Grantee Research Project Results
Final Report: Data Collection Platforms for Integrated Longitudinal Surveys of Human Exposure-Related Behavior
EPA Grant Number: R831541Title: Data Collection Platforms for Integrated Longitudinal Surveys of Human Exposure-Related Behavior
Investigators: Whitmore, Roy , Kizakevich, Paul
Institution: Desert Research Institute
EPA Project Officer: Aja, Hayley
Project Period: May 1, 2004 through April 30, 2008
Project Amount: $4,998,513
RFA: Aggregate Exposure Assessment: Longitudinal Surveys of Human Exposure - Related Behavior (2003) RFA Text | Recipients Lists
Research Category: Human Health
Objective:
The objectives are to develop and validate innovative methods for collecting exposure factor data in longitudinal surveys, including: (1) time, activity, location, exertion level, and environmental (TALE) data; (2) dietary consumption data; and (3) data on use of consumer products, including pesticide products, household cleaning products, and personal care products. An additional objective is to investigate innovations including voice diaries, passive microenvironment identification, wireless interfaces, intelligent prompting, and automated daily review to collect the data both accurately and with low participant burden.
Summary/Accomplishments (Outputs/Outcomes):
Development Approach: The project was conducted in two main phases. In Phase 1, we developed a set of alternative data collection platforms: paper questionnaires, Pocket PC-based menu, voice, and photo diaries, and passive sensors instruments to acquire, integrate, and store data. We pilot tested each platform with 40 participants for TALE, dietary, and consumer product data collection. In Phase 2, we developed a second-generation data collection platform, making improvements based on the initial pilot test results. We tested the second generation platform in 2 pilot field studies with 16 participants for TALE and 20 participants for dietary data collection.
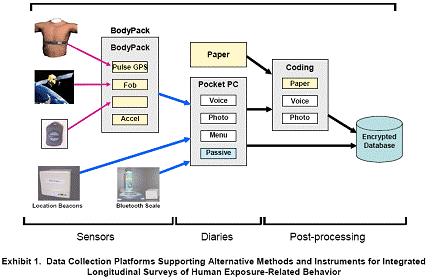
Data Collection System: The developed system comprises a handheld Pocket PC computer and various sensor instruments for passive and semi-automated monitoring (Exhibit 1). These include a wearable Body Pack™ sensor data collection instrument, location sensing “Beacons,” GPS devices, heart rate sensors, and a 3-axis accelerometer sensor. TALE, dietary, and product use data are collected on the Pocket PC via menu, voice, or photo-based diaries and stored in a secure, encrypted database. Sensor data are acquired using various sensors and instruments, and automatically transmitted to the Pocket PC via Bluetooth wireless for integration with diary data in the common secure database. Paper diaries also were developed so that we could compare various automated and semi-automated data collection technologies to traditional data collection methods.
Evaluation Approach: Each data collection platform was evaluated with regard to: (1) participant burden; and (2) ability to obtain complete and accurate information on human exposure factors. Participant debriefing questionnaires were the primary method used to evaluate participant burden. Evaluating the numbers of reported (1) activities and locations, (2) foods and drinks, and (3) product use events was the primary method used to determine data completeness. We assumed that the data were more complete and accurate for the data collection methodologies that resulted in substantially more reported events.
Phase 1 Pilot Test
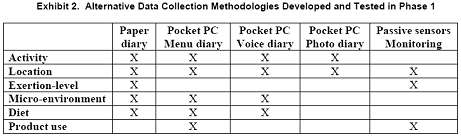
Exhibit 2 summarizes the types of alternative data collection methodologies that were developed and tested in Phase 1. The passive sensor monitoring required the participant to carry a Pocket PC for data storage as well as data collection via menus, voice, or photo-based diary data collection. Microenvironments relevant to exposure assessment for combustion products also were monitored.
Two types of passive monitoring of location were tested:
- A GPS chip was mounted in the Body Pack to monitor location continuously whenever the participant was in a location where the chip could receive GPS satellite signals, primarily outdoors.
- Beacons were placed in rooms of the home frequented by participants. Whenever the participant entered a room with a beacon present, the beacon would sense the presence of the Pocket PC and send a time-stamped signal to the Pocket PC. Hence, it was possible to passively monitor the participant’s room-to-room movements within his/her home.
Exertion level was monitored passively using a heart rate monitor. We developed a heart rate monitor that each participant was asked to wear as a direct indication of exertion level.
Use of consumer products was monitored passively using fobs (like those used to open and close the doors of an automobile). During the initial visit to set up the home for monitoring, the project staff placed a fob on or near each product to be monitored and collected inventory data regarding those products. Whenever the participant used a monitored product, he/she pressed the fob associated with the product, which sent a time-stamped signal through the Body Pack to the Pocket PC. Hence, with little effort on the participant’s part, we were able to determine when each product was used. The Pocket PC then prompted the participant to respond to a few questions regarding each use of a cleaning or pesticide product.
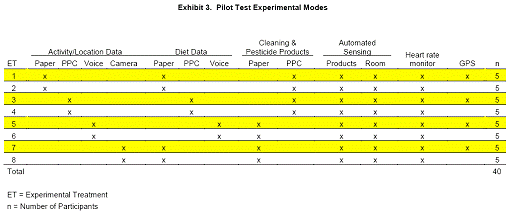
For the Pocket PC and photo diaries, participation in the Phase 1 pilot study began at noon each day because of longevity issues with the Pocket PC batteries. Each participant also used a Body Pack to collect heart rate, residential beacon, and fob-invoked product use data. Paper diary users carried a Pocket PC for storing Body Pack sensor data. Half of the participants carried an RTI- developed GPS subsystem which was integrated into the Body Pack as shown in Exhibit 3.
An important component of RTI’s comparison of the various methods for collecting human exposure factor data was a formal debriefing questionnaire administered to each pilot test participant. Some of the more salient comments from the participants regarding the four modes of data collection, as well as the Polar heart rate band and the Body Pack, are shown in Exhibit 4.
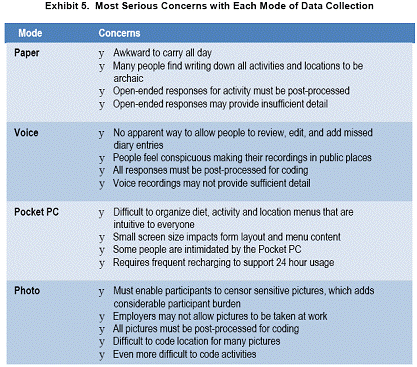
None of the data collection modes was optimal. Our most serious concerns from Phase 1 are summarized in Exhibit 5.
The key lesson learned from Phase 1 was that asking participants to simultaneously keep track of all three types of information – TALE, diet, and product use – was overwhelming. Although each mode had relatively low burden, being responsible for collecting all three types of data negatively affected data completeness and data quality.
Phase 2 Pilot Test
The Phase 1 results clearly indicated that the photo diary and voice diary methodologies were impractical, both for participants and for investigators. Data collection was flawed due to privacy concerns, both methods can be intrusive, and at best only a subset of the desired data can be acquired. Nonetheless, some participants liked the inherent convenience of these methods, leading us to consider how the positive aspects of these technologies could be used in an improved system.
The challenges for the Pocket PC mode are primarily technological challenges. With the growing use of smart touch-enabled cell phones (i.e., Apple iPhone), we felt that the general population will grow more familiar and facile with such devices and user-concerns with handheld devices will likely diminish. After considering all of these results, including participant feedback, we selected the Pocket PC as the preferred mode of data collection and developed a second generation data collection platform focusing on (1) improving the usability of the Pocket PC diaries and (2) enhancing the passive sensor data collection instruments.
The primary improvements made in the second generation data collection platform include:
- Easy configuration of a field study data collection scheme based on a “protocol” file defining which diaries, sensors, forms, and questionnaires are to be used in a particular study,
- Improved authoring system for data collection menus utilizing database tables,
- Burden-reducing improvements to the TALE, dietary, and product use diaries,
- Enabling three user-selectable means of reporting “other, specify” responses: virtual touch/tap keyboard, voice dictation, and stylus handwriting script,
- Enabling daily review and editing of diary data using a time-sequenced summary of data entries,
- Redevelopment of the Body Pack using a modular design and a rechargeable battery,
- Addition of an accelerometer for energy expenditure data and user compliance monitoring,
- Improvements to the heart rate monitor and the wireless scale instruments, and
- Automatic sensing of Body Pack presence and configuration of Body Pack settings.
In June and July 2008, we conducted a 20-person pilot test of the dietary dairy. In March and April
2009, we conducted a 16-person pilot test of the integrated TALE dairy and passive data collection instrumentation.
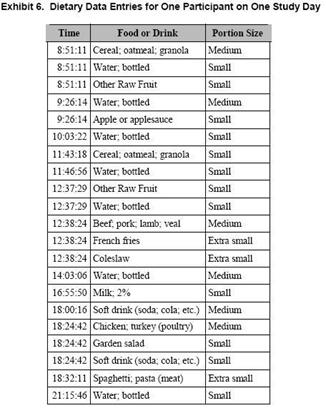
Exhibit 6 illustrates of a typical person’s dietary data for 1 day. Note that the foods and drinks that were consumed are typical of a normal daily diet, starting with breakfast foods.
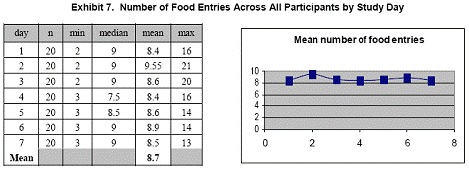
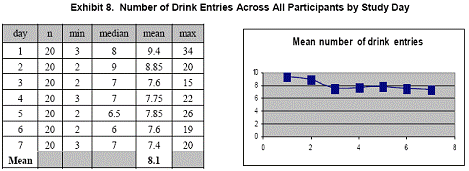
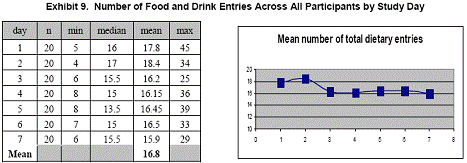
Summary statistics for the number of diet entries across all participants for each day are presented in Exhibits 7, 8, and 9. Although the mean number of total entries decreased slightly during the 7- day study, the number of daily entries remained remarkably consistent.
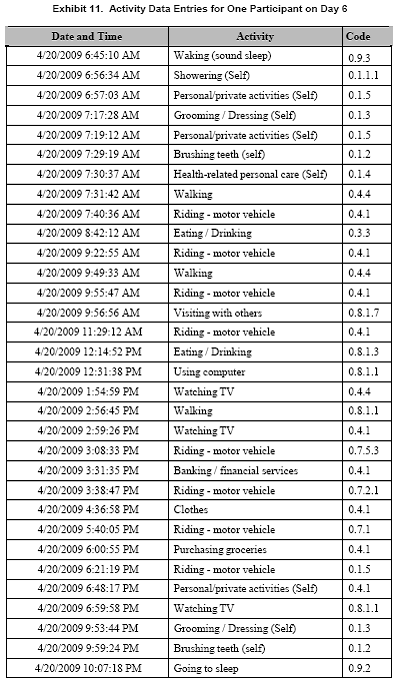
TALE diary location and activity entries are presented for one participant (#311000) on Day 6 of the collection period (Exhibits 10 and 11). This participant recorded 18 changes in location, after first reporting being at home at 6:45 am. The participant also recorded 32 activities throughout the day. These data are a good example of the close association between activities and location.
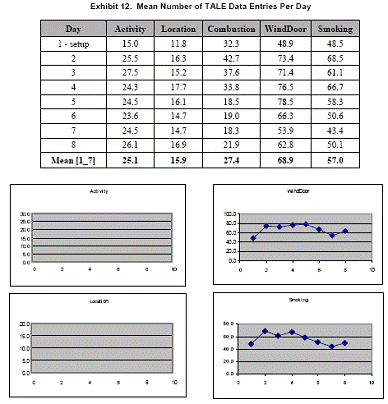
The mean number of data entries per day is presented for each of the TALE diary instruments in
Exhibit 12. Data are provided in table and chart formats below.
Technical Effectiveness/Economic Feasibility
The purpose of this project was to develop technologies that could be used to collect accurate, detailed data on human exposure factors for seven consecutive days, at least quarterly, in a longitudinal survey of the general U.S. household population. Based on data from the second pilot test, this objective was achieved for most of the data collection technologies. Certainly, it appears to have been achieved for the diet and TALE diaries, which provide detailed data regarding foods, drinks, activities, locations, and microenvironments. Users reported that these diaries could be used with relative ease, with little burden, and with little intrusion into their daily lives.
At the outset of the project, we realized that a clear, easy-to-use interface would be necessary for user acceptance and data entry compliance. After the initial pilot, we focused on the user interface, simplified look, a clear font, web-like navigation, and user options such as font-size control. The improved diary productivity (e.g., activity report rate) indicates that these efforts were effective.
We were not able to conduct a second test of the product-use monitoring methodology, but we feel confident that it also would produce good results with the improved, second-generation methodology. One potential technical issue with widely implementing this methodology is that the fob technology used in this study to signal product use has evolved, and the available fob technology may have different wireless communication characteristics. While this may require some retooling of the Body Pack to use the newer technologies, the modular Body Pack design will help minimize such costs.
The technologies developed for passive monitoring were fairly successful, especially considering the widely varied aspects of field data collection and changing operational conditions as users went about their day. Our project had challenging data collection goals: (1) 24-hour data acquisition for 7 days, (2) low-power, battery-based electronics, (3) wearable sensors and data recording, and (4) variant environments, including homes, vehicles, and various work, commercial, and other locations, and (5) user freedom of movement and locomotion. While we addressed these issues as much as possible in the development of the various systems, use of such technologies under these constraints somewhat limits their effectiveness. Nonetheless, the various sensors and technologies recorded excellent data whenever the conditions were amenable to the technology.
A significant cost advantage of handheld computer diaries, as compared to paper diaries, is that the data are immediately coded and stored in a secure database. Paper diaries, as well as photo and audio dairies, require a data analyst to code the data adding significantly to the cost. Furthermore, since the user can enter and review his or her data entries, the quality of collected data should be better; leading to improved modeling and lower overall cost. Handheld computers that have better performance than those used for this study now cost half as much. Smartphones offer the additional capability of daily uploading of data to a secure central database, and with integrated GPS receivers and accelerometers have a lower overall cost for diary and sensor data collection. These cost and technology improvements support the economic feasibility of this methodology.
Conclusions:
Technical Effectiveness/Economic Feasibility
The purpose of this project was to develop technologies that could be used to collect accurate, detailed data on human exposure factors for seven consecutive days, at least quarterly, in a longitudinal survey of the general U.S. household population. Based on data from the second pilot test, this objective was achieved for most of the data collection technologies. Certainly, it appears to have been achieved for the diet and TALE diaries, which provide detailed data regarding foods, drinks, activities, locations, and microenvironments. Users reported that these diaries could be used with relative ease, with little burden, and with little intrusion into their daily lives.
At the outset of the project, we realized that a clear, easy-to-use interface would be necessary for user acceptance and data entry compliance. After the initial pilot, we focused on the user interface, simplified look, a clear font, web-like navigation, and user options such as font-size control. The improved diary productivity (e.g., activity report rate) indicates that these efforts were effective.
We were not able to conduct a second test of the product-use monitoring methodology, but we feel confident that it also would produce good results with the improved, second-generation methodology. One potential technical issue with widely implementing this methodology is that the fob technology used in this study to signal product use has evolved, and the available fob technology may have different wireless communication characteristics. While this may require some retooling of the Body Pack to use the newer technologies, the modular Body Pack design will help minimize such costs.
The technologies developed for passive monitoring were fairly successful, especially considering the widely varied aspects of field data collection and changing operational conditions as users went about their day. Our project had challenging data collection goals: (1) 24-hour data acquisition for 7 days, (2) low-power, battery-based electronics, (3) wearable sensors and data recording, and (4) variant environments, including homes, vehicles, and various work, commercial, and other locations, and (5) user freedom of movement and locomotion. While we addressed these issues as much as possible in the development of the various systems, use of such technologies under these constraints somewhat limits their effectiveness. Nonetheless, the various sensors and technologies recorded excellent data whenever the conditions were amenable to the technology.
A significant cost advantage of handheld computer diaries, as compared to paper diaries, is that the data are immediately coded and stored in a secure database. Paper diaries, as well as photo and audio dairies, require a data analyst to code the data adding significantly to the cost. Furthermore, since the user can enter and review his or her data entries, the quality of collected data should be better; leading to improved modeling and lower overall cost. Handheld computers that have better performance than those used for this study now cost half as much. Smartphones offer the additional capability of daily uploading of data to a secure central database, and with integrated GPS receivers and accelerometers have a lower overall cost for diary and sensor data collection. These cost and technology improvements support the economic feasibility of this methodology.
Journal Articles:
No journal articles submitted with this report: View all 12 publications for this projectSupplemental Keywords:
observations in daily living, ecological momentary assessment, activity monitoring, mobile computing, windows mobile;, Health, Scientific Discipline, ENVIRONMENTAL MANAGEMENT, HUMAN HEALTH, Health Risk Assessment, Risk Assessments, Biology, Exposure, Biochemistry, Risk Assessment, dietary exposure, toxicity, micro environmental influences, data collection platforms, human exposure, long term exposure, food consumption habits, household study, human activities, food consumption behavior, human health riskRelevant Websites:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.