonylation for the Synthesis of Biologically Active Natural Products
EPA Grant Number: SU833911Title: Eco-Friendly Solvent Free Photodecarbonylation for the Synthesis of Biologically Active Natural Products
Investigators: Garcia-Garibay, Miguel A. , Family, Farnosh , Kuzmanich, Greg , Shiraki, Saori
Institution: University of California - Los Angeles
EPA Project Officer: Page, Angela
Phase: I
Project Period: August 15, 2008 through August 14, 2009
Project Amount: $10,000
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet (2008) RFA Text | Recipients Lists
Research Category: Nanotechnology , Pollution Prevention/Sustainable Development , P3 Awards , P3 Challenge Area - Chemical Safety , Sustainable and Healthy Communities
Objective:
These students contributed to this project: Farnosh Family, Department of Chemistry and Biochemistry, UCLA; Saori Shiraki, Department of Chemistry and Biochemistry, UCLA; Greg Kuzmanich, Department of Chemistry and Biochemistry, UCLA.
The objective of this project is to integrate sustainability into one of the largest and most profitable sectors of the economy – the pharmaceutical industry. To accomplish this objective, the primary goal was to develop eco-friendly methods to synthesize complex molecules that may become cancer medicines. The molecules we have focused on all contain a structural motif that has proved difficult to synthesize. This structural framework contains two adjacent carbon atoms each bonded to three other carbons. This crowding around the carbon-carbon bond creates steric hindrance that makes it difficult for any reaction to occur at that site. Yet, this crowded carbon-carbon bond appears in a number of natural products that may be useful medicines. Therefore, we sought out to modify current means of producing such molecules using a photoinduced decarbonylation reaction of suitable, easier to prepare, crystalline ketones.
Photodecarbonylation reactions that occur in crystalline ketones leads to the formation of a new bond between two tri-susbtituted carbon atoms after the expulsion of the carbon atom and oxygen atom between them. It is the breaking of two bonds and the formation of a new bond that leaves behind an interesting and hard to synthesize complex structure.
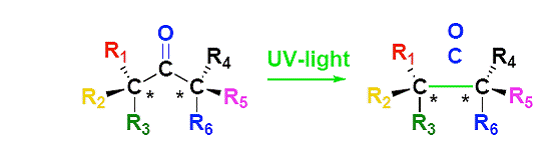
As shown in the scheme above, the two carbons labeled with asterisks can be chiral if groups R1, R2, and R3 are different, and groups R4, R5, and R6 are also different from each other. Chiral carbons pose a challenge in their synthesis, yet chirality is fundamental to many biologically relevant molecules. Reactions that produce chiral carbons normally have statistical chance of producing either enantiomer. Enantiomers are molecules with a chiral center that can either be arranged to the left or to the right. Enantiomeric ratios can be improved by use of chiral auxiliaries or chiral catalysts, which is the most commonly used practice in academic and industrial laboratories.
The environmental problem with the use of such auxiliaries and catalysts is the poor atom economy of the reaction. By design, the catalysts and auxiliaries are not a part of the product and therefore are a part of the waste. Furthermore, unless the reaction produces 100 percent of only one enantiomer, there must be separation and purification which leads to use of more solvent and waste.
Our project offers a new approach to the synthesis of chiral crowded and hard to synthesize molecules. In our approach, the chiral centers are created by a coupling of one a molecule with a chiral carbon with a carbonyl source – a carbon double bonded to an oxygen.

By creating the chiral centers before they are next to each and therefore less crowded, we have improved enantiomeric control. The ketone – the carbon double bonded to an oxygen – is now situated between the two chiral centers. This makes the molecule set up nicely for our photodecarbonylation reaction. The photodecarbonylation will then produce a molecules with adjacent chiral centers.
In addition to changing the traditional approach to such complex molecules, our project aimed at increasing the presence of green chemistry in the graduate school education of organic chemists. In addition to discussing it and our project in our group meetings with other fellow graduate and undergraduate students, we also invited pharmaceutical chemists to describe the impact of green chemistry on the industry.
Summary/Accomplishments (Outputs/Outcomes):
Our research focused on the use of the photodecarbonylation reaction in the production of adjacent chiral centers. We examined specifically the selectivity, and therefore efficiency, of the reaction. For proof of principle, we examined a class of compounds referred to as phenyl pyrrolidinones, shown below. The R group in the diagram has been varied among several different functional groups.
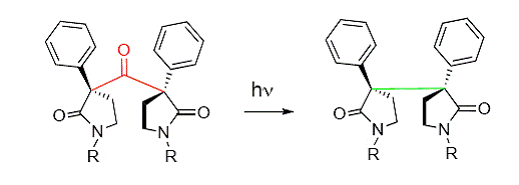
Our preliminary results are very encouraging. We have established that these compounds do photodecarbonylate. Additionally, we have found that the selectivity of the reaction is quite high. Depending on which R group is present and the reaction conditions, enantiomeric excesses of 80 percent have been achieved.
Conclusions:
The photodecarbonylation reaction is a highly selective reaction that is very promising for the formation of structurally complex molecules with crowded carbon-carbon bonds that may be useful cancer medicines.
Proposed Phase II Objectives and Strategies:
Now that the photodecarbonylation of the phenyl pyrrolidinones has been established, our next objective is to synthesize the nitro analogue of the phenyl pyrrolidinone and continue the synthesis to a natural product.
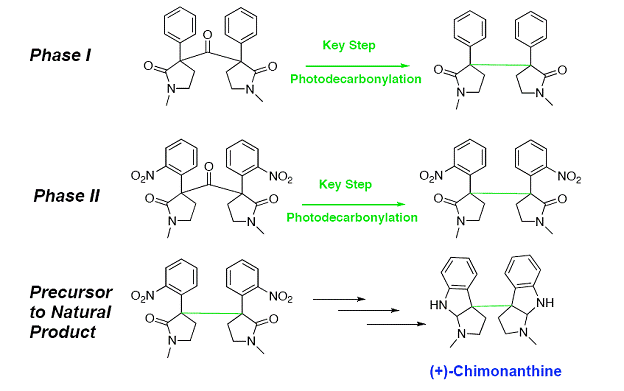
Phase II will focus on the synthesis of (+)-chimonanthine specifically. We will approach the synthesis with the knowledge we learned from Phase I. We have established a method for the formation of the ketone and the photodecarbonylation. From there, we will work out the proposed three synthetic steps to produce (+)-chimonanthine. This will allow us to explore other natural products with similar structural frameworks.
Journal Articles:
No journal articles submitted with this report: View all 1 publications for this projectSupplemental Keywords:
Green chemistry, photodecarbonylation, natural productsRelevant Websites:
www.chemistry/ucla.edu/mgghome.html
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
