Grantee Research Project Results
Final Report: Biogeochemistry of Arsenic in Contaminated Soils of Superfund Sites
EPA Grant Number: R830842Title: Biogeochemistry of Arsenic in Contaminated Soils of Superfund Sites
Investigators: Sarkar, Dibyendu , Datta, Rupali
Institution: The University of Texas at San Antonio
EPA Project Officer: Hahn, Intaek
Project Period: August 1, 2003 through July 31, 2005 (Extended to July 31, 2007)
Project Amount: $391,473
RFA: Superfund Minority Institutions Program: Hazardous Substance Research (2002) RFA Text | Recipients Lists
Research Category: Hazardous Waste/Remediation , Safer Chemicals , Land and Waste Management
Objective:
I. EXECUTIVE SUMMARY
In Phase I of the greenhouse study, column experiments were accomplished in June 2005. Two types of soils were selected based on the physico-chemical properties, which were most likely to influence As retention. Pesticides (sodium arsenate and DMA) were used at two different rates (675 and 1500 mg kg-1) representing superfund conditions. Rice (used as the test crop) was grown for a period of 6 months. Rice was harvested twice, after 3 and 6 months. Soils were collected at different time periods: immediately after spiking (time-0), after 6 months (time-final) and after 1 and 3 years.
The second set of column experiments was started in late June 2004. It involved two soils (Immokalee and Orelia), and two pesticides (Sodium arsenate and DMA) added at a rate of 1500 mg kg-1 of soil. Both soils were amended with Al-WTR and Fe-WTR at two rates (5% and 10%) expected to be effective in reducing soluble As. Rice was used as the test crop.
To better understand the effect of soil-aging on As bioaccessibility and speciation, columns from both of the experimental set ups were retained (without the cultivar) for three years and were sampled once in a year. The columns were maintained at optimum soil moisture conditions (“pot-holding capacity”). Aqueous speciation study of As was also conducted to see whether there were changes in the oxidation state of As.
Soil samples collected at different time periods were extracted for soil-As forms by a sequential extraction technique and for bioavailable As by in-vitro gastrointestinal method as described by Datta and Sarkar (2004). In all the three soils except for Pahokee Muck, most of the total extractable As was in the soluble form. Soluble form decreased significantly after 1 year of soil-pesticide equilibration. Arsenic bioaccessibility also decreased with time in all the four soils, indicating the affect of ageing as well as soil properties on bioaccessibility. Selected in-vivo study using As-contaminated soils were conducted on male/female BALB/c mice. In-vivo bioavailability was calculated from the blood collected by cardiac puncture at different time periods.
Results obtained with WTRs suggests that significantly higher levels of the added As was transformed into bound forms, thus decreasing its bioaccessibility in Fe-WTR and Al-WTR amended soils. Arsenic bioaccessibility decreased immediately after spiking for both the WTRs, with more reduction at higher application rate (10%). Geochemical speciation after 1 year of soil-WTR-pesticide equilibration showed that As in the WTR amended soils was mostly in Fe-and Al-bound form, therefore less available, thereby decreasing As bioaccessibility.
Summary/Accomplishments (Outputs/Outcomes):
II. INTRODUCTION
The Agency for Toxic Substances and Disease Registry (ATSDR) has ranked arsenic (As) as #1 in its 2001 Priority List of Hazardous substances at Superfund sites. There are many Superfund sites where soils are severely contaminated with As as a result of over-application of arsenical pesticides. Since As is classified as a Group-A carcinogen, an elevated health risk is associated with long-term human exposure to As in pesticide-applied soils. Arsenic exists in soils mostly in the +5 or +3 oxidation state. Arsenate (AsV) is the oxidized form and occurs in well-aerated soils, whereas in chemically-reduced soil environments, arsenite (AsIII) is the prevalent As form. Although arsenite is more toxic than arsenate, arsenate can also have deleterious effects on humans, plants, and microorganisms. Sodium arsenate and dimethyarsinic acid (DMA) are two pesticides extensively used.
There is an increasing need for data dealing with As bioavailability in As-contaminated soils, varying in physicochemical properties. Since the current methods employed to measure the amount of bioavailable As in soils are expensive, time-consuming and requires specialized facilities and personnel, it is a practice to use the conservative estimate of 100% As bioavailability in all soil types, ignoring differences in physicochemical properties between soil types. More realistic measures of As bioavailability in soils would decrease cost associated with clean-up of As-contaminated sites.
We choose to utilize the recently developed term “bioaccessibility” to better define the ambiguous use of bioavailability that has labeled common biological and chemical extraction assays. According to Semple et al. (2004), the bioaccessible compound is available to cross an organism’s cellular membrane from the environment, if the organism has access to the chemical. However, the chemical may be either physically removed from the organism or only bioavailable after a period of time (Semple et al., 2004). Contaminant bioavailability or bioaccessibility is increasingly being used as a key indicator of contaminant risk to environmental and human health (Adriano et al., 2004). Recent studies show that As bioaccessibility in soils can not be arbitrarily assigned a 100 % value (Rodriguez et al., 1999; Yang et al., 2002; Sarkar et al., 2004). Several studies have indicated that As bioaccessibility in soil is mostly a function of a soil’s chemical properties, using sequential As fractionations and in-vitro tests (Davis et al., 1992; Ruby et al., 1993; Sheppard et al., 1995). Thus, the calculated risk may be overestimated, sky-rocketing the clean-up cost for such contaminated soils. Lead is the only metal that has an explicit soil bioavailability adjustment (Dieter et al., 1993).
Increased cost and the objections set by several groups on experiments with animals have guided the scientific community to use in-vitro tests as an analogue to evaluate a contaminant’s bioavailability. One of the commonly used in-vitro tests is the physiologically based extraction test that has been developed to simulate metal bioavailability in the human gut (Ruby et al., 1993). The in-vitro test simulates As bioaccessibility in human gastrointestinal juices of soil particles containing As. Another in-vitro test was developed by Rodriguez et al. (2003) that attempts to predict As bioaccessibility in the stomach, as well as in the intestine phase, using an Fe hydroxide gel to simulate metalloid absorption by human membranes. A modified procedure developed by Sarkar and Datta (2004) includes Fe oxide strip bags for use with the intestine-absorbed in-vitro test.
To better understand the variability of soil As bioaccessibility, empirical models have been constructed, using soil chemical properties (Yang et al., 2002; Sarkar et al., 2004; Datta and Sarkar, 2004). Dithionite-citrate-bicarbonate-extractable Fe content and pH were the most significant (α = 0.05) chemicals properties that explained the reduction in As(V) bioaccessibility with time (Yang et al., 2002). Other studies showed that the oxalate-extractable Fe/Al content manifested the variability of As bioaccessibility in three acidic soils (Datta and Sarkar, 2004). We hypothesized that the Fe/Al hydroxide soil pools would be major sorbents (in addition to Ca/Mg) for soluble As in either acidic or alkaline soils.
Conventional remediation techniques involve excavation and some form of ex-situ treatment (soil washing, solidification, etc.), followed by disposal and long-term monitoring, which are often expensive and disruptive to the surrounding landscape (Seaman et al., 2003). Therefore, it is imperative to develop techniques that can treat and stabilize contaminants in situ in an efficient and cost effective manner. Examples of in-situ treatment techniques include soil flushing, electro kinetics, bioremediation, vacuum or air stripping, and immobilization. In-situ immobilization is a cost-effective approach where land-applied amendments are used to remove contaminants via adsorption and or precipitation reactions that render the contaminant immobile (Adriano, 1987). Numerous inorganic amendments (clays, Al/Fe/Mn oxides and hydroxides) may be land-applied to As- contaminated soils as a means of reducing As mobility (Hartley, 2004).
Use of Fe/Al hydroxide-containing materials to remediate As-contaminated sites is based on the general notion that As adsorption in soils is primarily controlled by Fe/Al (hydr)oxides (Livesey and Huang, 1981; Goldberg, 1986; Goldberg, 2002). A low-cost and potentially effective substitute for natural Fe/Al hydroxides could be the drinking-water treatment residuals (WTRs). The WTRs are the waste material generated during the drinking-water treatment process, and are primarily composed of Fe/Al (hydr)oxides, but they may also contain some activated C, and high molecular weight, long-chain, water soluble organic compounds (Elliot and Dempsey, 1991).
Our earlier work has demonstrated the efficacy of WTRs in removing As from water (Makris et al., 2006). However, there is no published work on As sorption by WTR-amended soils. Arsenic-contaminated soils pose serious risk to human health and such soils that are characterized by low As sorption capacities due to low Fe/Al hydroxide content may be most vulnerable to As mobility and transport. We hypothesized that land-applied WTRs would significantly increase the As retention capacity of such soils, decreasing As mobility, availability and bioaccessibility.
III. MATERIALS AND METHODS
i) Soil sampling, Preparation and Characterization
Four surface soils (0-12 cm) were used for the greenhouse study. Immokalee series soil was collected from the Southwest Florida Research and Education Center, Immokalee, Florida, Millhopper series soil was collected from the University of Florida campus at Gainesville, Florida. The Pahokee Muck series was collected from the Everglades Research and Education Center at Belleglade, Florida and Orelia soil was collected from Corpus Christi, Texas. Soil samples were air dried, passed through a 2-mm sieve and characterized for various soil properties. Soil pH, electrical conductivity, particle size, and water content, cation-exchange capacity were measured using standard protocols (Sparks, 1996). Organic matter was measured using the loss-on-ignition method (Sparks, 1996). Plant-available P was extracted by Mehlich III solution (Mehlich, 1984). Oxalate-extractable Fe and Al were obtained using Tamm’s reagent (Sparks, 1996). Total recoverable Ca, Mg, Fe, Al, P, and As was obtained by soil digestion according to USEPA method 3050B (USEPA, 1996). Phosphorus was measured colorimetrically by an UV/Visible light spectrophotometer using the molybdate-ascorbic acid method (Watanabe and Olsen, 1965). Calcium, Mg and Al were analyzed using the flame atomic absorption spectrometry (FAAS) and As was analyzed by graphite furnace atomic absorption spectrometry (GFAAS).
ii) Soil amendments and plant growth
All the four soils were spiked with sodium arsenate at two rates - 675 and 1500 mg As kg-1 soil. These two rates were selected to represent the higher and lower end of the superfund soil-As concentrations. Columns (13” tall x 6” i.d) made of PVC were used in this greenhouse study. The bottom 7” of the columns was filled with white sand that had no As retention capacity. Pesticide amended soil was packed in the top 6” of the PVC column. Each column was provided with a reservoir compartment to hold the excess leachate and a hole fitted with a nozzle connected to nalgene tubing collected the leachate. The columns were arranged in a randomized block design and were rotated periodically to account for variances in temperature and sunlight within the greenhouse. Rice was used as the test crop. The number of PVC columns used were 24 treatments (4 soils x 2 rates x 3 replicates) and 12 controls (4 soils x 3 replicates). Leaching was induced after 2 weeks of pesticide application. Seeds were sowed directly on the soils. Prior to germination, the seeds were surface-sterilized in 3% H2O2 and then rinsed with distilled water. Germinated seeds were counted 4 days after initiation. Rice was harvested in those columns where there was germination of rice seedlings. Shoot and root height were measured during harvesting. Shoot height was measured from the culm base to the tip of the longest leaf and root length was measured from the root-shoot junction to the tip of the longest root. Leachate water was analyzed for soluble As using the GFAAS. Soils were collected from the surface layer immediately after spiking (time-0), after six months, one year and 3 year of soil-pesticide equilibration and assessed for soil-As forms.
iii) Sequential Extraction Method
Sequential extraction scheme developed by Chungao and Zihui (1988) was employed to identify the various operationally defined forms of as follows.
1. Water-soluble phase: One gram of soil was extracted at room temperature for 30 min with 50 mL of deionized water under continuous shaking. The samples were centrifuged, filtered, and analyzed for soluble As.
2. Exchangeable phase: The residual soil from the water-soluble fraction step was shaken at room temperature for 30 min with 50 mL of 1M NH4Cl. The mixture was centrifuged, and the supernatant was analyzed for exchangeable As.
3. Fe- and Al-bound phase: The soil residue from step 2 was shaken at room temperature for 17 hr with 50 mL of 0.1 M NaOH. The residue after centrifugation was washed twice using 25 mL of saturated NaCl solution. The supernatant from these washes were pooled and analyzed to determine the As fraction bound to Fe/Al oxides
4. Ca- and Mg-bound phase: The residual soil was extracted using 50 mL of 0.25 M H2SO4 by shaking for 1 hr at room temperature. The residue after centrifugation was washed twice using 25 mL of saturated NaCl solution. The supernatant from these washes were pooled and analyzed to determine the As fraction bound to Ca/Mg compounds.
5. Organic matter and sulfide-bound phase: The residual soil was digested using 3 mL of 30% H2O2 (adjusted to pH 2.0 using HNO3) at 85oC for 3 hr. To prevent adsorption of extracted As to the oxidized soil, the samples were cooled and 5 mL of 3.2 M NH4OAc in diluted HNO3 was added. The samples were diluted to 20 mL with deionized water, followed by shaking for 30 min at room temperature, and then analyzed to determine the soil organic matter and sulfide-bound As.
6. Residual phase: The remaining soil was extracted using 25 mL of concentrated HNO3 at 105oC, until approximately 5 mL of solution remained. The samples were diluted to 25 mL with deionized water, centrifuged, and the supernatants were analyzed for As bound to the silicate framework.
All the above separations were done by centrifugation at 3000 x g for 30 min. The supernatants were filtered and analyzed for As using GFAAS.
iv) In-vitro Bioaccesibility Method
The fraction of bioaccessible As was estimated following the method of Rodriguez et al. (1999) with certain modifications made by Sarkar and Datta (2003). Reactions were carried out in 250 mL beakers in a 37oC water bath to stimulate body temperature. Anaerobic conditions were maintained by passing argon gas through the solutions. The pH of the solutions was continuously monitored. The in-vitro gastrointestinal method was conducted in two sequential phases, a low pH gastric phase, followed by a higher pH intestinal phase.
1. Stomach phase: The gastric-phase solution consisted of 0.15 M NaCl and 1% porcine pepsin (Sigma Chemical Co., St. Louis, Missouri). One gram of soil sample was added to 150 mL of gastric solution, and the pH of the solution was adjusted to 1.8 using 1 N HCl. The solution was incubated for 1 hr, at the end of which 10 mL of the solution was collected, centrifuged at 3000 x g for 30 min and analyzed by GFAAS.
2. Absorbed-intestinal phase: At the end of the stomach phase, 10 mL of the gastric solution was added to replace the solution removed for analysis The pH of the solution was adjusted to 7.0 using a saturated solution of NaHCO3, followed by the addition of 525 mg of porcine bile extract and 52.5 mg of porcine pancreatin (Sigma Chemical Co., St. Louis, Missouri). To simulate absorption through the intestinal lining a 40-cm2 filter paper strip coated with ferric oxide was used (Sarkar and O’Connor, 2001). The ferric oxide strip was placed in a square bag (sides 6.5 cm) made of nylon membrane filter of 8 μm pore size. The bag was tied with a string and suspended in the reaction vessel. The solution was incubated for 1 hr, at the end of which 10 mL of the solution was collected, centrifuged at 5000 rpm for 30 min and analyzed by GFAAS. As adsorbed by the ferric oxide strip was desorbed by shaking it vigorously in 80 mL of 1N HNO3 for 1 hr.
3. Preparation of ferric oxide strips: Ferric oxide strips were prepared according to Sarkar and O’Connor (2001). In brief, Whatman no 8 filter papers were immersed in a 10-g/100-mL ferric chloride solution for 1 hr. The filter-papers were air-dried, followed by immersion in a 2.7 M NH4OH solution for 1 min for deposition of ferric oxide. The iron oxide-coated papers were air-dried and used to mimic As adsorption in the in-vitro absorbed-intestinal phase.
All analyses were carried out in triplicates and the results are shown as mean values.
v) Physico-Chemical Characterization of Soils and WTRs
Two soils (Immokalee and Orelia series) and two types of WTRs (Al- and Fe-based) were used for the greenhouse study. Immokalee series soil was collected from the Southwest Florida Research and Education Center, Immokalee, Florida, and the Orelia series from Corpus Christi, Texas. Fe- and Al-WTRs used for the study were obtained from Tampa, Florida and Bradenton, Florida water treatment facilities, respectively. All the samples (soils and WTRs) were air-dried and passed through a 2 mm sieve prior using them to soil characterization studies. Soil pH, electrical conductivity and particle size (only for soils) were measured using standard protocols (Klute, 1996). Organic matter was measured using the loss-on-ignition method (Klute, 1996). Total C and N were determined by combustion at 1010ºC using a Carlo Erba NA-1500 CNS analyzer. Oxalate-extractable Fe and Al concentrations of the soils and WTRs were determined using Tamm’s reagent (Klute, 1996). Total-recoverable Fe and Al concentrations were measured in acid digests by USEPA 3050B method (USEPA, 2000). Inductively Coupled Plasma Mass Spectrometer (ICP-MS), Perkin Elmer Elan 9000 model, was used for elemental determinations. Needed QA/QC procedures were followed to assure a recovery of 90-110% of spikes and standards.
vi) Greenhouse Study Design
A 3y greenhouse study involved two soils (Immokalee and Orelia series) amended with two arsenical pesticides, namely sodium arsenate (arsenic occur as +5 oxidation state) and Dimethylarsinic acid (the most commonly used organic arsenical pesticide). In the beginning of the greenhouse experiment, both the soils were spiked with sodium arsenate (Na2AsO4.7H2O- KR Grade, Aldrich, USA) and cacodylic acid ((CH3)2AsO2H- Reagent Grade, Alfa Aesar, USA) to reach the load of 1500 mgAs kg-1. Moreover, soils were mixed thoroughly with Al- and Fe-WTRs at two rates (5% and 10% w/w) expected to be effective in reducing soluble As. The high soil As value has been chosen following the assessment of Ng et al. (1993) who estimated soil arsenic in cattle dip sites to range between 700-2000 mg kg-1. Control was the soils amended with arsenical pesticides and no WTR. These amended samples were shifted to the columns in the greenhouse. Soil columns were made of PVC (0.38m high x 0.15m diameter). An outlet nozzle was made at the bottom of each column and connected with a tube to collect the leachate in 1.0 L clean plastic bottle. To prevent the soil washing from the column, a fine nylon mesh (8 μm mesh size) was incorporated at the bottom of the column and above the outlet nozzle. Each column is filled with 0.18m height of play sand, followed by 0.15m of amended soil. There were two columns (4 replicates) for each treatment. Rice, a fast growing and high biomass plant, was the test crop grown for a period of 0.5 yr. The columns were maintained at 70% water holding capacity. Columns were arranged in a randomized block design and were rotated periodically to account for variations in temperature and sunlight within the greenhouse.
The first soil sampling was done immediately after spiking (time zero), and then after 0.25, 0.5, 1 yr of equilibration time. The columns were over-watered in order to introduce leaching after 0.25 yr of equilibration time. The leachates were analyzed for soluble As. Plant samples were harvested twice (0.25 and 0.5 yr) during the entire experimental period. These samples were digested and analyzed for As using standard protocol (Carbonell et al., 1998). Columns were retained for longer periods (without grass cover) to understand the effect of soil/WTR-aging on As speciation and bioaccessibility. After 1st year, soils were sampled on an annual basis until the end of 3rd year. Soil samples were analyzed for total soil As (USEPA, 2000), in vitro bioaccessibility (Sarkar and Datta, 2003) and sequentially extracted forms (Chunguo and Zihu, 1988) after each sampling as outlined in sections iii and iv.
vii) Plant Tissue Digestion Protocol
Total As in rice shoot and root was determined using acid digestion method by Carbonell et al. (1998). A 0.5 g of dry plant tissue was digested with 5 mL of HNO3 for 3 hrs at 95°C. Then 5 mL of 30% H2O2 was added and the sample was heated at 95°C for another 1 hr. After cooling, the total digest was transferred by making the volume up to 50-mL in a volumetric flask. Samples were analyzed for As with Perkin Elmer Elan 9000 ICP-MS. All tests were performed in triplicate for each plant material. Adequate blanks, duplicates and matrix spikes were analyzed to meet QA/QC requirements.
IV. RESULTS AND DISCUSSION
i) Soil Arsenic Fractionation (sodium arsenate):
Four soils (Immokalee, Millhopper, Pahokee, and Orelia) differing in their physicochemical properties were subjected to a sequential fractionation scheme for a period up to 3-years. For the sodium arsenate amended Immokalee soil, the water-extractable As fraction represented the largest As fraction, regardless of soil aging up to three years. Specifically, water-extractable As concentrations decreased from 94% at time zero to 48% after three years (Figure 1). A concomitant increase was observed for the Fe/Al As fraction, suggesting that the portion of As being unavailable was due to sorption by the low content of Al/Fe hydroxides. Similar increase was also observed for the exchangeable fraction as a function of soil aging. Minimal partitioning of As was observed for the Ca/Mg, organic and residual As fraction throughout the 3-year period. Similar As fractionation data were obtained for the 1500ppm sodium arsenate load (Figure 2), suggesting that increases in As load did not influence the pattern of As fractionation in the Immokalee soil (Figure 2).
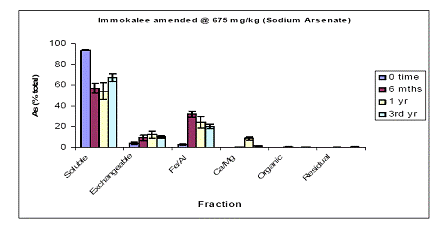
Figure 1. Sequential soil As fractionation data for the Immokalee soil treated with 675 mg/kg of sodium arsenate pesticide. Data are the average of three replicates ± one standard deviation.

Figure 2. Sequential soil As fractionation data for the Immokalee soil treated with 1500 mg/kg of sodium arsenate pesticide. Data are the average of three replicates ± one standard deviation.
For the Millhopper soil, the initial fractionation at time zero revealed that most of the soil As was extracted with the simple water phase (~ 90%) (Figure 3). The rest of soil As was measured in the exchangeable and the Fe/Al-hydroxide As phase (Figure 3). Soil aging had a prominent effect on Millhopper soil As fractionation. Water-extractable As concentrations decreased as a function of soil aging, reaching the lowest of 20% after 3-years of incubation (Figure 3). This reduction in the magnitude of water-extractable As concentrations were manifested in the concomitant increase of the Fe/Al hydroxide fraction. Arsenic concentrations in this fraction increased from 8 to 70% within 6- months and remained unchanged thereafter (up to 3 years) (Figure 3). Negligible As concentrations were measured in the organic, residual and Ca/Mg As fractions. Similar data to the 675 ppm As load were observed for the 1500 ppm load (Figure 4).

Figure 3. Sequential soil As fractionation data for the Millhopper soil treated with 675 mg/kg of sodium arsenate pesticide. Data are the average of three replicates ± one standard deviation.

Figure 4. Sequential soil As fractionation data for the Immokalee soil treated with 1500 mg/kg of sodium arsenate pesticide. Data are the average of three replicates ± one standard deviation.
For the Pahokee soil, soil As fractionation data showed the majority of As being partitioned at time zero in the Fe/Al hydroxide As fraction (60%) (Figure 5). About 25% was partitioned in the water-soluble As phase, and minimal amounts of As were measured in the exchangeable (10%) and the Ca.Mg phase (5%) (Figure 5). Soil aging resulted in further reduction in soil As being most available, since water-extractable As concentrations further decreased to 10% within 6 mo, and remained unchanged thereafter. This slight reduction was indeed reflected by increases (minor) in both the Fe/Al and Ca/Mg As fractions (Figure 5). Again, similar data were obtained for the soil As fraction of Pahokee when the initial As load was 1500 ppm (Figure 6).
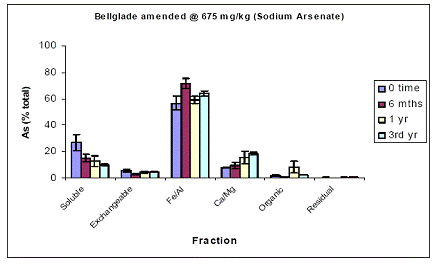
Figure 5. Sequential soil As fractionation data for the Belleglade soil treated with 675 mg/kg of sodium arsenate pesticide. Data are the average of three replicates ± one standard deviation.
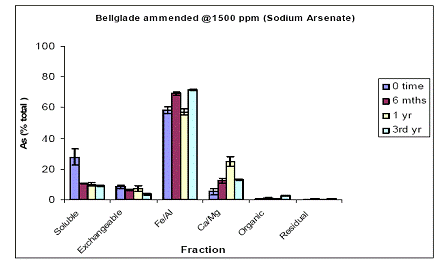
Figure 6. Sequential soil As fractionation data for the Belleglade soil treated with 675 mg/kg of sodium arsenate pesticide. Data are the average of three replicates ± one standard deviation.
For the Orelia soil, the greatest amount of soil As at time zero was measured in the water-soluble phase (75%), followed by the Fe/Al hydroxide As phase (12%) and minor constituents of the exchangeable, Ca/Mg and residual As phases (Figure 7). Soil aging decreased the magnitude of water-soluble As to 50% after a three year incubation. This decrease was illustrated in the Fe/Al hydroxide As fraction which increased from 10 to 40% within the first 6 months and stayed unchanged up to 3 years of incubation (Figure 7). Again, similar data were obtained for the soil As fraction of Pahokee when the initial As load was 1500 ppm (Figure 8).
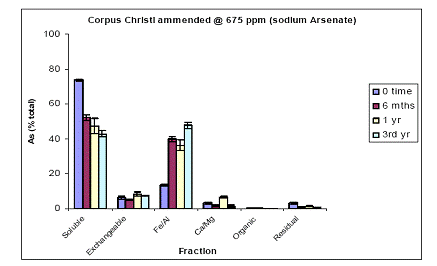
Figure 7. Sequential soil As fractionation data for the Orelia soil treated with 675 mg/kg of sodium arsenate pesticide. Data are the average of three replicates ± one standard deviation.
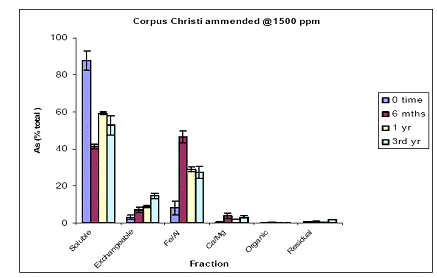
Figure 8. Sequential soil As fractionation data for the Orelia soil treated with 1500 mg/kg of sodium arsenate pesticide. Data are the average of three replicates ± one standard deviation.
ii) Soil Arsenic Fractionation (DMA):
The same four soils earlier described were also spiked with another arsenical pesticide-DMA. At time zero for the Immokalee soil, all of the soil As was measured in the water-extractable As fraction (Figure 9). There was a significant (p<0.05) decrease in the water-extractable As concentrations of Immokalee soil after 6-months of incubation to 65%, and a parallel increase in the exchangeable and Fe/Al hydroxide As fraction of the soil. Extending the incubation to 3 years did not significantly change the fractionation of soil As into these operationally defined fractions (Figure 9). Use of a greater load (1500 ppm As added as DMA) did not change the observed patterns (Figure 10).
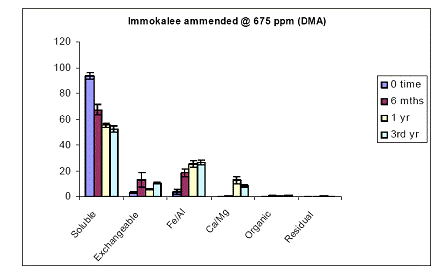
Figure 9. Sequential soil As fractionation data for the Immokalee soil treated with 675 mg/kg of DMA pesticide. Data are the average of three replicates ± one standard deviation.
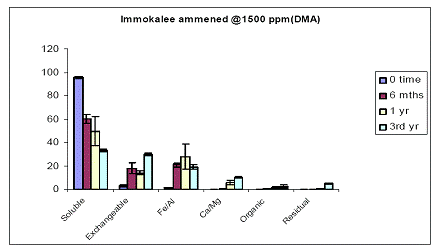
Figure 10. Sequential soil As fractionation data for the Immokalee soil treated with 1500 mg/kg of DMA pesticide. Data are the average of three replicates ± one standard deviation.
In the case of Millhopper soil, water-soluble As constituted almost 100% of soil As at time zero (Figure 11). After 6 months of incubation, water-extractable As concentrations decreased to 35%, due to soil aging. Water-extractable As concentrations remained unchanged up to a year of incubation, but there was a further reduction in water-extractable As concentrations to 20 % of total soil As after 3-years of incubation (Figure 11). The reduction in water-extractable As initiated a concomitant increase in the magnitude of the Fe/Al hydroxide As fraction from 2% (time zero) to 60% (6 and 12 months). Similar to the water-extractable As kinetic, there was a significant increase in the magnitude of Fe/Al hydroxide fraction (up to 70%) after a three-year incubation period (Figure 11). Similar data were obtained when using a greater DMA load (Figure 12).
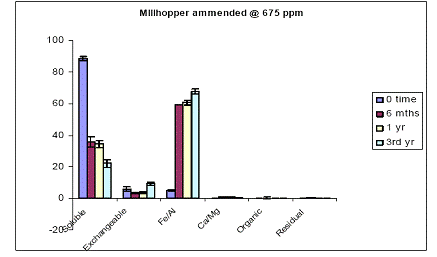
Figure 11. Sequential soil As fractionation data for the Millhopper soil treated with 675 mg/kg of DMA pesticide. Data are the average of three replicates ± one standard deviation.
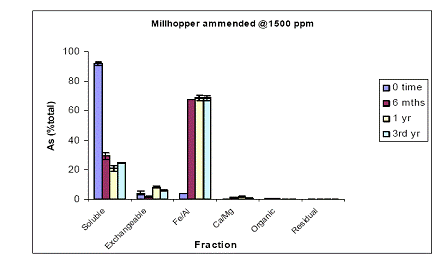
Figure 12. Sequential soil As fractionation data for the Millhopper soil treated with 1500 mg/kg of DMA pesticide. Data are the average of three replicates ± one standard deviation.
For the Pahokee soil, soil As fractionation data showed the majority of As being partitioned at time zero in the Fe/Al hydroxide As fraction (60%) (Figure 13). About 30% was partitioned in the water-soluble As phase, and minimal amounts of As were measured in the exchangeable (5%) and the Ca/Mg phase (5%) (Figure 5). Soil aging resulted in further reduction in soil As being most available, since water-extractable As concentrations further decreased to 10% within 6 mo, and remained unchanged thereafter. This slight reduction was indeed reflected by increases (minor) in both the Fe/Al and Ca/Mg As fractions (Figure 13). Again, similar data were obtained for the soil As fraction of Pahokee when the initial As load was 1500 ppm (Figure 14).
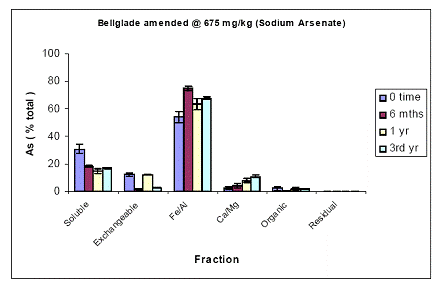
Figure 13. Sequential soil As fractionation data for the Belleglade soil treated with 675 mg/kg of DMA pesticide. Data are the average of three replicates ± one standard deviation.
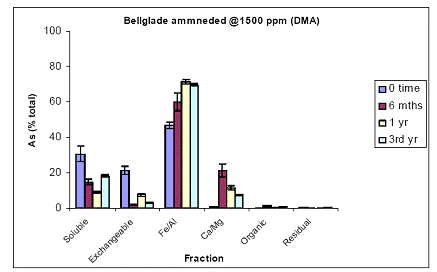
Figure 14. Sequential soil As fractionation data for the Belleglade soil treated with 1500 mg/kg of DMA pesticide. Data are the average of three replicates ± one standard deviation.
For the Orelia soil, the greatest amount of soil As at time zero was measured in the water-soluble phase (80%), followed by the Fe/Al hydroxide As phase (10%) and minor constituents of the exchangeable, Ca/Mg and residual As phases (Figure 15). Soil aging decreased the magnitude of water-soluble As to 40% after a three year incubation. This decrease was illustrated in the Fe/Al hydroxide As fraction which increased from 10 to 50% within the first 6 months and stayed unchanged up to 3 years of incubation (Figure 15). Again, similar data were obtained for the soil As fraction of Pahokee when the initial As load was 1500 ppm (Figure 16).
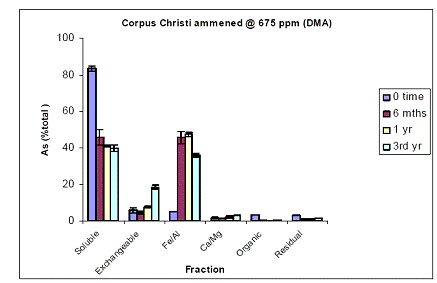
Figure 15. Sequential soil As fractionation data for the Orelia soil treated with 675 mg/kg of DMA pesticide. Data are the average of three replicates ± one standard deviation.
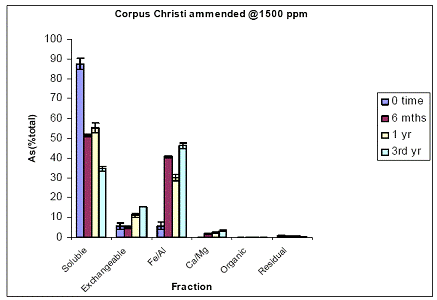
Figure 16. Sequential soil As fractionation data for the Orelia soil treated with 1500 mg/kg of DMA pesticide. Data are the average of three replicates ± one standard deviation.
Assuming that DMA did not change form via several biotic degradation reactions to the inorganic As(V) or As(III) species, then there seems to be no effect of arsenic pesticide source on the distribution of soil As in As-contaminated soils. In addition, soil aging seems to proceed in similar kinetic pattern, regardless of the original As pesticide source.
iii) In-Vitro Soil As Bioaccessibility(sodium arsenate)
Soil As bioavailability in the human gastric phase was simulated with a gastric test for soil As bioaccessibility for all four soils. At the initial load of 675 ppm As added in the form of sodium arsenate pesticide, results showed that in-vitro soil As bioaccessibility was ~ 90 % for all four soils (Figure 17). Specifically, there was no soil aging effect on the soil As bioaccessibility values of the gastric phase test (Figure 17). Even after three years of incubation, there was no significant difference in the gastric As bioaccessibility values between time zero and three years for all four soils studied. Despite the vast differences in their physical and chemical properties, soil As bioaccessibility did not change within the three year incubation period. Similar data were obtained for soil As bioaccessibility data obtained with the intestinal phase in-vitro test. There was no change in soil As bioaccessibility by extending the gastric to the intestinal phase test at any sampling period. Soil aging did not have any significant effect on the intestinal soil As bioaccessibility (Figure 18).
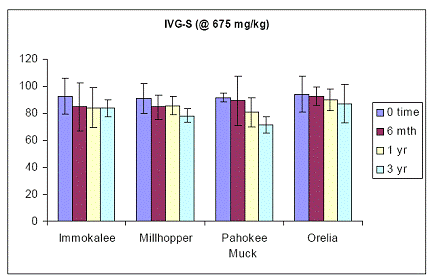
Figure 17. Soil As bioaccessibility changes as a function of soil aging with the human gastric phase test (in-vitro) for the four soils studied. Initial soil As load is 675 mg As(V) kg-1. Data are the average of three replicates ± one standard deviation.
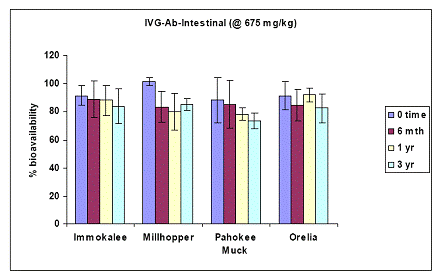
Figure 18. Soil As bioaccessibility changes as a function of soil aging with the human intestinal phase test (in-vitro) for the four soils studied. Initial soil As load is 675 mg As(V) kg-1. Data are the average of three replicates ± one standard deviation.
Increasing the load to 1500 ppm As, did not significantly change the trends observed with the 675 ppm data (Figures 19 and 20).
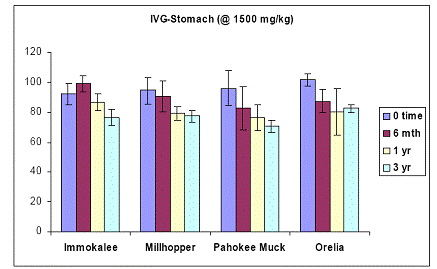
Figure 19. Soil As bioaccessibility changes as a function of soil aging with the human gastric phase test (in-vitro) for the four soils studied. Initial soil As load is 1500 mg As(V) kg-1. Data are the average of three replicates ± one standard deviation.
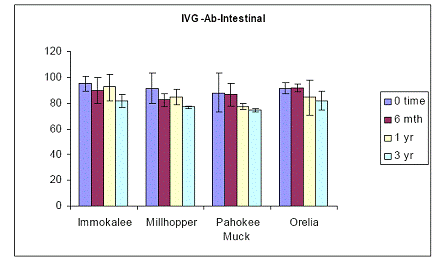
Figure 20. Soil As bioaccessibility changes as a function of soil aging with the human intestinal phase test (in-vitro) for the four soils studied. Initial soil As load is 1500 mg As(V) kg-1. Data are the average of three replicates ± one standard deviation.
iv) In-Vitro Soil As Bioaccessibility(DMA)
In the case of using the DMA-contaminated soil samples, in-vitro soil As bioaccessibility trends were similar to those obtained with the sodium arsenate pesticide. At the initial load of 675 ppm As added in the form of sodium arsenate pesticide, results showed that in-vitro soil As bioaccessibility was ~ 100 % for all four soils (Figure 17). Specifically, there was no soil aging effect on the soil As bioaccessibility values of the gastric phase test (Figure 21). Even, after three years of incubation, there was no significant difference in the gastric As bioaccessibility values between time zero and three years for all four soils studied. Similar data were obtained for soil As bioaccessibility data obtained with the intestinal phase in-vitro test. There was no change in soil As bioaccessibility by extending the gastric to the intestinal phase test at any sampling period. Soil aging did not have any significant effect on the intestinal soil As bioaccessibility (Figure 22).
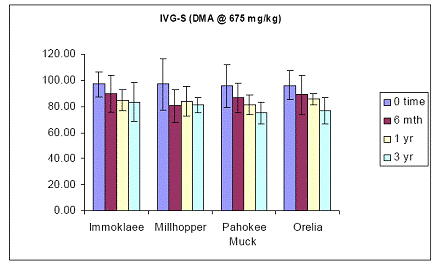
Figure 21. Soil As bioaccessibility changes as a function of soil aging with the human gastric phase test (in-vitro) for the four soils studied. Initial soil As load is 675 mg DMA kg-1. Data are the average of three replicates ± one standard deviation.
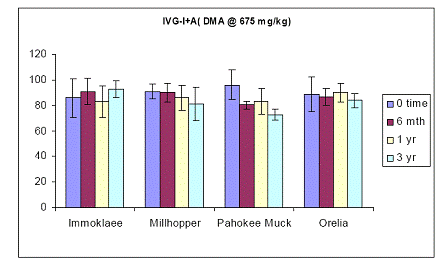
Figure 22. Soil As bioaccessibility changes as a function of soil aging with the human intestinal phase test (in-vitro) for the four soils studied. Initial soil As load is 675 mg DMA kg-1. Data are the average of three replicates ± one standard deviation.
In the case of soils amended with 1500 ppm As in the form of DMA, trends in soil As bioaccessibility varied somewhat. Immokalee soil showed no soil aging effect, but the other three soils showed significant reduction in the in-vitro soil As bioaccessibility between time zero and after three years of incubation (Figure 23). This trend was not observed in the intestinal phase, since the Immokalee, Millhopper and Orelia soils did not change the intestinal soil As bioaccessible concentrations during the three-year incubation, except the Pahokee soil, which is a organic-muck soil (Figure 24).
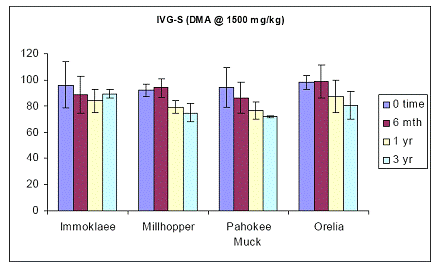
Figure 23. Soil As bioaccessibility changes as a function of soil aging with the human gastric phase test (in-vitro) for the four soils studied. Initial soil As load is 1500 mg DMA kg-1. Data are the average of three replicates ± one standard deviation.
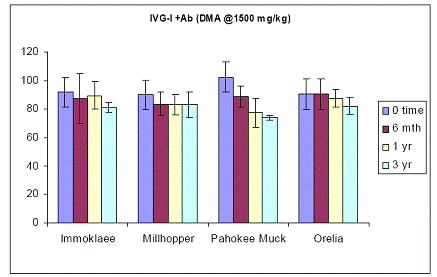
Figure 24. Soil As bioaccessibility changes as a function of soil aging with the human intestinal phase test (in-vitro) for the four soils studied. Initial soil As load is 1500 mg DMA kg-1. Data are the average of three replicates ± one standard deviation.
v) Arsenic Mass Balance:
After the completion of the three year incubation experiment in the greenhouse, columns were sacrificed, rice plants were harvested and cumulative leached As were calculated to determine the distribution of As in the soil-plant-leachate interactive system. In the case of the Immokalee soil, > 90% was measured in the leachate, and the rest 10% was sorbed by the soil particles; minimal amounts of As were taken up by rice plants (Figure 25).
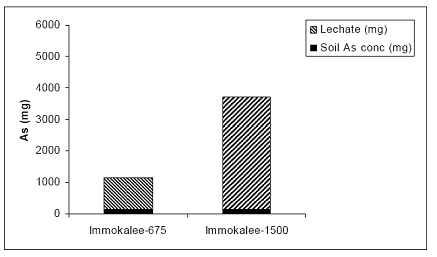
Figure 25. Arsenic mass balance in the greenhouse-based system of the Immokalee soil-plant-leachate components. Arsenic partitioned to rice plants was negligible, relative to the soil and leachate compartments and it not included here during the three year greenhouse study.
In the case of Millhopper soil, mass balance analysis showed that most of the added As was equally shared between the soil and the leachate As fraction. Obviously, the greater amount of the Al/Fe hydroxide fraction in the Millhopper increased As sorption, and decreased the potential for As to leach out the system (Figure 26). Again, minimal amounts of As were measured in the tissues of rice grown on this soil. Arsenic load had no effect on the mass balance calculations.

Figure 26. Arsenic mass balance in the greenhouse-based system of the Millhopper soil-plant-leachate components. Arsenic partitioned to rice plants was negligible, relative to the soil and leachate compartments and it not included here during the three year greenhouse study.
In the case of the muck soil, there was a 90% partitioning in the soil solid phase and only 10 % were measured in the leachate; none was detected in the rice plants at the 675 ppm As load (Figure 27). Arsenic load had a significant effect on the partitioning of added As, changing the amount of As sorbed by the soil. Specifically, As partitioning in soil decreased to approximately 75% and the rest was measured in the leachate, suggesting that the increase in the As load saturated the soil sorption sites.
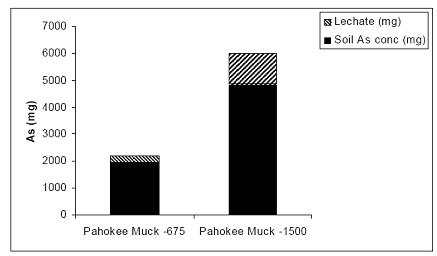
Figure 27. Arsenic mass balance in the greenhouse-based system of the Belleglade (Pahokee) soil-plant-leachate components. Arsenic partitioned to rice plants was negligible, relative to the soil and leachate compartments and it not included here during the three year greenhouse study.
In the case of the Orelia soil, most of the As was measured in the soil solid phase (> 90%) (Figure 28). Minimal (< 10%) was measured in the leachate and none in the rice tissues, suggesting that Orelia soil had a high As sorption capacity (Figure 28). Arsenic load had no effect on the mass balance analyses of As. Orelia and Pahokee showed the greatest sorption of As in the greenhouse scale experiments.
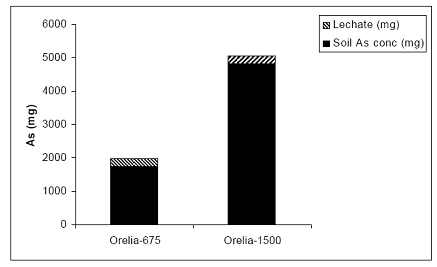
Figure 28. Arsenic mass balance in the greenhouse-based system of the Orelia soil-plant-leachate components. Arsenic partitioned to rice plants was negligible, relative to the soil and leachate compartments and it not included here during the three year greenhouse study.
vi) Soil Arsenic(V) Fractionation:
A) WTR Effect on Immokalee soil: The effect of WTR added to the contaminated soils on soil As fractionation was also assessed in the greenhouse experiments. In the unamended (no WTR) Immokalee soil, water-extractable As concentrations were very high (> 80%) at time zero of the experiment, but soil aging (after 3 years) resulted in significant reduction to approximately 50% (Figure 29). Upon the mixing of the contaminated soil with 5% Fe-WTR, there was a significant reduction in water-extractable As concentrations for all sampling times (time zero up to 3-years). Water-extractable As concentrations were reduced from 65% to 30% and < 10% after 0.25 and 0.5 years at the 5% Fe-WTR application rate (Figure 29). No further significant reduction in water extractable concentrations were observed after 1 and after 3-years of equilibration in the greenhouse, suggesting the stability of the beneficial WTR effect in immobilizing soil As in-situ. When the WTR application rate was increased to 10%, there was a slight increase in the beneficial effect of WTR on the reduction of water-extractable As concentrations, although not significant (p>0.05) (Figure 30). The observed reduction in soil As bioavailability was in parallel to the concomitant increase in the magnitude of the Fe/Al hydroxide As fraction for both 5% and 10% Fe-WTR application rate (Figures 29 and 30). Again 0.5 years were adequate to stabilize the reduction of As availability to low levels in the WTR-amended soil.
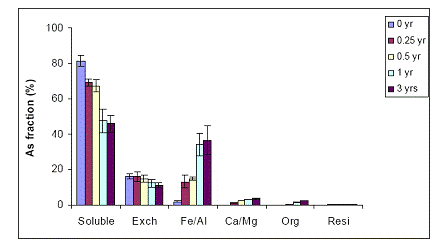
Figure 29. Sequential As fractionation data as a function of soil aging for the unamended (no WTR) Immokalee soil in the greenhouse. Data are the average of three replicates ± one standard deviation.

Figure 30. Sequential As fractionation data as a function of soil aging for the 5% Fe-WTR application rate on the Immokalee soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
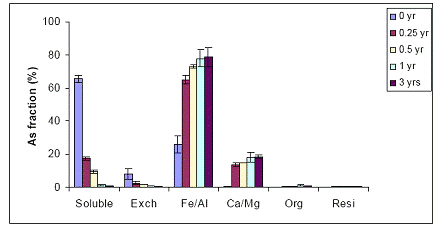
Figure 31. Sequential As fractionation data as a function of soil aging for the 10% Fe-WTR application rate on the Immokalee soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
In the case of the Al-WTR Immokalee soil samples, there was also a significant reduction in the water-extractable As concentrations for all sampling times (time zero up to 3 years). Water-extractable As concentrations were reduced from 70% to 25% and < 10% after 0.25 and 0.5 years at the 5% Al-WTR application rate, similar to the data obtained with the Fe-WTR, suggesting the effectiveness of both WTRs in decreasing soil As availability (Figure 32). No further significant reduction in water extractable concentrations were observed after 1 and after 3-years of equilibration. When the WTR application rate was increased to 10%, there was a slight increase in the beneficial effect of WTR on the reduction of water-extractable As concentrations, although not significant (p>0.05) (Figure 33). The observed reduction in soil As bioavailability was in parallel to the concomitant increase in the magnitude of the Fe/Al hydroxide As fraction for both 5% and 10% Al-WTR application rate (Figures 32 and 33). Again 0.5 years were adequate to stabilize the reduction of As availability to low levels in the WTR-amended soil.
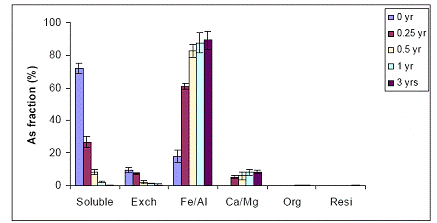
Figure 32. Sequential As fractionation data as a function of soil aging for the 5% Al-WTR application rate on the Immokalee soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
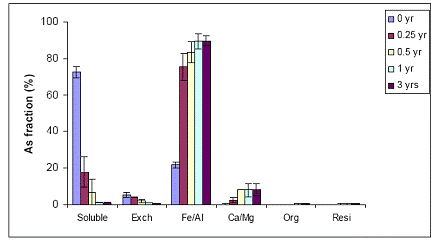
Figure 33. Sequential As fractionation data as a function of soil aging for the 10% Al-WTR application rate on the Immokalee soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
B) WTR Effect on Orelia soil: The Orelia soil contains greater amount of Fe/Al hydroxides, which seems to be the dominant fraction of As sorption by the soil. The unamended Orelia soil samples exhibited a greater soil aging effect on water-extractable As concentrations when compared with the sandy Immokalee soil (Figure 34). There was a gradual reduction in water-extractable As concentrations of the Orelia soil, reaching 40% and 20% after 1 and 3 years, respectively of reaction in the greenhouse (Figure 34). The Orelia soil samples amended with 5% Fe-WTR exhibited a significant (p< 0.005) reduction in water-extractable As concentrations at all sampling time intervals (Figure 35). Similar to data obtained with the Immokalee soil, there was a significant kinetic effect on the WTR effectiveness in decreasing water-extractable As concentration is in the 5% Fe-WTR rate treatment (Figure 35). The Fe-WTR effectiveness was unaffected by differences in soil chemical properties between Orelia and Immokalee soils. Despite the inherent ability of the Orelia soil in decreasing water-extractable As concentrations, when the Fe-WTR was mixed with the contaminated soil there was a significant WTR effect. In specific, the Fe-WTR effect was complete within 0.5 years of equilibration in the greenhouse soil PVC columns (Figures 35 and 36). The 5 and 10% Fe-WTR amended soil samples exhibited a significant decrease in water-extractable As concentrations of the Orelia soil within 0.5 years of incubation, by reaching ~ 10% of the total soil As. The decrease in water-extractable As concentrations was attributed to sorption by the Fe/Al hydroxide As soil fraction that increased in parallel (Figures 35 and 36).
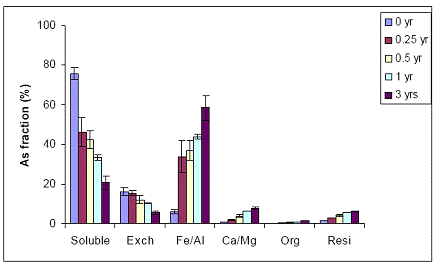
Figure 34. Sequential As fractionation data as a function of soil aging for the unamended (no WTR) Orelia soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
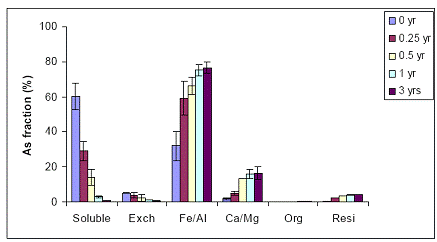
Figure 35. Sequential As fractionation data as a function of soil aging for the 5% Fe-WTR amended Orelia soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
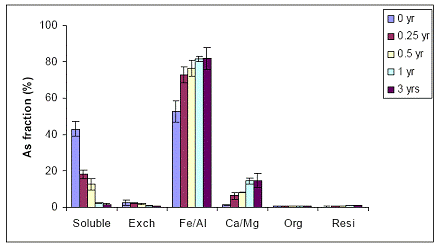
Figure 36. Sequential As fractionation data as a function of soil aging for the 10% Fe-WTR amended soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
When the Orelia soil was amended with the Al-based WTR, similar As fractionation data were obtained to the Fe-WTR (Figures 37 and 38). The Al-WTR did not differ in the effectiveness of WTR in decreasing soil As availability, by decreasing water-extractable As concentrations via sorption to Fe/Al hydroxides.
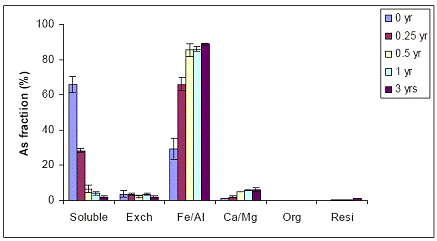
Figure 37. Sequential As fractionation data as a function of soil aging for the 5% Al-WTR amended soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
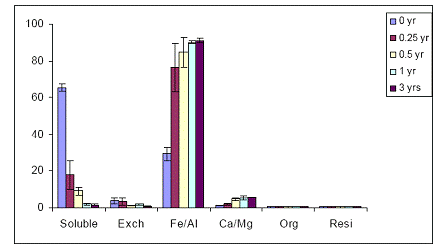
Figure 38. Sequential As fractionation data as a function of soil aging for the 10% Al-WTR amended soil in the greenhouse. Data are the average of three replicates ± one standard deviation.
vii) Soil DMA Fractionation:
WTR Effect on Immokalee and Orelia soils: The WTR effect in decreasing soil As availability was also assed in DMA-treated Immokalee soil. Very similar As fractionation data were observed for the WTR effect between the As(V)- and DMA-treated Immokalee and Orelia soils. The source of As did not have any effect on the beneficial WTR effect in immobilizing As in both soils. It seems that both WTR (Fe- and Al-based) are capable of immobilizing As(V) and DMA onto their surfaces, assuming that DMA did not undergo transformation to inorganic As. Arsenic speciation data on both soils treated with DMA will be presented later in this final report.
viii) Soil As(V) Bioaccessibility:
A) WTR Effect on Immokalee soil: In the unamended control Immokalee soil samples (no WTR added), there was no significant decrease in the in-vitro bioaccessible As concentrations even after 3-year of equilibration of the soil in the greenhouse (Figure 39). On the contrary, WTR-amended soil samples showed a gradual decrease in the in-vitro bioaccessible As concentrations, w
2004 Progress Report
2005 Progress Report
2006 Progress Report
Journal Articles:
No journal articles submitted with this report: View all 47 publications for this projectSupplemental Keywords:
Health, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, Waste, Water, POLLUTANTS/TOXICS, Hazardous, Risk Assessments, Contaminated Sediments, Hazardous Waste, Geochemistry, Arsenic, Environmental Monitoring, Water Pollutants, arsenic mobility, contaminant transport, assessment methods, contaminated sediment, sediment quality survey, water quality, biogeochemistry, contaminated soil, bioaccumulation, sediment transport, water treatment, ecology assessment models, superfund site, reservoir sediments, risk management, arsenic exposureProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
Site Navigation
Related Information
Contact Us to ask a question, provide feedback, or report a problem.
Last updated April 28, 2023
