Grantee Research Project Results
Final Report: Dose-Response of Nitrate and Other Methemoglobin Inducers on Methemoglobin Levels of Infants
EPA Grant Number: R829781Title: Dose-Response of Nitrate and Other Methemoglobin Inducers on Methemoglobin Levels of Infants
Investigators: VanDerslice, Jim
Institution: Washington State Department of Health
EPA Project Officer: Page, Angela
Project Period: September 2, 2002 through September 1, 2005 (Extended to September 1, 2007)
Project Amount: $594,934
RFA: Health Effects of Chemical Contaminants in Drinking Water (2001) RFA Text | Recipients Lists
Research Category: Drinking Water , Human Health , Water
Objective:
INTRODUCTION
In Washington State nitrate is the most commonly found contaminant of drinking water wells, both private wells and wells serving public water supplies (1,2). Recent studies indicate that almost one-fourth of the wells sampled in the Columbia Basin had nitrate levels greater than the U.S. Environmental Protection Agency (USEPA) Maximum Contaminant Level (MCL) of 10 mg/L (2). Given that over 80 percent of these wells were either domestic or public water supplies, there is a great potential for human exposure to high levels of nitrates.
Ingestion of water containing elevated levels of nitrate may, in some situations, lead to methemoglobinemia, a condition that renders the hemoglobin in an individual’s red blood cells less capable of transporting oxygen from the lungs to the rest of the body (3). At particular risk are infants less than 1 year old, any person with reduced gastric acidity, or persons lacking cytochrome b5 reductase, an enzyme that changes comethemoglobin back to normal hemoglobin (3,4). Individuals who are diagnosed with this condition can be effectively treated. However, given sufficient exposure and left untreated, the condition can be fatal. Normal levels of methemoglobin have been cited as ≤1 percent of total hemoglobin to < 2 percent (3,4,5,6).
The current MCL for nitrate is based primarily on a study of 139 cases in Minnesota in 1950 (7) and a survey in 1951 of state health officials regarding methemoglobinemia cases (8). These studies found that methemoglobinemia cases occurred only when the nitrate level in the infant’s drinking water was greater than 10 mg/L; however, only 5 of the 214 cases reported occurred when the level was less than 20 mg/L (8). The USEPA set the MCL for nitrate at 10 mg/L (9). Since the available data represented clinical cases of methemoglobinemia in a sensitive subpopulation (i.e. infants), no safety factors were used in setting the MCL.
A number of scientists have questioned the role of nitrate-contaminated water as a cause of methemoglobinemia. Some researchers have noted that bacterial infections, perhaps resulting from fecally-contaminated wells, may lead to the endogenous production of nitrite in the body, and that this may lead to the formation of methemoglobin (5,10,11,12,13,14). Other researchers have suggested that other chemicals found in drinking water may also induce the formation of methemoglobin (15). At the same time, public health officials cite cases that they claim demonstrate the risks of nitrate contaminated water supplies (16).
This ambiguity has made it difficult for local and state public health officials to effectively respond to the problem of nitrate contamination of non-federally regulated water systems and private wells. The widespread nature of the problem, the high cost of treatment to remove nitrate or to develop alternative water sources, the lack of observed cases of frank methemoglobinemia and the knowledge that the condition can be fatal combine to make this issue nearly intractable for local and state environmental health practitioners.
OBJECTIVES
The long-term goal of this research is to improve public health actions to reduce the risk associated with nitrate contamination of private and public water supplies, by improving our understanding of the exposures to chemicals which may form methemoglobin, and the relationships of those exposures to measured levels of methemoglobin among a highly susceptible sub-population, that is, infants in an area with high nitrate waters. The specific objectives of this study are:
1. To estimate the intake of methemoglobin inducers (i.e., nitrate, copper, chlorination products, medications) by infants and their effects on methemoglobin levels.
2. To estimate the effect of potential endogenous production of nitrite (i.e. symptoms of infection and GI distress) on the level of methemoglobin in infants.
3. To examine mothers’ knowledge and attitudes regarding the risks associated with the use of private well water for infants.
Summary/Accomplishments (Outputs/Outcomes):
III. METHODS
A. Study design
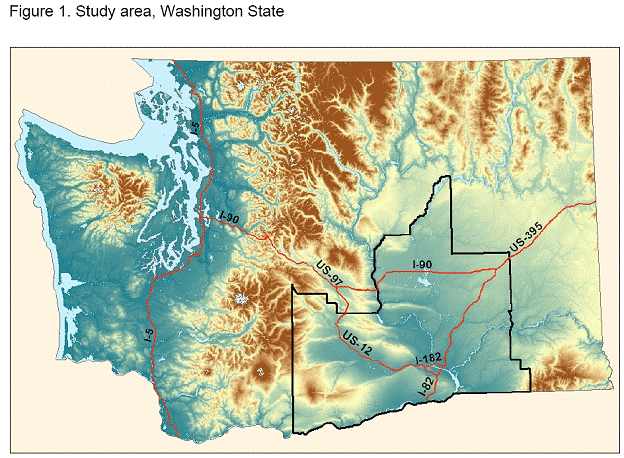
This was a community-based cross-sectional cohort study of infants 6 months of age or younger who lived in the study area. Infants who had any malformations or abnormalities at birth were excluded. The study area included the following counties of Washington state: Grant, Adams, Benton, Franklin, Yakima, Walla Walla, and Klickitat (Figure 1). The sampling process targeted infants whose families were not served by Community Water Supplies (CWS), that is, those not served by water systems regulated by the Safe Drinking Water Act. Data collected during a home visit included demographic, health, water use and diet information through a structured interview, nitrate and bacteriological analysis of household drinking water sources, length and weight, and methemoglobin (MetHb) levels.
B. Subject selection and recruitment
Infants were identified using birth certificate follow-back. Birth certificate data from all births in Washington State where the mother’s address was in one of the seven study counties, and which there was no indication of a birth defect, abnormality or other serious health problem, was provided by the Washington State Department of Health (DOH) Center for Health Statistics on a weekly basis. These records were geo-coded using a geo-coding process developed by DOH personnel which used street network and county parcel data to establish the geographic coordinates for each potential study subject.
Data from the Office of Drinking Water (ODW) at the DOH were used to estimate whether a given household was served by a CWS. For some cities, ODW had a GIS layer delineating the extent of the CWS distribution system. In other areas, information was collected from the utilities and some field validation was conducted to more precisely define the extent of the distribution system. ArcGIS® version 8 (ESRI, Redlands, CA) was used to determine which of the residences fell outside of the estimated service boundaries of the CWS. Infants who lived in residences which fell within these boundaries were not eligible. Infants whose mothers lived within the boundaries of the Yakima Indian Nation were also not included.
For each eligible infant the age at interview was randomly assigned to occur between birth and 6 months of age. A target interview date was calculated based on the target interview age and the date of birth. Approximately 3 weeks before the target interview date, a letter was sent on DOH letterhead to the mother explaining the study and asking her to call a toll-free number to schedule an appointment. If there was no response from the mother, a bi-lingual recruiter attempted to contact the mother via telephone. Up to five attempts were made covering different times and days. In cases where the recruiting letter was returned as undeliverable, efforts were made to locate and contact the mother using publically-available search engines.
When contact was made with the infant’s parent (usually the mother), the recruiter explained the purpose of the study, what participation would entail, and that participants would receive a $35 Safeway gift card for participating in the study. If the mother agreed to participate, then an appointment was set up. Regular appointments took place during the weekend; however, a few appointments were scheduled during the week to accommodate the parents’ work schedule.
A field team consisting of a nurse and a technician, one of whom was bilingual, went to the mother’s home at the appointment time. They explained the procedures for the study and asked if the parent wanted to participate. If the parent agreed, then the fieldworkers administered an informed consent. Data collection did not begin until the informed consent process was completed.
The study protocol was approved by the Washington State Institutional Review Board (DOH Project A-121703-H).
C. Data collection
Data collection included a structured interview with the infant’s caregiver(s), sampling and analysis of water used for drinking, measurement of the infant’s height and weight, and analysis of a sample of the infant’s blood to determine the level of MetHb.
1. Interviews
Interviews were conducted in either English or Spanish using a structured instrument. This instrument had been pre-tested for comprehension with Spanish and English-speaking mothers from the study area before being used in a previous study. The version used in this study incorporated minor modifications.
The first module covered several areas: who had cared for the child in the past 24 hours; the infant’s health, including any serious illness since birth, and the occurrence of specific common illnesses in the past 3 days (i.e., diarrhea, fever, vomiting, fussiness, earache). A 24-hour diet history was then collected starting with the most recent feeding and moving backwards in time. For each item, the amount prepared and the amount consumed was estimated by the mother based on model baby food containers or in terms of specific volumes (e.g., fluid ounces). These models included large and small bottles, small and medium sized bowls, and baby food jars. The amount and source of water used to prepare the food item, if any, was estimated by the mother. If the child was breastfed, then the duration and time of the feedings was recorded.
The next module covered the specific source of the household’s tap water, and for participants with private wells, whether the well had ever been tested by the family, and the mother’s perception of the water quality. Race, ethnicity, and language spoken in the home were the last items collected.
2. Anthropometric measures
After the interview, the baby was weighed on a portable baby scale and measured using a SECA baby measuring mat. This was carried out by the nurse.
3. Methemoglobin levels
The infant’s blood was analyzed for levels of oxyhemoglobin, carboxyhemoglobin and methemoglobin, as well as the concentration of hemoglobin using a portable co-oximeter (Avoximeter 4000, Avox Corporation, San Antonio, TX). The blood sample was obtained via heel stick, conducted by the nurse, immediately expelled into a specialized cuvette and analyzed. Before each analysis, the calibration was checked and recorded using colored standards supplied by the manufacturer.
4. Water quality
Samples of water were collected from the household’s kitchen tap, and from all other household sources of water the parent(s) reported giving to the infant. This included bottled water (both from small or 1-gallon containers and 5-gallon "water cooler" containers), pitchers (some of which had carbon filters), filtered faucets mounted on the sink dispensing filtered water, and water from taps mounted on the door of the refrigerator. A second water sample was collected from the kitchen tap after removing the aerator and running the water for 3 minutes.
Nitrate levels were determined using an ion-selective probe (HACH, Loveland, CO). At each household, the meter was calibrated using a three-point calibration curve (1, 5, 10 mg/L) and the calibration slope was checked against the manufacturer’s specifications. If the slope was not within these limits, the meter was re-calibrated, the membrane was replaced, a different probe was used, or if all these measures failed, an indicator strip was used to measure the nitrate level. The temperature of the standards and the water samples were checked to ensure that temperature differences did not affect the calibration. Copper, which can be a metHb-inducer, and chloride, which can interfere with the measurement of nitrate, were measured using indicator strips.
Samples for bacteriological analysis were collected using sterile technique into sterile 100 mL bottles. Samples bottles with sodium thiosulfate were used for households served by public water systems or when the household had its own chlorinator. In these cases, total and free chlorine were measured using indicator strips (HACH, Loveland CO). The samples were immediately put into a cooler with cool packs. At the end of the day, Colilert® media was added to the samples which were then incubated at 37ºC for 24 hours. A yellow color indicated the presence of total coliforms; fluorescence indicated the presence of E. coli.
Quality control procedures were carried out at approximately 10 percent of the houses. These houses were randomly selected and the fieldworkers did not know which houses were selected until the day of the interview. For the nitrate analysis, standards containing 0, 5, and 10 mg/L nitrate-nitrogen were analyzed. A split sample from the kitchen tap was also analyzed. For the bacteriological samples, positive and negative controls were run using distilled water and ATCC E. coli culture, respectively.
D. Participant feedback
One of the valuable aspects of this study was the ability to immediately give the parents feedback on the health of their child and their water quality, to answer questions about their infant’s development and the water quality from their well, as well as to offer referrals to the local environmental health specialist and other state and county programs.
The growth indicators, metHb levels and nitrate levels were immediately provided to the parents. Staff called the parents the following day with the water bacteriological results. A letter was sent to all parents within a week of the interview reviewing the major results and providing, in writing, a summary of the finding and recommendations for follow-up.
E. Data processing and reduction
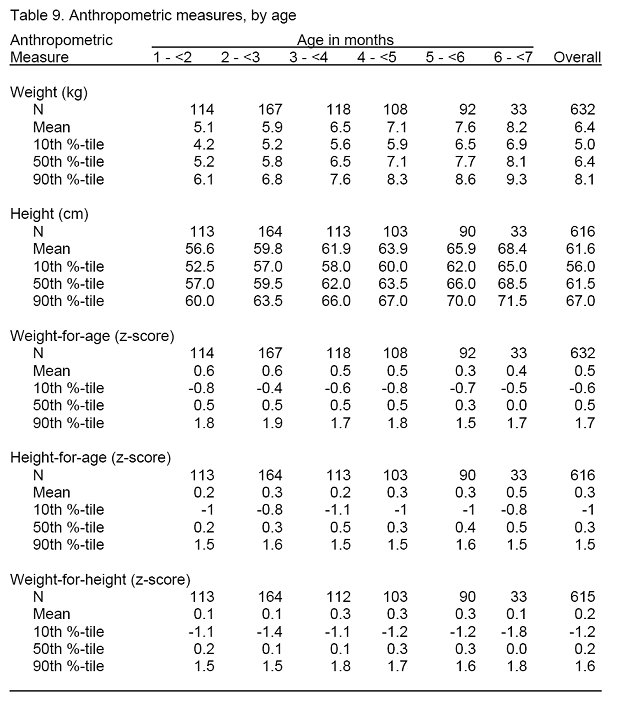
Data were entered into a MS Access® relational database with a custom front end. The front end application included automatic skip-patterns and out-of-range checks. The data were imported into a number of SAS® databases. SAS® version 9 (Cary, NC) was used to merge infant, water quality, anthropometry and diet data tables, create composite and summary variables, and conduct logical consistency checks. The original data forms were consulted to resolve discrepancies and to verify accurate data entry. Anthropometric measures based on the 2000 Centers for Disease Control and Prevention Growth Charts were generated using a SAS program provided for that purpose (17).
F. Data analysis
Data analyses were conducted using SAS® version 9 (Cary , NC), and STATA® version 10 (College Station, TX). Bivariate comparisons were conducted using t-tests and Mantel-Haenszel chi-square statistics, with a 5 percent significance. Multi-variable analyses were carried out using linear regression on the level of methemoglobin, and two logistic regressions on binary variables indicating that the methemoglobin level was greater than 2 and 3 percent of the total hemoglobin. Initial models included all risk factors with bi-variate associations with at least a 0.20 significance level. Risk factors that were non-significant in multi-variable models were removed sequentially, and the impact on the parameter estimates and standard errors of the remaining variables were noted. Exposure to bacteriologically contaminated water, nitrate intake, and age were retained in all but the final model due to their etiological significance and the objectives of this analysis. Interactions of age with nitrate intake and exposure to bacteriologically contaminated water were tested. Goodness-of-fit was assessed for the final model.
IV. RESULTS
A. Participation
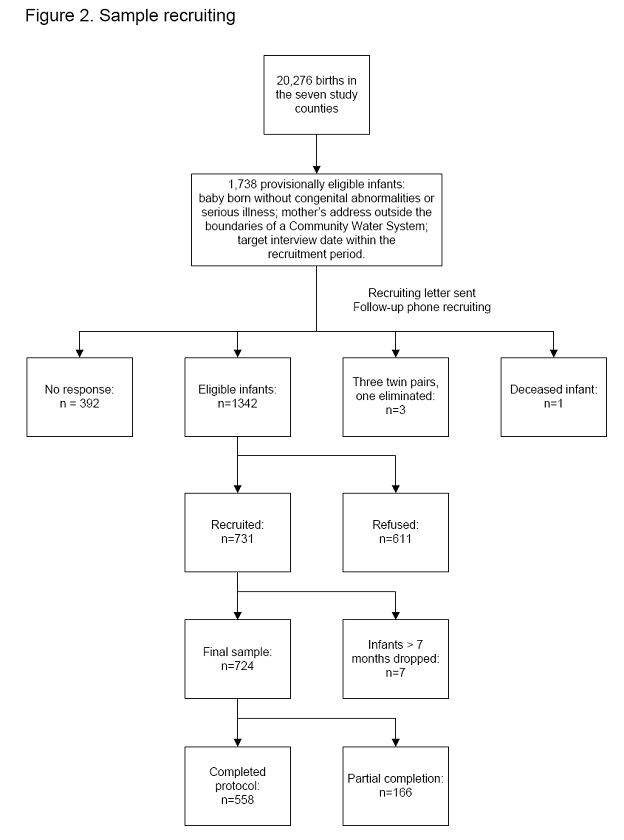
Some 20,276 births whose mother’s reported address was in one of the seven study counties were reported to the study PI during the study period, September 15, 2004, and October 15, 2005. The study period was extended 1 month past the planned recruitment period because of a low number of recruited infants during the first month of recruitment. Of these, 1,738 were provisionally eligible; that is, the baby was born without congenital abnormalities or other serious health problems noted on the birth certificate; the mother’s reported address was outside the approximate CWS distribution system boundaries available to the study; and the target date of the interview was within the recruitment period.
Of those provisionally eligible, 392 (23%) did not respond to the recruitment letter and were unable to be contacted by phone. In some cases, the letter was returned as undeliverable with no known forwarding address. We heard anecdotal reports of mothers giving incorrect addresses for the birth certificates due to fears associated with immigration status. Attempts to contact these parents by telephone were also unsuccessful because the telephone number was disconnected, incorrect, or not answered, despite multiple attempts at different times and days of the week. There were three sets of twins in this group, so one from each set was dropped. One infant had died by the time of recruitment, leaving 1,342 eligible infants.
Of these, 611 (45%) parents declined to participate; in 92 of these cases, the parent had initially agreed but then declined when the fieldworkers went to their home. As a result, 731 infants were enrolled in the study. Seven of the infants were 7 months of age or older at the time of the interview and were dropped from further analysis, leaving 724 eligible participants. While demographic and some water quality data were collected for all these infants, only 558 completed all parts of the protocol, including the diet history, anthropometry, and blood sampling. The results presented below use all data that were collected for each specific topic area.
Depending on how many of the non-contacts are assumed to have been eligible (i.e., actually living in the study area and outside a CWS service area) ,the response rate ranges from 41.8 percent (all non-contacts were eligible) to 64.5 percent assuming half the non-contacts were ineligible because they were no longer in the study area at the time of recruitment or not living outside a CWS area. Refusal to allow a blood draw was the most common reason for having only partial completion. This factor on its own accounted for 59 percent of the 166 partial responses. No nitrate water sample result was the next most important factor, on its own accounting for 13 percent of the partial completions. In 6 percent of the cases, only minimal information was collected before the respondent discontinued the interview.
B. Study population
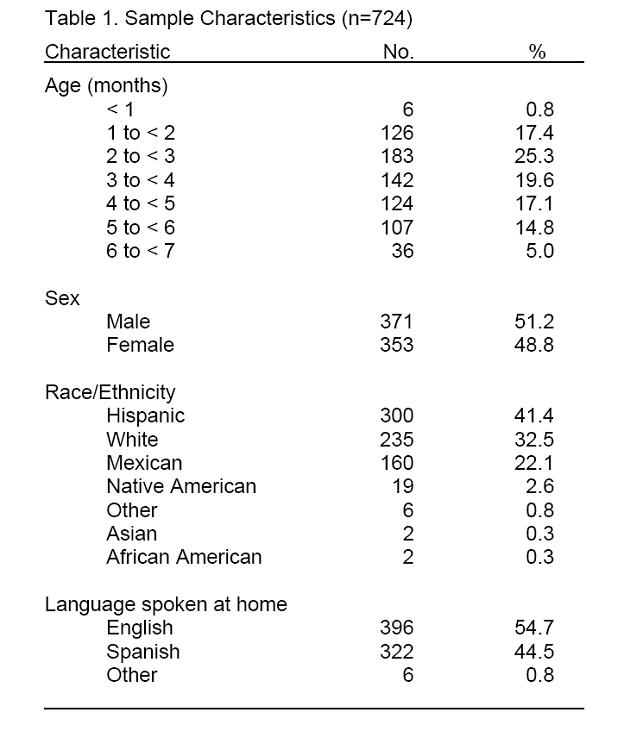
There were slightly more males in the final sample as compared to females (51.2% vs. 48.8%). The distribution of infants by age was relatively uniform, with the highest rate of participation occurring in the 2 to 3 month age group (Table 1). Only 6 infants completed the study before 1 month of age, due to the lag time in receiving the birth records from the hospitals and time needed to contact the women by mail to recruit them into the study. All of these infants were within 5 days of being 1 month old. As such, these infants are grouped with the 1 to < 2 month age group for the subsequent analyses.
English was the language most commonly spoken in the home (54.7%) followed by Spanish (44.5%). Four respondents reported speaking languages indigenous to Mexico. Of those who primarily spoke English, 24 percent also reported speaking a second language, predominantly Spanish. Only two of the respondents who predominantly spoke Spanish also spoke another language (indigenous dialect of Mexico).
Almost two-thirds of the study participants reported the infant’s ethnicity as Hispanic or Mexican (63.5%). Thirty-two percent identified their infant as white. Three percent were African American.
C. Current illness and use of medication
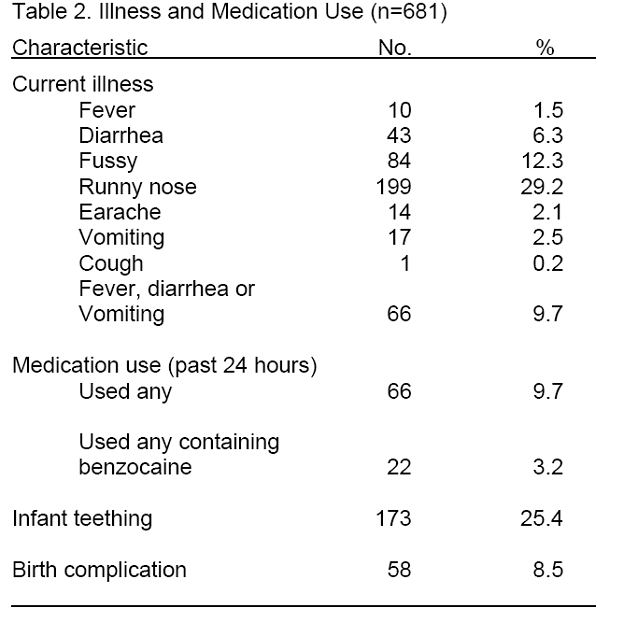
Overall 9.7 percent of the infants had had a fever, vomiting, or diarrhea up to and including the day of the interview (diarrhea 6.4%; vomiting 2.5%; fever 1.5%) (Table 2). Ear ache was reported for 2.0 percent of the infants, runny nose for 29.2 percent and "fussiness" for 12.4 percent.
Sixty-six infants had been given some type of medication. Most of these were medications associated with teething. While just over a quarter of the infants were teething at the time of the interview (26.1%, n=66), only 22 infants (3.2%) had been given a medication in the previous 24 hours which had an active ingredient that could form methemoglobin. In virtually all cases this was benzocaine, an ingredient of teething gels.
D. Water quality
1. Nitrate
The measured nitrate levels are presented in Table 3. Tap water samples were the highest with a mean of 3.5 mg/L; 8.6% of the samples were > 10 mg/L. Filtered tap water, from a filtered faucet or a refrigerator dispenser, had only slightly lower levels. Both types of bottled water were significantly lower in nitrates with only three samples greater than 5 mg/L.
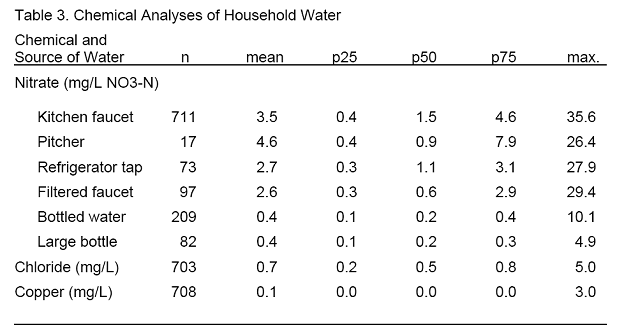
Just over half of the households had private wells (Table 4). The remaining 44% were served by water systems; about 40% of the respondents thought their water system was a large, municipal or water system. Few of the respondents had water bills or other information to verify that the water system was a Community Water System.

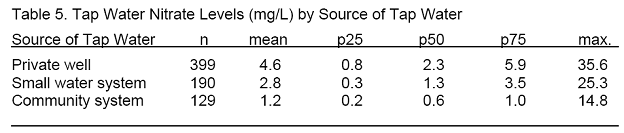
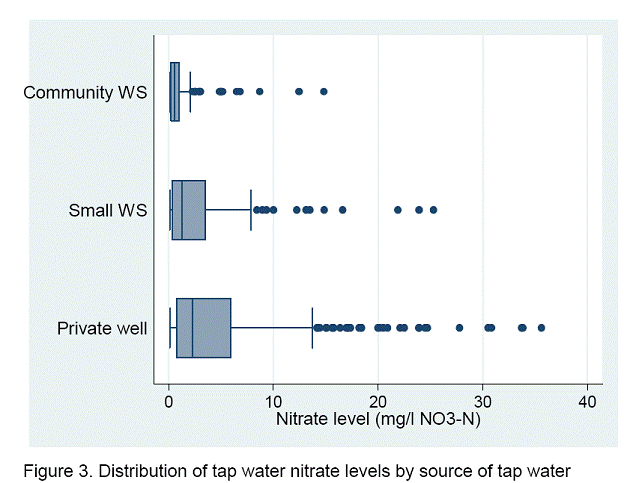
Tap water nitrate levels varied by the source of tap water: private wells had the highest levels while community water systems had the lowest (Table 5 and Figure 3). While over 10% of the private wells had nitrate levels over 10 mg/L, only nine of the samplefrom households served by small community systems (4.7%) and two of the samples from community water systems (1.6%) were over 10 mg/L.
A total of 71 split samples were analyzed. Eighty-nine percent (n=63) were within 1 mg/L, and an additional five were within 2 mg/L. Of these, one had a relative difference of 33 percent (3.5 vs. 2.3 mg/L); the rest had relative differences < 20 percent. The three split samples with larger absolute differences were: 9.8 vs. 12.8; 5.9 vs. 2.5; and 5.0 vs. 0.1.
Seventy-two sets of controls were run in the field using a blank (distilled water) and 5 mg/L and 10 mg/L standards. Ninety percent of the blanks had levels at or below 0.5 mg/L. There were two values ≥ 1 mg/L with a maximum of 1.3 mg/L. The average absolute difference of the 5 mg/L and 10 mg/L controls were -0.3 mg/L and -0.2 mg/L respectively, indicating good accuracy. Seventy percent of the 5 mg/L control samples and 78 percent of the 10 mg/L control samples were within 20 percent of the standard value (1 mg/L and 2 mg/L respectively), indicating lower precision for nitrate levels less than 10 mg/L. The 90th percentile for the 5 mg/L controls was 2.1 mg/L, with the top 3 samples over 3 mg/L. The 90th percentile for the 10 mg/L controls was 4.4 mg/L, with the top three ranging from 6.4 to 8.0. The potential impact of the precision of these measurements is addressed in the discussion section.
2. Chloride
The mean chloride level was 0.7 mg/L. Approximately 10 percent of the samples were greater than 1 mg/L. The selectivity coefficient for chloride relative to nitrate is approximately 0.005, so that for samples where both the nitrate and chloride levels were 1 mg/L, the presence of chloride would increase the reading by 0.5 percent or 0.005 mg/L. At 1 mg/L NO3-N, the presence of chloride of 5 mg/L (maximum observed) would be expected to increase the reading by 2.5 percent or 0.25 mg/L. As such, chloride in the samples did not have a significant effect on nitrate readings.
3. Copper
Only 1 percent of the samples had 1 mg/L or higher of copper. The maximum value was 3 mg/L.
4. Bacteriological Quality
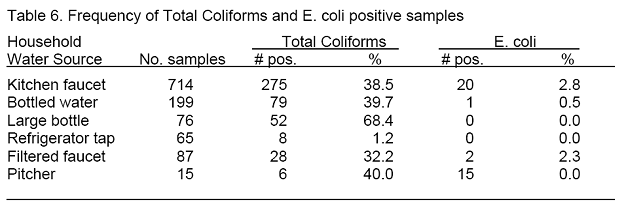
Total coliforms (TC) were found in a large proportion of household water samples, regardless of the source. E. coli (EC) were isolated from only 23 of the 1,156 samples taken, and 20 of these were from kitchen faucets (2.8%) (Table 6). About 40 percent of the tap water samples and the small bottled water samples were TC positive. More than two-thirds of the large 5-gallon water bottles were TC positive. Only 1.2 percent of the samples from the refrigerator dispensers were positive, significantly lower than the other sources of household water.
Fifty-one positive and negative controls were run for TC and EC. All negative controls were negative indicating good technique. One positive control was TC and EC negative. The incubator temperature was recorded at four times during the incubation. All readings were within 1º of 35ºC. This result may have been due to inactive growth media, or an inactive positive control.
E. Water source and consumption
During the interview, the mother or father was asked about all sources of water that were given to the child, either directly or used in the preparation of any drink or food. Tap water was at least occasionally given to 78.3 percent of the infants while bottled water was given to 43.8 percent. Just over half of the infants (54.0%) were typically given tap water but no bottled water; just under one-fifth (19.5%) were typically given only bottled water. Only 15 respondents said that they never give their child any water.
F. 24-hour diet
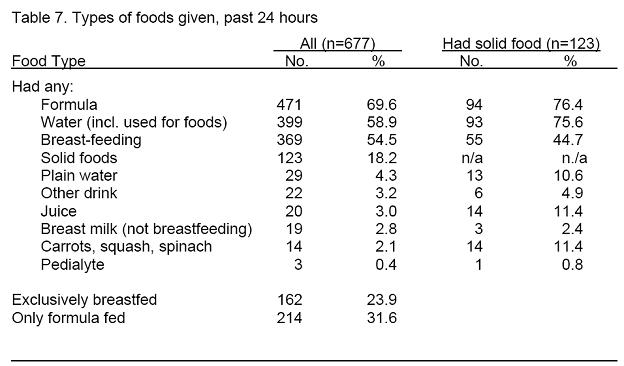
Every feeding the child received in the previous 24 hours was recorded, and the data were summarized for each of the 677 infants for whom information was provided. Just under a fourth of the infants (23.9%) were exclusively breast-fed during the previous 24 hours, and an additional 30.6 percent were breast-fed and given other foods and/or drinks (Table 7).
Overall, 69.5 percent were given formula and nearly one-third were given only formula during that day. Over 80 percent of the formula preparations included water as part of the mixture. Almost a fifth of the infants were given solid food. Of these, 76.4 percent were also given formula and 44.7 percent were also breast-fed. Other diet items included juices (3.1%), other drinks (3.4%), and Pedialyte (0.4%). Overall, only 14 infants consumed any carrots, spinach or squash, foods that may have appreciable levels of nitrate.
Just under 60 percent of the infants had some food or drink the previous day which included water. In just over half of these cases, this was tap water, predominantly unfiltered and directly from the tap. Over 80 percent of the kids who were fed formula had it mixed with water, and 95.5 percent of the infants given water, were given water as part of formula, although many of these infants also received water as part of other foods or drinks. Only 29 (4.2%) were given plain water. Overall, 31.5 percent were given water from the tap, while 26.0 percent were given bottled water. Only 4 infants consumed both tap and bottled water during that day.
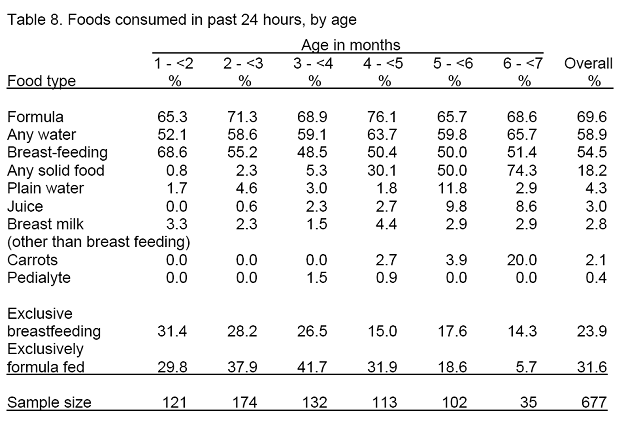
Feeding patterns changed somewhat between the younger and older age groups (Table 8). Most noticeably, breastfeeding, and exclusive breastfeeding in particular was far less prevalent among the older infants. Solid food consumption increased from less than a percent among the 1 to 2 month age group to almost three-quarters of the 6 to 7 month age group.
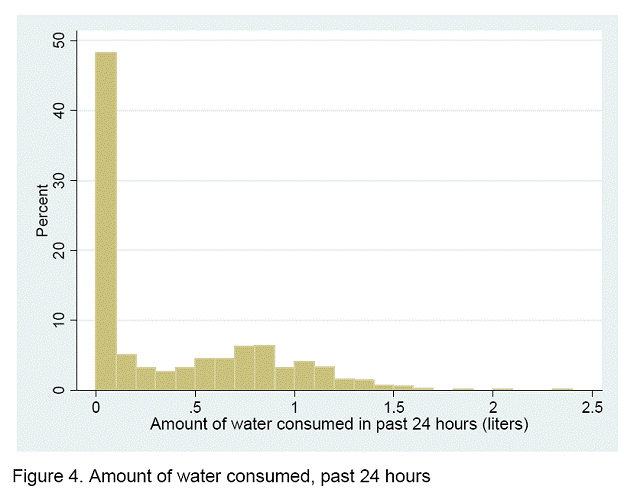
The total amount of water consumed ranged from 0 to 2.3 liters. Only 12.6 percent consumed more than 1 liter (Figure 4). Among those who consumed water, the mean amount consumed increased between 1 and 3 months, but remained relatively constant through 7 months of age.
G. Nitrate intake
Nitrate intake was estimated as the total daily amount of nitrate in water ingested divided by the infant’s weight. The amount of nitrate ingested was estimated as the amount of water consumed with each food item multiplied by the nitrate concentration measured in the household water source used in preparing that food item. Only 14 infants had ingested any foods that could have had appreciable levels of nitrate; this potential exposure was analyzed separately.
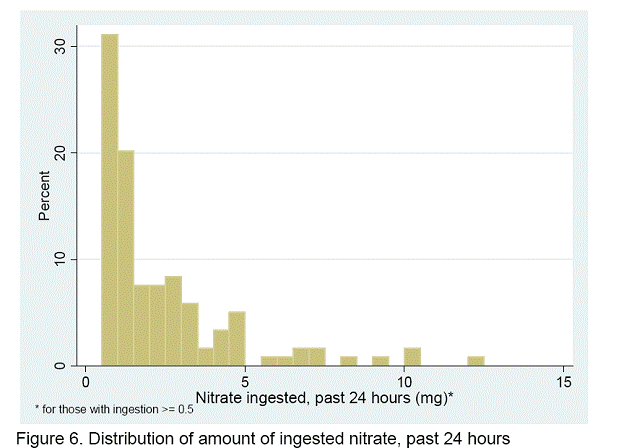
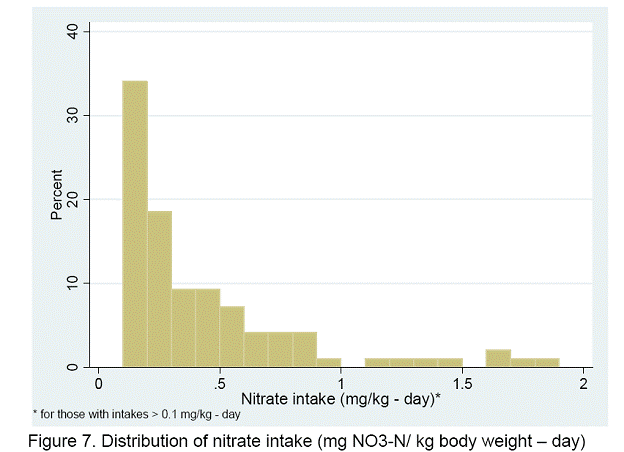
Over half of the infants ingested less than 0.1 mg NO3-N, while 12.0% ingested ≥ 1.0 mg during the preceding 24 hours (Table 10). Only three infants (0.3%) ingested more than 10 mg (Figure 6). Nitrate intake ranged from zero to 1.9 mg NO3-N/ kg body weight – day. Almost 10 percent of the infants (9.6%) had intakes ≥ 0.2 mg/kg day; 4.3 percent (n=28) had intakes ≥ 0.5 mg/kg day (Figure 7). Only 4 infants were above the RfD of 1.6 mg NO3-N/kg day. Infants whose intake was ≥ 0.5 mg NO3-N/kg day were considered to be in the high intake group.
As would be expected, high nitrate intake was related to both the amount of water consumed and the concentration of nitrate. Over 90 percent of the infants in the high intake group consumed 500 mL of water or more in the previous 24 hours. However, among those consuming 500 mL or more, the proportion of infants in the high intake group did not increase with increasing consumption. Only 3 of the 28 with high nitrate intakes consumed less than 500 mL, and all these infants were consuming water with > 10 mg/L nitrate.
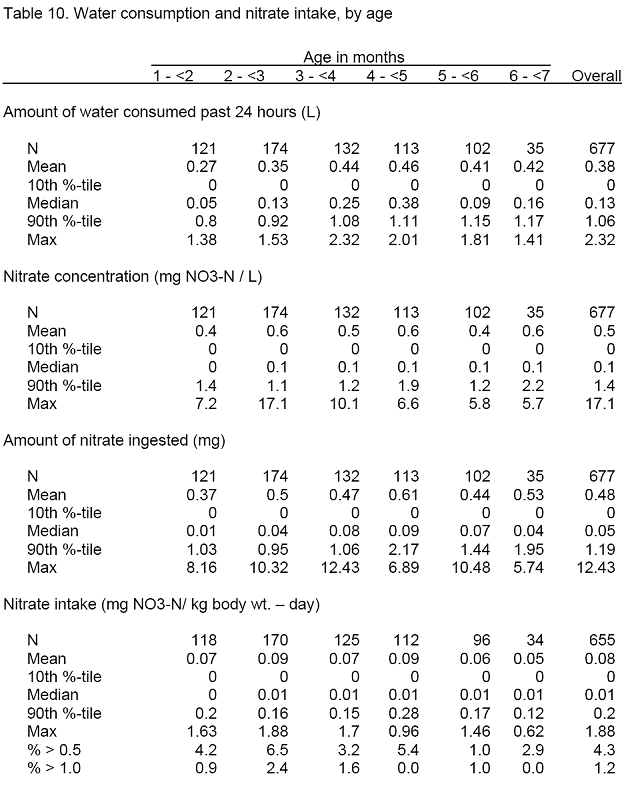
While 35.7 percent of those with high intakes were consuming water < 5 mg/L nitrate, 63.6 percent of those with water between 5 mg/L and 10 mg/L, and 57.1 percent of those with water > 10 mg/L had high nitrate intake. All but one of the 22 infants who consumed more than 100 mL of water at or above 10 mg/L were in the high intake group. About 45.4 percent of infants ≤ 2 months of age consumed 100 mL or more; the proportion ranged between 48.0 percent to 58.4 percent in the older age groups (Table 10).
H. Exposure to bacteriological water contamination
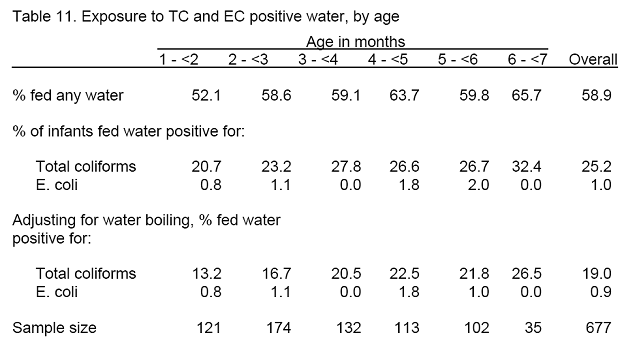
Of the 58.9 percent who were given water, 43.8 percent of the water samples from the household source given to the infant were positive for total coliforms (TC), as were 1.8 percent for E. coli (EC) (Table 11). Thus, a quarter of all infants consumed TC-positive water, and 1 percent consumed EC-positive water (n = 7).
About a third (31.8%) of the mothers who gave their infants water reported boiling the water before using it. Assuming that boiling effectively purifies the water, the proportion of infants consuming water positive for TC drops to 19.0 percent, but remains virtually the same for EC (0.9%, n=6). A larger proportion of women using tap water reported boiled it as compared to women using bottled water (43.3% vs. 18.7%); however, a larger proportion of the infants given bottled water consumed TC-positive water as compared to infants given tap water (52.7% vs. 36.6%).
I. Methemoglobin levels
Methemoglobin levels were obtained from 558 infants. In 21 cases, the co-oximeter calibration was not within the recommended standards. These observations were eliminated from further analyses.
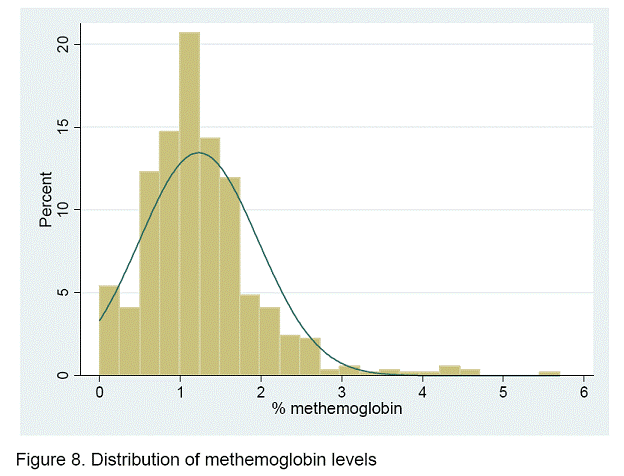
Of the remaining observations, 63.5 percent had MetHb levels 1 percent or greater, 11.7 percent had levels of 2 percent or greater, and 2.6 percent had 3 percent or more of their hemoglobin in the methemoglobin form (Table 12, Figure 8). Seven infants had levels above 4 percent. Less than 1 percent MetHb is considered normal, while some indications of oxygen deficiencies appear at 3 percent. None of the infants demonstrated any signs or symptoms indicating a frank case of methemoglobinemia.
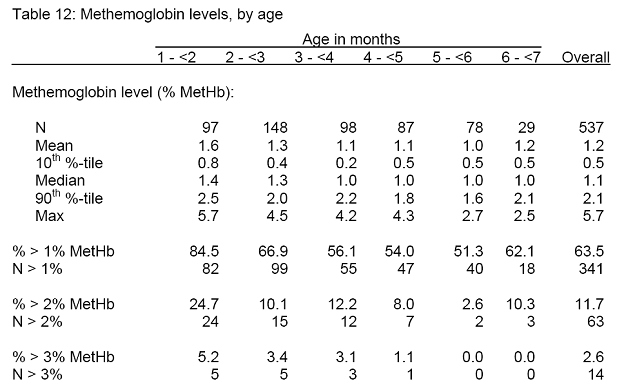
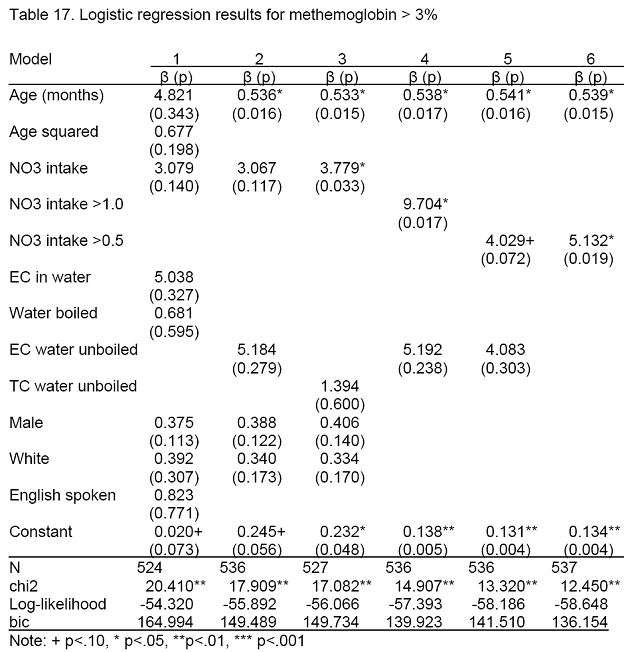
J. Risk factors for elevated methemoglobin levels
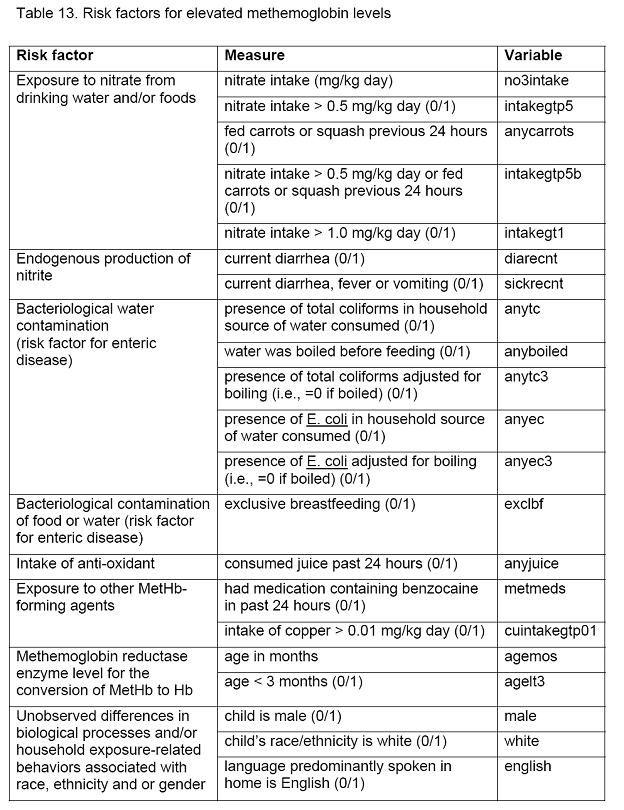
The risk factors for elevated metHb levels are presented in Table 13 below. As would be expected, many of the risk factors are related to the infant’s age and race/ethnicity (Table 13).
There was strong correlation between several of the risk factors. Several of the risk factors increased significantly with age, particularly the factors related to diet: consuming any carrots or juice, exposure to TC in water (adjusted for boiling), metHb forming medications. Exclusive breastfeed decreased significantly with infant’s age. There was no relationship between gender or race and age.
Risk factors associated with diet were themselves associated. Nitrate intake and exposure to TC were strongly associated; 60.7 percent of those with high nitrate intakes also had TC positive water, as compared to only 22.8 percent of those not exposed to high nitrates (p < 0.0001). Further, those in the high nitrate intake group were half as likely to have diarrhea (3.6 vs. 6.2%), more than twice as likely to have their water boiled (46.4 vs. 17.4%), and less likely to be male (39.3 vs. 52.2%). None of the infants fed carrots and none of the infants who were exclusively breast-fed were in the high nitrate intake group. Nitrate intake was somewhat lower among those currently sick, and none of those fed carrots were sick.
Male infants were less likely to come from English-speaking homes, but none of the other risk factors were significantly associated with gender. Those from English-speaking homes were more likely to have higher copper levels, less likely to have used teething gels, less likely have been given juice, more likely to be exclusively breast-fed, less likely to be exposed to TC-positive drinking water, less likely to have had their water boiled, and less likely to be currently sick.
Race/ethnicity was strongly associated with the language most commonly spoken at home: 99 percent of those self-described as "white" spoke English, as did 33 percent of those who described themselves as a race other than white. As such, the same associations of risk factors with language were present.
K. Relationship between risk factors and methemoglobin levels
1. Outcome
Three measures of metHb were used in these analyses; the measured level, a dichotomous variable indicating that the level was 2 percent or higher, and a variable indicating a metHb level of 3 percent or greater.
2. Bi-variate analyses
Only four of the risk factors were associated with metHb levels greater than 3 percent: high nitrate intake (OR =5 .9, p = 0.003), high nitrate including consumption of carrots (OR = 4.1, p = 0.026), consumption of EC-positive water (OR = 10.0, p = 0.014) and age less than 3 months (OR = 3.1, p = 0.05). Gender had a relationship that was of borderline significance (OR = 0.36, p = 0.07). Drinking TC-positive water had a very weak association (OR = 2.0, p = 0.19).
Age less than 3 months was the only statistically significant risk factor for having metHb > 2 percent (OR=2.1, p=0.006). Consuming TC-positive water (OR = 1.63, p = 0.08) and water boiling (OR = 1.73, p = 0.07) were the only risk factors even marginally associated with metHb > 2 percent, and the estimated effect of water boiling was the opposite of what was expected. High nitrate intake had a weak association (OR = 1.86, p = 0.22), as did gender (OR = 0.70, p = 0.19) and speaking English in the home (OR = 0.68, p = 0.14).
3. Linear regression
Linear regression was used to model the effects of the risk factors on the level of metHb. Only two of the risk factors in the fully specified model (model 1) were statistically significant: age and race/ethnicity. A quadratic age term was added and was highly significant (model 2). Variables were removed from the model based on their statistical significance. Interactions of age with EC-positive water and nitrate intake were not significant (results not shown).
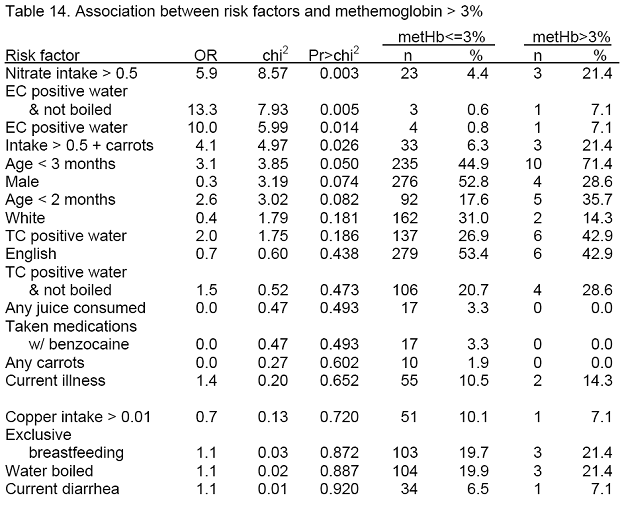
The statistically significant parameter estimates (age, age squared and race/ethnicity) were stable across model specifications. Infants who were identified as being "white" had a small (-0.15 to -0.17) but statistically significant lower metHb level than other infants. Consuming TC-positive water, accounting for boiling or not, was not associated with the level of metHb in any of the models, however there was some evidence of an association with consuming EC-positive water (p = 0.15). There was also some indication of an association between nitrate exposure > 1 mg/kg – day and metHb, but it was not statistically significant (p = 0.15, model 5). The precision of the estimates was likely affected by the small number of infants who were exposed to EC contaminated water (n = 6) and nitrate intake greater than 1.0 (n = 8).
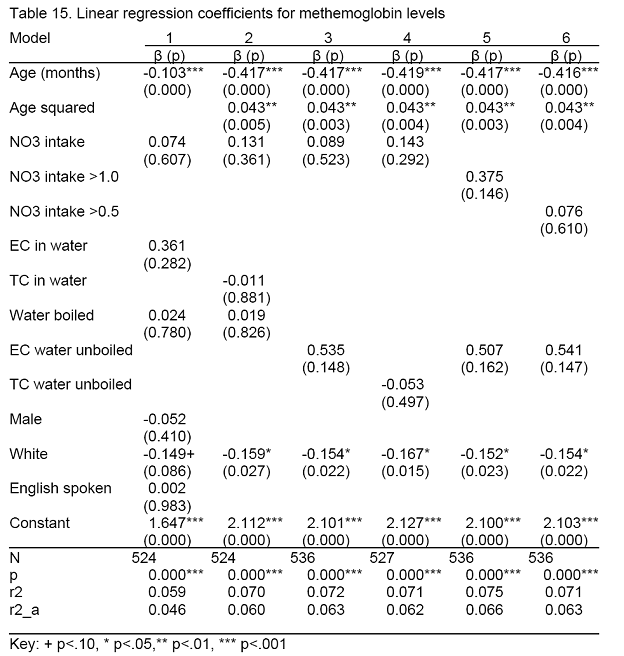
4. Logistic regression
Logistic regression was used to model the probability that metHb levels were greater than a set level. Two levels were used: 2 percent metHb and 3 percent metHb. Some researchers have indicated 2 percent metHb to be elevated. Other references state that 3 percent metHb is the lowest level of clinical significance.
a) MetHb > 2 percent
Having consumed juice and having had medications with benzocaine were dropped from the analysis because none of the kids who had either of these risk factors had metHb > 2 percent. While this might be expected if the anti-oxidants in juice were acting in a protective manner, taking methemoglobin-forming medications would be expected to increase the probability of having metHb > 2 percent.
Age was highly significant, however age-squared was not (model 2). None of the other risk factors were statistically significant in the full model, or in reduced models. TC-positive water was marginally significant when all other risk factors except age and nitrate were in the model (model 4, OR = 1.7, p = 0.075). Nitrate concentration was not statistically significant. When nitrate was removed from the model (model 6), TC-positive water became statistically significant (OR = 1.77, p = 0.047). Even when TC-positive water was removed from the model (model 5), nitrate was not statistically significant.
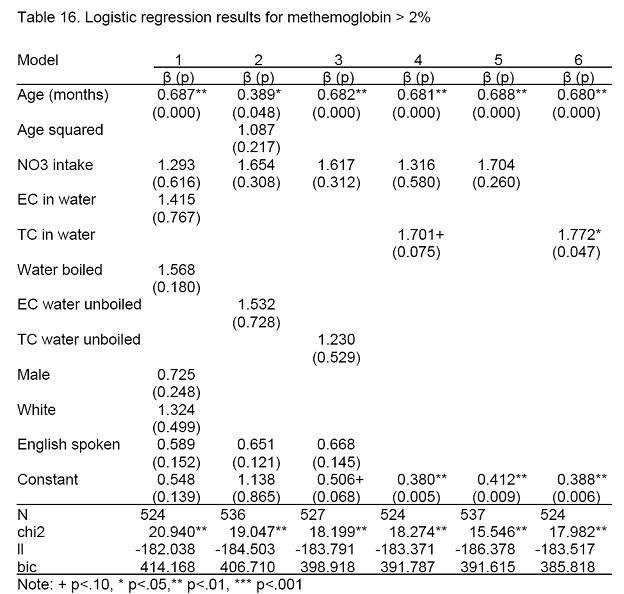
b) MetHb > 3 percent
In the full model, none of the risk factors were statistically significant (model 1). Age was highly significant when age-squared was dropped (model 2). Nitrate exposure was significantly associated with the risk of methemoglobin > 3 percent, especially when TC-positive drinking water was included rather than EC-positive water (models 3-5). Neither measure of bacteriological contamination was statistically significant in any of the models. There were no significant interactions between age and nitrate exposure when coded either as categorical, dichotomous or continuous variables. In each case, the interaction term was small and not significant (p > 0.7). The most parsimonious model (model 6) included only two significant risk factors: age in months (OR = 0.54, p = 0.015) and nitrate intake > 0.5 mg/kg – day (OR = 5.13, p = 0.02).
L. Perceptions and attitudes about water quality
Parents were asked whether they felt their tap water was safe for their baby to drink, for them to drink, and whether anyone in their family had gotten sick from drinking their tap water. Overall, just under half (48.4%) felt that their tap water was not safe for their baby to drink, with an additional 16.0 percent saying it was safe but only because it had been boiled or because they had a water filter. A somewhat lower proportion (33.9%) felt that their tap water was not safe for them to drink. Five percent thought that someone in their home had been ill because of the tap water. Just under half of those with private wells (48.2%) reported having tested it. About half had tested the well within the past 2 years.
Perceptions of water quality varied somewhat by the type of water system serving the household (Table 18). While the differences were not statistically significant, respondents with private wells were more likely to think that their tap water was safe to drink, and was safe for their baby to drink. Perceptions were associated with feeding patterns. Those who thought the water was safe for their baby to drink were more likely to have given their baby tap water as compared to those who thought the water was not safe (45.7% vs. 6.3%). However, almost all of those in the second group had boiled the tap water given to their infant (82.4%).

V. DISCUSSION
The controversy regarding the importance of nitrate contaminated drinking water as a cause of methemoglobinemia has been discussed for decades. The primary argument against a significant role for nitrate has been that in agricultural areas fecal contamination of wells frequently co-occurs with nitrate contamination, and that this leads to enteric infections, which leads to the endogenous production of nitrite. In short, nitrate contamination is correlated with enteric infections, the actual cause of methemoglobinemia.
Several factors make it difficult to address this issue directly. First, frank cases of methemoglobinemia are rare in the United States, and the condition is transient making it nearly impossible to conduct a study with sufficient sample size to assess the relative importance of the various risk factors on clinically observed cases of methemoglobinemia. While using the percent of blood in the metHb form allows feasible subject recruitment, metHb can be readily reduced back to hemoglobin, with a half-life of 4 to 6 hours. The transient nature of increased methemoglobin levels after exposure adds unexplained variability when analyzing the association of methemoglobin levels and potential risk factors.
Further, the etiology of the disease is complex and multi-factorial. Because nitrate is present in drinking water, any infant food prepared with water, as well as specific vegetables, many sources can contribute to nitrate exposure. Endogenous production of nitrite is nearly impossible to observe directly, and indirect measures, such as reported symptoms of enteric distress or fecal contamination of drinking water, are likely poor surrogates.
Many biologic factors may mediate the relationship between nitrate exposure and methemoglobin formation, and these mediating factors vary by age, and between individuals of the same age. In particular, the rate of re-conversion of metHb back to Hb is highly variable, adding unexplained variability in the assessment of the effect of exogenous or endogenous exposure on methemoglobin levels. Age and individual dependent rates of conversion of nitrate to nitrite in the stomach add yet more variability in the relationship between ingested nitrate exposure and nitrite levels in the blood. Further, the percent of the blood in the "infant Hb" state affects the blood nitrite – metHb relationship.
Finally, the major exposure factors, nitrate and fecal water contamination, are generally correlated. Further, exposure to these factors, as well as the mediating factors, varies by age. Thus, it is very challenging to disentangle the effects of these intertwined risk factors and measure their actual and independent effects.
Given the rarity of methemoglobinemia cases and the multiple potential risk factors, it seemed plausible that a frank case would arise when several of the risk factors occurred concurrently, so that exposure to methemoglobin-forming agents was maximized and the infant’s ability to reduce metHb back to hemoglobin was low. Studying this by retrospectively gathering data after a case of methemoglobinemia seemed impractical given the low incidence of the disease. Assuming that frank cases arose from a combination of risk factors, with each acting to increase the level of methemoglobin, we designed the study to observe the effects of relevant risk factors on changes in metHb levels.
One of the strengths of this study was the careful assessment of exposure. Even though the study was cross-sectional, the rapid impact of methemoglobin-forming agents on methemoglobin levels, and the consistency of water nitrate levels over the course of a day, made it possible to accurately estimate the level of nitrate in the water consumed by the infant during the relevant exposure period. Furthermore, this study collected detailed consumption information from the mother for the preceding 24 hours, including the amount of food prepared, the amount of water used in the preparation, and the amount actually consumed by the infant, using commonly used food and drink serving containers. Estimates of nitrate intake were based on the overall amount of water consumed, the household source(s) of this water, and actual nitrate measurements for each specific household water source. Exposure to fecally contaminated water was estimated similarly. While standard indicator organisms were used to measure water contamination, total coliforms are not specific to fecal contamination, while E. coli are not a sensitive indicator for all types of fecal contaminants.
MetHb levels were measured using an accepted method which has been shown to have good accuracy and better precision than the standard lab test based on titration. Information on other risk factors was collected directly from the caregiver in face-to-face interviews conducted in Spanish or English by experienced fieldworkers, including nurses, from the local area.
The major limitation of this study was the response rate. Depending on the proportion of non-responders who were assumed to have been eligible, the response rate was between 42 and 65 percent, potentially limiting the generalizability of the results. While a low response rate may indicate a bias due to differential participation, these effects may be more important for descriptive measures of associated with infant feeding behaviors, than with the modeling of the exposure-disease relationship, which is primarily a biological process.
The analytical power of the study was limited by the relatively few infants with elevated methemoglobin levels; about 10 percent of the infants were above "normal", while only 3 percent had physiologically significant levels (> 3% metHb). In addition, only 4 percent of the sample had nitrate intakes at 0.5 mg/kg day or higher, roughly one-third of the RfD; Only three infants had estimated nitrate intakes greater than the RfD. This limits the power to observe associations between elevated nitrate exposure and elevated methemoglobin levels, should it exist. There may have been some exposure misclassification due to the level of precision of the nitrate measurements. There were 44 infants who had average nitrate levels between 0.5 and 10 mg/L, and who had estimated nitrate intakes greater than 0 and less than 1.0 mg/kg – day. In this group, all infants who had water with an average nitrate level > 4.7 mg/L were categorized as having "elevated" intakes (i.e., > 0.5 mg/kg – day), while all those whose average nitrate levels < 2.8 mg/L had intakes < 0.5 mg/kg –day. Given the distribution of the measured nitrate values for the 5 mg/L control samples, variation in the measured nitrate levels from the actual values would likely have changed the exposure classification of fewer than 5 infants. However, the effect of this potential misclassification is difficult to predict.
The physiological processes leading from exposure to elevated metHb are complex and non-linear, so it is not clear what functional relationship should be used to model this process. As such three sets of models were constructed. Linear regression was used to model the effect of the risk factors on variation in methemoglobin levels. In addition to the variability in methemoglobin levels, and the non-linear relationship between risk factors and metHb level, modeling changes in metHb levels mostly within the normal range may not be clinically relevant. Two logistic regression models were also developed, modeling the relationship between the risk factors and having methemoglobin levels >2 and > 3 percent, respectively. While there is a loss of information when using dichotomous outcomes, this does reduce the underlying "noise" and can identify those factors that are most strongly associated with having a high level, and thus, most likely to be an important risk for a clinical case of methemoglobinemia.
Age demonstrated a strong inverse association with high MetHb levels in each type of model. In the linear regression models, the effect was non-linear as a quadratic term was highly significant, indicating a decreasing marginal effect as age increases. Age may be capturing the effects of several age-varying factors: the infant’s intrinsic ability to re-convert methemoglobin back to hemoglobin, unmeasured dietary factors, or the risk of endogenous production of nitrites.
Nitrate and bacterially contaminated water were correlated, and this was reflected in their parameter estimates in the various models. In the linear model, there was weak evidence of an association of both nitrate intake and EC-contaminated water (adjusted for boiling) with metHb. TC-contaminated water was marginally associated with metHb > 2 percent (OR = 1.7, p = 0.08), and statistically significant when nitrate intake was removed (OR = 1.8, p = 0.05). Removing nitrate intake did not affect the parameter estimate, indicating that there was little confounding present.
Nitrate intake, measured as a continuous variable or as an indicator, was significantly associated with having metHb > 3 percent, even with TC or EC contaminated water in the specification. Neither TC or EC contaminated water was associated with metHb > 3 percent. Removing these factors did increase both the parameter estimate and the significance of the nitrate intake variable. The estimated effect of nitrate was substantial; having an intake > 0.5 was associated with a 4- to 5-fold higher risk of having elevated metHb. Using the continuous measures, one would have a 3- to 4-fold risk of having elevated metHb for every unit increase in intake.
One of the primary factors missing from these models is a measure of each infant’s ability to reduce metHb back to Hb. If age was acting as a measure of the activity of NADH methemoglobin reductase, the effect of nitrate exposure would be expected to larger among infants with lower enzyme activity, that is, among younger infants. Interactions between age and nitrate exposure (both categorical and continuous) were not statistically significant in any of the three model specifications.
The lack of association between bacteriological water quality or illness and methemoglobin levels should not be interpreted as evidence that endogenous production of nitrite is not a cause of methemoglobinemia. Methemoglobinemia has been documented in infants that suffer from diarrhea and who not had any exposure to ingested nitrate. In many cases, these were infants with serious diarrhea. The measure of having diarrhea, or a fever, vomiting or diarrhea, may not be capturing those cases serious enough to trigger significant endogenous production of nitrite. Bacterial water quality may also have been a poor measure of this risk. Total coliforms are commonly found in well water as well as in the household environment, and may not necessarily indicate the presence of pathogens. E.coli are a much more specific indicator of pathogenic contamination, but they were rarely present in the water samples tested. In the bi-variate analyses, the presence of E. coli in the household source of water given to the infant was very significantly associated with high metHb (OR = 13.3, p = 0.005), but this was based on only seven EC-positive samples. The relationship was not statistically significant in the multi-variable models.
Illness, the use of benzocaine-containing medications and exposure to ascorbic acid (as measured by intake of juice) were not associated with metHb levels. Being white as associated with a small (-0.2) but statistically significant reduction in metHb levels in the linear model. In some models, male and white infants had a lower risk of having high met levels; however, these effects were not statistically significant with p-values ranging between 0.10 and 0.20. None of the other risk factors were associated with methemoglobin levels.
While the risk of a clinical case of methemoglobinemia posed by the consumption of water at the current MCL cannot be estimated based on the results of this study, the results do provide evidence that ingesting nitrate above 0.5 mg/kg-day significantly increases the risk of having methemoglobin levels considered to be physiologically significant (i.e., > 3%) with an OR of three to five. Further, such levels of exposure are not uncommon; virtually all infants (16 of 17) fed water with 5 mg/L NO3-N or higher fell into this high exposure category. Even 12 percent of infants fed water with low levels of nitrate (0.5 to 5.0 mg/L) nitrate had exposures greater than 0.5 mg/kg day; the high intake levels were due to high consumption levels (> 500 mL/day).
Serious clinical symptoms of methemoglobinemia do not appear until the level of metHb reaches about 10 percent. Such cases are rare, with only a few documented cases each year in the United States. Reaching a high methemoglobin level likely results from several factors coming into play at the same time: high exposure to nitrate in foods and/or water, high endogenous production of nitrite, and a low level
2003 Progress Report
2004 Progress Report
2005
2006 Progress Report
Conclusions:
The results of this study provide evidence that exposure to nitrate from drinking water significantly and substantially increases the risk of an infant having physiologically elevated levels of methemoglobin. Furthermore, this risk is associated with intake levels above 0.5 mg NO3-N/kg day, approximately one-third of the RfD value. Overall, about 4 percent of the infants had this level of exposure, and it occurred uniformly from 1 to 5 months of age. In this sample, virtually all infants (94.1%) who were given water containing nitrate above 5 mg/L NO3-N had exposures above 0.5 mg/kg day.
Exposure to TC or EC positive drinking water was associated with the level of methemoglobin, and with the probability of having > 2 percent metHb. Current diarrhea or combined illness (i.e., diarrhea, fever or vomiting) were not associated with methemoglobin levels in any of the models.
The multi-factorial nature of methemoglobinemia makes it very difficult to assess the risks associated with any one risk factor. These results indicate a relationship between exposure to relatively low levels of nitrate and elevated metHb levels. Elevated metHb levels likely increase the risk of clinically significant methemoglobinemia. Thus, consumption of drinking water containing nitrates may be a contributing factor for the development of methemoglobinemia.
References:
1. G. Stern, DOH Division of Drinking Water, personal communication, 2000.
2. Ryker SJ, Frans LM. Summary of Nitrate Concentrations in Ground Water of Adams, Franklin, and Grant Counties, Washington, Fall 1998—A Baseline for Future Trend Analysis, U.S. Geological Survey Water-Resources Investigations Report 99-4288, U.S. Department of the Interior, Tacoma, Washington, 2000, pp. 1-39.
3. Wright RO, Lewander WJ, Woolf AD. Methemoglobinemia: Etiology, Pharmacology, and Clinical Management, Ann Emerg Med, 1999;34:646-656.
4. Jaffe ER, Hultquist DE. Cytochrome b5 reductase deficiency and enzymopenic hereditary methemoglobinemia, in Scriver CR, Beaudet AL, Sly WS, Valle, D (eds.), The Metabolic and Molecular Bases of Inherited Disease (ed 8), New York, NY, McGraw Hill, 2001, p 4555.
5. Hegesh E, Shiloah J. Blood nitrates and infantile methemoglobinemia, Clinica Acta, 1982;125:107-115.
6. Mansouri, A, and Lurie AA. Concise review: Methemoglobinemia. Am J Hematology, 1993;42:7-12.
7. Bosch, HM, Rosenfield, AB, Huston R, Shipman, HR, and Woodward, FL. Methemoglobinemia and Minnesota well supplies. J. AWWA. 1950:42:161-170.
8. Walton G. Survey of literature relating to infant methemoglobinemia due to nitrate-contaminated water, Am J Public Health, 1951;41:986-995.
9. Office of Drinking Water, Environmental Protection Agency, The drinking water criteria document on nitrate/nitrite, December 21, 1990, pp. III-IX-14.
10. Hanukoglu A, Danon PN. Endogenous methemoglobinemia associated with diarrheal disease in infancy, J Pediatr Gastro Nutr, 1996;23:1-7.
11. Felsot AS, Re-examining the link between nitrates and “Blue-Baby” syndrome: A necessary first step for managing ground water quality to protect public health, Agri Environ News, 1998;150:1-14.
12. Avery AA. Infantile methemoglobinemia: Reexamining the role of drinking water nitrates, Environ Health Pers, 1999;107:1-8.
13. Gupta SK, Fitzgerald JF, Chong SKF, Croffie JM, Garcia JGN. Expression of inducible nitric oxide synthase (iNOS) mRNA in inflamed esophageal and colonic mucosa in a pediatric population, Am J Gastro, 1998;93:795-798.
14. Levine JJ, Pettei MJ, Valderrama E, Gold DM, Kessler BH, Tractman H. Nitric oxide and inflammatory bowel disease: Evidence for local intestinal production in children with active colonic disease, J Pediatr Gastro Nutri, 1998;26:34-38.
15. Langlois CJ, Calabrese EJ. The interactive effect of chlorine, copper and nitrite on methemoglobin formation in red blood cells of Dorset sheep. Human Exp. Toxicol. 1992;11:223-228.
16. Knobeloch L, Salna B, Hogan A, Postel J, Anderson H. Blue-babies and nitrate contaminated well-water. Environ Health Perspect. 2000;108:675-678.
17. Centers for Disease Control and Prevention. A SAS Program for the CDC Growth Charts. http://www.cdc.gov/nccdphp/dnpa/growthcharts/resources/sas.htm. (Accessed May 15, 2008).
Journal Articles:
No journal articles submitted with this report: View all 6 publications for this projectSupplemental Keywords:
RFA, Health, Scientific Discipline, Water, Health Risk Assessment, Risk Assessments, Environmental Chemistry, Analytical Chemistry, Children's Health, Biochemistry, Drinking Water, health effects, human health, dietary exposure, other - exposure, methemoglobin formation, nitrate contamination, water quality, human exposure, chemical contaminants, dietary ingestion exposures, chemical exposure, children, drinking water contaminants, exposure, groundwater, methemoglobin, children's vulnerablity, human health effects, human health risk, assessment of exposure, exposure and effects, exposure assessmentProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Contact Us to ask a question, provide feedback, or report a problem.
Last updated April 28, 2023
