Grantee Research Project Results
Final Report: A Long-Life, Low-Cost Sorbent for the Conversion of HCl to Chlorine
EPA Contract Number: 68D98158Title: A Long-Life, Low-Cost Sorbent for the Conversion of HCl to Chlorine
Investigators: Copeland, Robert , Gebhard, Steven , Cesario, M. , Dubovik, Margarita , Gershanovich, Yevgenia
Small Business: TDA Research Inc.
EPA Contact: Richards, April
Phase: II
Project Period: September 1, 1998 through September 1, 2000
Project Amount: $225,000
RFA: Small Business Innovation Research (SBIR) - Phase II (1998) Recipients Lists
Research Category: SBIR - Pollution Prevention , Pollution Prevention/Sustainable Development , Small Business Innovation Research (SBIR)
Summary/Accomplishments (Outputs/Outcomes):
Purpose of Research:
The primary purpose of this research was to develop a long life sorbent to remove hydrogen chlorine (HCl) from wastes streams and convert it to elemental chlorine (Cl2). The initial work was based on a copper oxy-chloride sorbent with a temperature swing process. The later word identified and developed a new sorbent and process that operated isothermally. The goal was to remove HCl from the waste stream of a plant and convert the HCl into Cl2 at a cost significantly less than purchasing chlorine from external sources.
Project Description:
In order to minimize the cost of the final product, TDA Research, Inc. (TDA) developed two types of regenerable HCl sorbents with high HCl absorption capacity and the ability to be regenerated for multiple cycles. In this Phase II research we investigated methods of forming the sorbents and measuring the physical and chemical properties of those sorbent. We then produced sorbents using techniques that would be used in commercial practice (i.e., extrusion). We measured their crush strength, pore volume, and capacity. We then tested the best sorbent for multiple cycles measuring its breakthrough characteristics and endurance.
Research Findings:
During Phase II we produced numerous sorbents at TDA using low cost materials such as copper oxide, inert materials (e.g., alumina), and activity aids. We identified several with the potential to meet the requirements for conversion of HCl to Cl2. We then selected two for multiple cycle testing. We tested those formulations (identified as Rita-3 and Rita-4) for multiple cycles and found that they had a long life and remained active for all cycles. We discovered that the sorbent did not spall, even under the most severe regeneration conditions.
However, the thermodynamics of the process with the copper-based sorbent were not favorable. Chlorine at high concentrations was observed at the start of the oxidation. Unfortunately, most of the Cl2 was released at low concentrations (e.g., 3% Cl2, balance O2) and the regeneration was very slow. Due to the large reactors and the cost of separating the low concentration of chlorine, our industrial partners (Air Products and Chemicals, Inc. (APCI) and Millennium Inorganic Chemicals (MIC) considered the process to be uneconomical.
Given that the copper thermodynamics were unfavorable, we developed a new sorbent and a modified Deacon process with favorable thermodynamics. It operates isothermally and produces Cl2 concentrations that are theoretically over 50%, with a measured average of over 30%. We measured high HCl loadings (>20%) and a capacity that was retained after multiple cycles at high Space Velocities (SV's). We estimated the cost of chlorine recovery at less than $84/ton Cl2 delivered, less than half the cost of fresh chlorine produced by other means.
Potential Applications:
This new sorbent production method and the new process can reduce the chlorine for industrial chemical plants significantly. Additional testing of the sorbent and process is recommended. Applications include chlorinated plastics plants and the production of inorganic chemicals (e.g., TiO2).
SBIR Rights Notice (March 1994)
These SBIR data are furnished with SBIR rights under U.S. Environmental Protection Agency Contract No. 68-D-98-158. For a period of 4 years after acceptance of all items to be delivered under this contract, the Government agrees to use these data for Government purposes only, and they shall not be disclosed outside the Government (including disclosure for procurement purposes) during such period without permission of the Grantee, except that subject to the foregoing use and disclosure prohibitions, such data may be disclosed for use by support contractors. After the aforesaid four-year period the Government has a royalty-free license to use, and to authorize others to use on its behalf, these data for Government purposes, but is relieved of all disclosure prohibitions and assumes no liability for unauthorized use of these data by third parties. This Notice shall be affixed to any reproductions of these data, in whole or in part.
Acknowledgements
TDA gratefully acknowledges the guidance and funding provided by both Air Products and Chemicals, Inc. (APCI) and Millennium Inorganic Chemicals (MIC). Merrill Brenner, Robert Coraor, Chuck Coe, John Tao, and Robert Weimer of APCI and Mike Robinson of MIC were invaluable in the performance of this work.
1. Executive Summary
1.1. Abstract
Air Products and Chemicals, Inc. (APCI) and Millennium Chemicals (MIC), as part of a coalition named CIETEP (the Chemical Industry Environmental Technology Partnership), are developing a copper-based process to recover HCl from chlorination processes. The goal of this research was to develop a long life sorbent to remove hydrogen chloride (HCl) from wastes streams and convert it to elemental chlorine (Cl2) at a cost significantly less than the market price of chlorine from external sources. In order to minimize the cost of the final product, TDA Research, Inc. (TDA) developed two types of regenerable HCl sorbents: one copper-based and one manganese based. TDA tested those formulations for multiple cycles and found that they had long life and remained active for many cycles.
While the copper-based sorbent performed far better than the HCl sorbents developed by previous groups, the thermodynamics of the copper-based process were not favorable. During regeneration, the Cu-based process provided some high concentrations of Cl2, but most of the Cl2 was released at low concentrations. The regeneration was very slow. Due to the large reactors and the cost of separating the low concentration of chlorine, our industrial partners, APCI and MIC, considered the process to be uneconomical.
TDA then identified a new process, herein identified as "Reductive Chlorination," and developed a manganese based sorbent for that process. An analysis of this new process showed that it had very favorable thermodynamics. In testing the new sorbent under conditions representative of the Reductive Chlorination process, we measured high HCl loadings after multiple cycles at high Space Velocities (SVs) an order of magnitude improvement in the chlorine concentration in the regeneration stream. We estimated the cost of chlorine recovery to be less than $84/ton Cl2 delivered, less than half the cost of fresh chlorine purchased from external sources (~$200/ton). We are continuing to evaluate different operating conditions for the Reductive Chlorination process under funding provided by APCI and MIC.
1.2. Introduction
Chlorine is a raw material used in the large-scale production of a range of valuable chlorinated solvents, polymers and fine chemicals. Unfortunately, one of the byproducts of the production of chlorinated hydrocarbons is HCl, a very reactive, toxic material that requires special precautions for transport and is expensive to neutralize. The U.S. demand for a range of chlorinated hydrocarbons is large (over 15 million tons annually) and, as a consequence, the production of "waste" HCl from the manufacturing of chlorinated hydrocarbons and inorganic chemicals (e.g., TiO2) is also large.
The Deacon reaction, Equation 1, can oxidize the waste HCl to water and chlorine.
Equation 1. Deacon reaction.
4HCl(g) + O2(g) = 2H2O(g) + 2Cl2(g)
@400oC, DH = -28.039 kcal ; DG = -6.252 kcal; K = 1.071(10)2; Log(K) = 2.03
This reaction has been known for decades but, unfortunately, is equilibrium limited. Since waste streams also have relatively low concentrations of HCl, the Deacon reaction inherently produces a low concentration of chlorine mixed with HCl. The Deacon process is not economical.
To overcome the limitations of the Deacon reaction, Minet et al. (1991) identified a copper-based sorbent to convert dilute HCl to chlorine. The process uses pure oxygen (O2) instead of air to oxidize HCl back to chlorine (Cl2) and water (H2O). The use of pure oxygen improves the Deacon process by shifting the equilibrium to produce more Cl2 (versus air), reduces the size of the equipment and reduces the energy required to heat and cool the gas streams. Equation 2 presents the reactions in this modified Deacon reaction process. Note that the overall net reaction is the Deacon reaction, and has the same net heat of reaction (DH = -28.039 kcal) and change in free energy (DG = -6.252 kcal). Therefore, the available energy is divided between the two steps. A temperature swing is incorporated into the process to improve the equilibrium conversion (i.e., the added thermal energy improves the free energy change by conducting the absorption at low temperature and the regeneration at high temperature).
Equation 2. Modified Deacon reaction with copper oxy-chloride sorbent.
Absorption: 4HCl +2Cu2oCl2 < 2H2O + 4CuCl2
Regeneration: 4CuCl2 + O2 < 2Cu2oCl2 + 2Cl2
Net: 4HCl + O2(g) < 2H2O + 2Cl2
APCI led a consortium to develop this modified Deacon process. The consortium had been operating a Product Development Unit (PDU) at their Carburos Metallicos plant in Barcelona, Spain to evaluate sorbent concepts for the recovery and conversion of HCl to Cl2. Their test system uses a sorbent made of CuO impregnated upon a zeolite or alumina support. Both fixed bed and fluidized bed operation is possible with the PDU. Although the impregnated copper sorbent is very effective at recovering all of the HCl and converting it to Cl2, a process analysis conducted by an independent engineering contractor for the consortium found that, as initially designed, the process economics are not favorable, primarily due to the limitations of the sorbent. The consortium's Cu/zeolite/alumina sorbent has a relatively low copper content (about 10% by wt), and not all of the copper is accessible. As a result, the chloride loadings on the sorbent are low, about 1-weight % in practice (Mortensen et al. 1995), and the cyclic capacity (the difference in HCl content between the fully loaded and regenerated sorbents) was even lower (about 0.5%). In addition, large temperature swings (~200?C) are required to regenerate the sorbent and produce chlorine.
A further problem with the initial fluidized bed system is that the violent motion of the sorbent particles in the fluidized bed causes mechanical attrition of the sorbent. APCI and the consortium have since focused on fixed bed systems to minimize the attrition losses of their relatively expensive sorbent. They have stopped pilot development, and are looking for 1) better sorbents to concentrate the HCl, 2) improved Deacon catalysts and 3) process modifications that will improve the process economics. The most crucial need is for a durable, selective, high loading sorbent.
While high Cl2 concentrations were measured by Minet (1991) and at the PDU at the Carburos Metallicos plant in Barcelona, Spain, those measurements were not continuous. Further, the researchers could not determine the thermodynamic properties of the copper oxy-chloride compound and, therefore, the thermodynamics of the modified Deacon process shown in Equation 2. At the start of the EPA Phase II contract, the heat of reaction and the change in free energy for each step in Equation 2 were unknown. As part of the Phase II work, TDA developed thermodynamic data on the copper oxychloride based on the measured experimental data.
1.2.1. EPA SBIR Phase II Base Contract
During the Phase I Small Business Innovation Research (SBIR) contract TDA developed a copper-based sorbent for the modified Deacon process. The goal for the Phase II contract was to develop improved sorbents and to measure their performance for multiple cycles. The results of that research and development are documented herein.
1.2.2. EPA SBIR Phase II Option Contract
TDA received additional funding for research beyond the EPA SBIR Phase II contract. The EPA provided an additional $70,000 and the Chemical, Industrial and Environmental Technologies Projects (CIETP) provided $110,000. The research goal was to improve the test apparatus and evaluate alternative operating conditions for the modified Deacon process. Some of the funds were used to purchase on-line analysis instruments to improve the data generated during the contract.
As part of the EPA funded Phase II option, TDA also identified a new approach for the modified Deacon process, which we call "Reductive Chlorination." TDA developed manganese based sorbents for this new approach, conducted tests and performed an economic analysis. That research and development, performed under the EPA portion of the Phase II option, is documented here.
This report documents the work done under EPA funding (both the base contract and the option). As of this writing, TDA is continuing to evaluate process options for the Reductive Chlorination under the CIETP funding. That work will be reported separately.
1.3. Copper-Based Sorbent Development
TDA prepared several different copper-based sorbents for the modified Deacon reaction (Equation 2) during the Phase II research. These sorbents incorporated different precursor materials, formulations, additives, and firing conditions. We prepared >40 different sorbents and screened them for strength, surface area, porosity and HCl sorbent capacity. Of the >40 samples, 28 sorbents had adequate crush strength, porosity, and surface area. Those 28 samples were then tested for chloride capacity in screening tests that measured their maximum Cl loading (exposure to HCl between one and four hours) and their Cl content following regeneration for a similar period. Ten where selected for screening in a Thermo-Gravimetric Analyzer (TGA).
Out of the >40 samples, we selected two sorbents -- Rita-3LCM and Rita 4 -- as promising, and performed additional testing on them (i.e., multiple cycle breakthrough tests) on only those two sorbents.
1.3.1. Breakthrough Apparatus
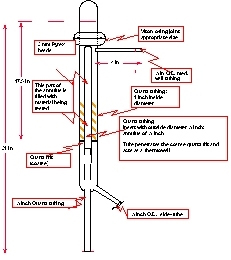
Breakthrough tests on the sorbents were conducted in the apparatus shown in Figure 1. During absorption ~5% HCl gases diluted with a balance of helium entered the bottom of the reactor and flowed up through an annular reaction zone containing the sorbent. An external tube furnace maintained the sorbent at the desired temperature.
Figure 1. Experimental Reactor
The gases leaving the reactor where then analyzed for HCl, Cl2, O2, and water. HCl and Cl2 were measured with continuous infrared (IR) analyzers. O2 and water were measured in a Gas Chromatograph (GC). To simplify the measurement of O2, we used helium as the inert gas in absorption since GCs have difficulty separating nitrogen and oxygen when water vapor, HCl, and Cl2 are also present in the column.
TDA selected an annular reaction zone to keep the sorbent isothermal. Since the absorption of HCl is known to be exothermic, we designed the reaction zone to allow transfer of this heat with minimal temperature differences. This approach allowed us to measure the performance of the sorbent as a function of temperature.
The reactor was constructed of quartz to minimize reactions with HCl. A quartz tube in the center of the reactor formed the annulus. We inserted thermocouples into this tube to measure the temperature of the sorbent bed while keeping the thermocouples isolated from the very corrosive HCl, Cl2, H2O, Cl2 gas stream.
1.3.2. Breakthrough Results
TDA identified two different copper-based sorbents as having good potential: Rita-3LCM and Rita-4.
Rita-3LCM
Fresh Rita-3LCM sorbent was ground and sieved to a 20-40 mm mesh size and then loaded into the annular test reactor. It was tested under experimental conditions that were identical to the ones used for the Rita-4 sorbent: 330oC and 400oC isothermal cycling and 330oC - 400oC temperature swing cycling. The physical characteristics of the fresh sorbent, including crush strength and surface area, were measured. The XRD spectrum of the fresh sorbent was taken and then compared with an XRD pattern of fresh copper oxide.
Prior to cycling, the sorbent was conditioned with 20%HCl/He at 350oC and then regenerated at the same temperature for 27 hours. The sorbent's activity was tested under isothermal conditions at 330oC and 400oC and under temperature swing conditions with 300oC absorption followed by a 400oC regeneration.
For the first performed 330oC isothermal cycle, the sorption capacity of the sorbent was measured to be 24.9 wt%; for comparison, the sorption capacity of the Rita-4 sorbent was only 13wt%. One possible reason for such a high sorption capacity for the Rita-3LCM sorbent could be that the sorbent was not aged enough; more cycles were needed in order to get representative data. Two more consecutive 330oC isothermal cycles were performed. The sorption capacity of the sorbent was measured to be 18.6% and 15.6%, respectively. The slight decrease in sorption capacity was due to the fact that the second absorption was performed on a partly regenerated sorbent. (It took ~12 hours to establish a 55% Cl- balance.) Due to a very long regeneration time, the 330oC isothermal process was not of particular practical interest and, therefore, no more 330oC isothermal cycles were performed.
Figure 2. Temperature swing cycling details for the Rita-3LCM sorbent.
Next, temperature swing cycles were performed (330oC absorptions, 400oC regenerations). Conducting the process in the temperature swing regime has two major advantages: 1) higher loading and 2) shorter regeneration time. As can be seen from the plot in Figure 2, regeneration is on the same time scale as absorption (4 and 5 hours, respectively). The Cl2 evolution level during the regeneration process was ~2.5 vol%.
For the next two temperature swing cycles performed, a problem with the mass flow controller led to uncertainty in the oxygen flow rate, and more cycles were run to get reliable data. The sorbent also underwent one isothermal cycle at 400oC. As with the Rita-4 sorbent, the Rita-3 sorbent's 400oC isothermal sorption capacity was lower than the sorption capacity for the temperature swing and 330oC isothermal cycles: 12.7 wt% compared to 24.0 and 17.1 wt%, respectively. Absorption lasted for ~3 hours, while a 9-hour regeneration (Cl2 tailing time) was not counted, resulting in an 85% Cl2 balance. The level of Cl2 liberated during regeneration was only ~1.6 vol%. Table 1 presents a summary of the cycles conducted.
| Absorption temperature (oC) | Regeneration temperature (oC) | Cl- balance | Sorption capacity |
| 330 | 330 | 55 | 18.6 |
| 330 | 330 | - | 15.6 |
| 330 | 400 | 82.6 | 19.6 |
| 330 | 400 | 114* | 22.2 |
| 330 | 400 | 99* | 30.35 |
| 330 | 400 | 73 | 23.82 |
| 400 | 400 | 85 | 12.87 |
Rita-4
We conducted multiple cycle tests on RITA-4. Three cycles were conducted on the sorbent with 1) temperature swing with 330oC absorption and 400oC regeneration and 2) isothermal absorption and regeneration at 330oC. These tests were conducted with 5.6% HCl in He as the absorption gas (dry and no oxygen). Regeneration was conducted with 100% O2 in the inlet. The initial temperature swing cycle was conducted with equal regeneration and absorption times; unfortunately, that did not regenerate the sorbent. We changed the cycle to include overnight regeneration in oxygen, but even then, in most cases the sorbent was not fully regenerated (a small concentration of Cl2 was still exiting the bed). With the overnight regeneration, we measured a cyclic bed capacity for HCl of 19% wt.
We then conducted isothermal absorption and regenerations at 330oC on the same sample of Rita-4. With overnight regeneration, a significant fraction of the bed was still not regenerated (~75% chlorine recovery). In this case we measured a 13% (wt) HCl capacity of the sorbent. The last set of experiments performed was the isothermal cycling at 400oC.
A stream of 5.6% HCl/He balance was directed through the sorbent bed until the HCl concentration in the stream exiting the sorbent bed became equal to the HCl concentration in the inlet stream. Next, the flow of HCl through the sorbent bed was stopped, and the He flow continued, until a stable HCl level was established. Thus, all of the adsorbed HCl was removed. Finally, a flow of pure O2 was started at a rate of 50ml/min. The sorbent was regenerated in O2 until the Cl2 concentration in the effluent stream reached a stable level of 0.5%. Two cycles were conducted; each had a measured sorption capacity of ~8.61 wt% and a calculated Cl- balance of ~85%.
Figure 3. Details of the isothermal cycling at 400oC for Rita-4 sorbent.
The plot in Figure 3 presents the details of the 400oC isothermal cycling. The sorbent's 400oC isothermal sorption capacity was lower than the sorption capacity for the temperature swing and 330oC isothermal cycles: 8.6 wt% compared to 18.8 and 13.0 wt%, respectively. Table 2 presents the overall summary of the conducted cycles.
Figure 4. Details of the 400oC isothermal complete regeneration cycle with reduced O2 flow rate.
The last 400oC isothermal cycle performed differed from all of the previously conducted cycles. (The cycling details are given in Figure 4.) Regeneration lasted until the sorbent was fully regenerated. As can be seen from the plot, it took ~35 hours to establish a 100% Cl- balance. The level of Cl2 liberated during regeneration was only ~3 wt% for all of the conducted cycles, which was low when compared to the theoretical expected value of 10 wt%. One possible reason for a low Cl2 evolution level was that regeneration could be a reaction rate limited process. To test for this possibility, the flow rate of O2 during the last regeneration was reduced from 60 ml/min to 24 ml/min. If regeneration were a reaction rate limited process, the level of Cl2 would go up. But, as can be seen from the plot, the Cl2 level did not change, thus proving the hypothesis.
| Absorption temperature (oC) | Regeneration temperature (oC) | Cl- balance (%) | Sorption capacity (wt %) |
| 330 | 400 | 81.8 | 18.5 |
| 330 | 400 | 83.0 | 19.0 |
| 330 | 400 | 86.5 | 19.0 |
| 330 | 330 | 64.4 | 12.7 |
| 330 | 330 | 77.7 | 13.6 |
| 330 | 330 | 83.7 | 12.8 |
| 400 | 400 | 85.0 | 8.6 |
| 400 | 400 | 83.4 | 8.6 |
1.3.3. Thermodynamic Analysis of the Copper Oxy-Chloride System
Both the Rita-3LCM and Rita-4 sorbents demonstrated high chlorine capacity and the ability to be regenerated for multiple cycles. Unfortunately, even with temperature swing and regeneration in a large excess of pure oxygen, the Cl2 concentration in the regeneration was very low. While there were periods where the Cl2 concentration was relatively high, most of the Cl2 on these copper-based sorbents was released at low concentrations and over a time frame of several hours.
We conducted thermodynamic analyses to estimate the properties of the copper oxychlorides (Cu2oCl2). The data qualitatively matched the measured data for our Rita-3 and Rita-4 sorbents with the predicted low concentrations of Cl2 during regeneration.
These analyses show that the reactions sequence is more complicated than described in Equation 2. The absorption step is still the chlorination of copper oxy-chloride to cupric chloride. However, the regeneration oCcurs in two steps. The first step is simply the disproportionation of cupric chloride to cuprous chloride and chlorine (CuCl2 -> CuCl + ?Cl2). The disproportionation oCcurs rapidly and generates the high concentrations of chlorine seen in the tests (i.e., at the start of the regeneration in Figure 2 and Figure 3). However, since CuCl2 and CuCl are soluble in each other, solution chemistry affects the equilibrium chlorine concentration. Therefore, as the reaction produces chlorine, the equilibrium concentration of chlorine in the gas continuously declines as the solid solution is formed. The Cl2 concentration rapidly falls from its high value.
Equation 3. Steps in modified Deacon reaction with copper oxy-chloride sorbent.
Absorption: 4HCl +2Cu2oCl2 < 2H2O + 4CuCl2
Regeneration: Two Steps: Disproportionation followed by oxidation
Disproportionation: 4CuCl2 < 2Cl2 + 4CuCl
Oxidation: 4CuCl + O2 < 2Cu2oCl2
Net: 4HCl + O2 < 2H2O + 2Cl2
The second step in the regeneration is the oxidation of the cuprous chloride (CuCl) to copper oxy-chloride (Cu2oCl2). Based on our experimental data, we estimate the thermodynamic properties of the copper oxy-chloride (Cu2oCl2), properties that were unknown at the start of this work. The data show that the reaction is equilibrium limited and that about 3% Cl2 in 97% O2) is required to regenerate the sorbent. Equation 3 shows the actual steps in the modified Deacon reaction using copper as the sorbent.
High concentrations of Cl2 were measured by TDA and predicted from the thermodynamics; but these concentrations were of short duration.
Minet (1991) and the PDU tests at Carburos Metallicos also measured high concentrations of Cl2, but their gas analyzers did not measure continuously. To determine if our sorbent operated by a different mechanism than the previous sorbents, APCI supplied TDA with some of the sorbent that was tested at Carburos Metallicos, YBA-347991. We conditioned the sorbent, then conducted four cycles with temperature swings using the same conditions as the previous tests with Rita-3 and Rita-4.
Figure 5. Temperature swing on YBA-347991.
Figure 5 presents the breakthrough data on YBA-347991. The sorbent shows the same high initial Cl2 peak followed by a slow regeneration that produces low concentrations of Cl2 over a long period of time. The maximum Cl2 concentration was 13% (versus 18% with Rita-3). The pre-breakthrough HCl loadings of YBA-347991 (6.6% maximum) are low compared to TDA's Rita series of sorbents (e.g., ~20% HCl for Rita-3) but that was expected, given that the YBA-347991 was an impregnated sorbent. Qualitatively, YBA-347991 and TDA's Rita sorbents are similar, and the sorbents probably have similar chemistry.
TDA provided the experimental data on the copper-based sorbents (Rita-3, Rita-4, and the YBA-347991) to our industrial partners, APCI and MIC. Since all of the copper-based sorbents release most of their Cl2 at low concentrations and over a long period of time, the data clearly showed that the copper-based sorbents would require very large reactors and a large cost for oxygen, or expensive Cl2/O2 separation equipment to recycle the oxygen.
Having previously conducted an economic analysis on the process, our industrial partners informed us that none of the copper-based sorbents (Rita-3LCM, Rita-4 or their own YBA-347991) would meet their cost/performance goals.
1.4. Reductive Chlorination Sorbent and process Development
Given the poor results of the copper-based sorbents, TDA evaluated a new approach to HCl conversion to Cl2. One way to avoid the low Cl2 concentrations during the regeneration in the copper system is to provide additional chemical potential in the form of a chemical reactant. By using the reducing power of CO, which is already present in some of the waste gas steam from which we wish to recover chlorine, one has available a very large chemical potential: (CO(g) + ?O2(g) = CO2(g) DH= -67.79 kcal, DG =-53.57 kcal, K = 6.109 (10)34 at 400oC). That chemical potential could be used to shift equilibrium and avoid the limitations of the Deacon reaction.
We call this new process that uses the reducing power of CO or another reducing gas (e.g., H2, CH4, etc.) "Reductive Chlorination." This new process will work with cerium, iron, manganese, tin, and possibly other redox metal oxides (i.e., metals with more than oxide). Since manganese does not have stable, volatile chlorides (and is not vaporized in either reducing or oxidizing gases), we selected manganese as the redox metal oxide for our evaluation of this new approach.
Table 3. Reductive Chlorination with manganese.
MnO + 2HCl(g) = MnCl2 + H2O(g)
2MnCl2 + 3.5O2(g) = Mn2O3 + 2Cl2 +2O2(g)
Mn2O3 + CO(g) = CO2(g) + 2MnO
Net Reductive Chlorination Reaction
CO(g)+1.5O2(g)+4HCl(g) = CO2(g)+2H2O(g)+2Cl2(g)
TDA prepared a process description based on the reactions given in Table 3; Table 4 presents the heat of reaction, free energy change, and equilibrium constants for each reaction.
Table 4. Reactions using manganese as the redox sorbent for the conversion of HCl to Cl2.
MnO + 2HCl(g) = MnCl2 + H2O(g): DH= -36.047kcal, DG = -16.523 kcal, K =2.32 (10)5 at 400oC.
2MnCl2 + 3.5O2(g) = Mn2O3 + 2Cl2 +2O2(g) DH= -0.432 kcal, DG = +0.235 kcal, K = 0.839 at 400oC.
Mn2O3 + CO(g) = CO2(g) + 2MnO DH = -23.301 kcal, DG = -27.01 kcal, K = 5.88 (10)8 at 400oC.
Net Reductive Chlorination Reaction
CO(g)+1.5O2(g) +4HCl(g) = CO2(g)+2H2O(g)+2Cl2(g); DH -95.827kcal, DG = -59.819kcal at 400oC.
Versus Deacon Reaction:
4HCl(g) + O2(g) = 2H2O(g) + 2Cl2(g); DH = -28.039 kcal, DG = -6.252 kcal; K = 1.07(10)2 at 400oC
The equilibrium constant is very favorable in both the reduction step (Mn2O3 + CO(g) = CO2(g) + 2MnO) and the chlorination step (MnO + 2HCl(g) = MnCl2 + H2O(g)). However, the equilibrium constant is near unity for the regeneration reaction, so we anticipate that an equal number of moles of chlorine and oxygen are present in the regenerated gas. A high pressure Cl2 condenser is used to separate the oxygen from the chlorine, and the O2 is recycled (see discussion in Section 1.4.3 Reductive Chlorination process and Economics).
The addition of the reducing gas and the redox metal oxide shifts the equilibrium (K = 2.648(10)19) versus the Deacon reaction (K = 1.07(10)2) for the net reactions as written. Since all of the equilibrium constants are relatively large in each step of the reductive chlorination, the process can proCeed as written.
1.4.1. Impregnated Manganese Chloride (MnCl2) Sorbent
To see if practice follows theory, we prepared one manganese based HCl sorbent by impregnating preformed alpha alumina pellets with MnCl2.. We selected a 550oC isothermal operation for the sorbent system during reduction, chlorination, and regeneration. We loaded our existing apparatus with this sorbent and conducted several cycles in a fixed bed reactor at 550oC and ~545 torr. The inlet concentration was 4.5% HCl, 2% H2, 2% CO2, with the balance He. We conducted regenerations in both oxygen and simulated air (21%O2, with the balance He). During the reduction we used 2% H2, 2% CO2 and the balance He.
The first three cycles, conducted at low Space VeloCity (SV), showed that the sorbent 1) needs to be reduced before HCl is added, 2) regenerates rapidly with air or O2, and 3) reaches <100ppmv HCl before breakthrough during the absorption (or chlorination).
Figure 6 shows the reduction, chlorination and regeneration of the Mn based sorbent during the fourth cycle. We first reduced the sorbent with 2%H2 for 2 hours and then we flowed 4.5% HCl gas through the reactor at a SV of 1,400 h-1, which was the highest rate that the apparatus would allow at that time. The HCl outlet concentration was very low (<100ppmv or 0.01%) at pre-breakthrough, and the sorbent absorbed 12.6% HCl (wt). After breakthrough, we purged the reactor with He and then regenerated the sorbent by flowing O2 at a SV of 700 h-1. We measured a high concentration of Cl2 (30-35% Cl2, with the balance O2), more than 10 times the average concentration of the copper based sorbents.
Figure 6. Cycle 4 with high O2 flow during regeneration.
1.4.2. Manganese Oxide Geode Sorbent
Given the success of the impregnated manganese sorbent, TDA prepared a new geode sorbent that had both a high Mn content and a high Cl loading: Cl = >24% (wt). The first 4 cycles were conducted at SV = 1,700-1. In the first cycle the Cl2 exceeded the range of the analyzer (50% vol). At the test conditions the equilibrium concentrations were 54% Cl2 and 46% O2. Later testing found measured concentrations of up to 50% Cl2 during regeneration.
We then mixed the Mn geode sorbent with inert alumina (so that the sorbent only oCcupied 33% of the bed) and conducted additional tests at much higher SVs. The tests with or without the inert alumina showed the same results. We then conducted 15 cycles at a SV=5,100h-1 which demonstrated repeatable performance with low HCl concentrations pre-breakthrough and high Cl2 concentrations during regeneration with high chloride loadings. We then conducted two more cycles at an even higher SV ( 6,000h-1).
Figure 7 presents the 17th and 18th cycles on this sorbent at the highest SV (6,000 h-1). The three steps of the reductive chlorination process are conducted in each cycle.
? Cycle 17: Reduction from 0 to 0.5 hr, chlorination for 0.5 to 1.25 h, purge from 1.25 to 1.35 hr, regeneration from 1.35 to 2.15 h and purge from 2.15 to 2.3 h.
? Cycle 18: Reduction form 2.3 to 2.8 h, chlorination for 2.8 to 3.3h, purge from 3.3 to 3.4 h, and regeneration from 3.4 to 4.2 hr.
High levels of chlorine are seen in both regenerations (>30%), with very low pre-breakthrough HCl concentrations.
Figure 7. High SV testing of manganese geode for reductive chlorination.
1.4.3. Reductive Chlorination process and Economics
Based on the process chemistry, we prepared a preliminary process description and sized all of the major components.
Figure 8 shows the system's three reactors. Each of the reactors is a fixed bed containing the manganese sorbent. When a reactor finishes its function in the indicated mode, a system of valves redirects the gas flow to the next reactor. These valves are not shown for the sake of simplicity.
Figure 8. Reductive chlorination reactors.
In Reactor 1 the low-pressure wet feed (~5 psig) is mixed with the oxygen and chlorine that are being purged from the chlorine separation unit. In the reactor, the dry feed and additional natural gas are fed into the combustor, while the oxidizing and reducing gas are burned to raise the gas streams to the operating temperature (550oC). The hot gases flow through the MnO and react to the MnCl2.
In Reactor 2 the MnCl2 is regenerated with high-pressure oxygen (10 atm, 147 psia) to Mn2O3. The regeneration exhaust gas is assessed to contain 27.2% Cl2, with the balance O2 (even though equilibrium is >35% O2 at these conditions). The regeneration exhaust gas stream then flows to the chlorine separation unit where the gases are cooled, the water and HCl removed, and the chlorine condensed.
Table 5 presents the major process assumptions used in that analysis. Note that the analysis was performed with assumptions that were more conservative than our measured data [lower SV than measured (i.e., 4,300 versus 6,000 h-1, lower Cl2 concentrations than measured (i.e., 27.2% versus >35%) and lower Cl loadings than measured (i.e., 11% versus 24% wt)]. Thus, our actual performance is considerably better than the performance assumed in this analysis.
Table 5. process assumptions.
o 550?C absorption, regeneration, & reduction
- Low pressure reduction and absorption (chlorination)
- SV = 4,300 h-1; less than measured in Figure 7
o Avoids heat exchangers in bed
- CO + natural gas burned in inlet HCl to heat gases to 550?C
- Water quench reduces low HCl gas to <400?C
o 3 Fixed Beds (SVabsorption = 4,300 h-1)
- 11% Cl loading on sorbent
o Pure oxygen supplied
- 27.2% Cl2 in regeneration; Less than measured with Mn geode sorbent
- 10% of O2 supplied purged to removed impurities, Cl2 in purge is recycled.
- High pressure operation, 10 atm in reactors
o Refrigeration to separate Cl2 from O2
- Regeneration gas is cooled to condense Cl2
- Recuperative cooled and Cl2 re-evaporated
TDA conducted the analysis with two sets of space veloCities and cycle times and with two sets of economic assumptions in order to evaluate the sensitivity to these items. We also evaluated the cost of the chlorine produced with either a wet or dry feed.
Table 6. Results of the economic analysis.
o 25% Fixed Charge Rate (FCR) (4-year payback)
- With wet oxidizing gas feed (1996$)
- $7.1 million capital cost
- $84/ton ($72/ton with 15% FCR)
o $64 /ton with dry reducing gas feed ($52/ton with 15% FCR)
o Versus goal of <$100/ton
- Less than half the cost of fresh Cl2 @$200/ton
- with 15% capital charge and <$20 million capital cost
o Latest test data shows SV up to 6,000 h-1
- 24% Cl loading on sorbent
Currently, fresh chlorine from the chloro-alkali process costs about $200/ton (Chemical Marketing Report, March 10, 1997). Our industrial partners set a goal of half the current market price (i.e., $100/ton with a 15% capital charge rate) as the cost at which they would be willing to invest in a new technology. Given the results of TDA's economic analysis, the estimated costs of reductive chlorination could easily meet that goal.
Using our test data, APCI conducted an independent cost analysis of the reductive chlorination process, and came up with estimated capital and operating costs that were essentially equal to those estimated by TDA.
1.5. Conclusions and Recommendations
This project was originally designed to develop improved sorbents for the modified Deacon reaction, but it evolved into the development of a new process and sorbent. Conclusions and recommendations are described below.
1.5.1. Copper-based Sorbents
TDA developed high capacity copper-based sorbents for the modified Deacon reaction that demonstrated the property of regeneration for multiple cycles and removed most of the HCl during absorption. However, the chlorine concentrations were relatively low during regeneration and required very long times to fully regenerate the sorbent.
Based on the experimental data, TDA estimated the thermodynamic properties of copper oxychloride, which were unknown prior to this work. We conducted a thermodynamic analysis of the process, and concluded that the process has several steps as shown below:
Absorption: 4HCl +2Cu2oCl2 < 2H2O + 4CuCl2
Regeneration: Two Steps: disproportionation followed by oxidation
Disproportionation: 4CuCl2 < 2Cl2 + 4CuCl
Oxidation: 4CuCl + O2 < 2Cu2oCl2
Net regeneration 4CuCl2+ O2 < 2Cl2 + 2Cu2oCl2
Net reaction: 4HCl + O2 < 2H2O + 2Cl2
TDA concluded that the same process was oCcurring for all of the copper-based sorbents -- Rita-3LCM, Rita-4, copper chromite and YBA-347991. Due to the disproportionation step, all copper-based sorbents will have periods of high Cl2 concentrations during regeneration. However, since CuCl2 and CuCl are soluble in each other, the concentration of the Cl2 continuously decreases as the regeneration proCeeds. Unfortunately, the oxidation step is much slower than the disproportionation step; therefore most of the chlorine is released at low concentrations over a long regeneration time. These effects mean a large excess of oxygen is required to regenerate the sorbent.
Prior to the initiation of the EPA Phase II, APCI and MIC performed an economic analysis on the original modified Deacon process using the YBA-347991 sorbent, and it did not meet the cost and performance goals. Although our improved sorbents (Rita-3LCM and Rita-4) have better chloride capacity, high Cl2 concentrations, and equal HCl removal, the slow regeneration and low average Cl2 concentration for all of these sorbents caused them also to fall below the performance goals. We concluded that the copper-based sorbents for the modified Deacon reaction process would never reach their cost/performance goals.
1.5.2. Reductive Chlorination
We selected manganese as the redox metal oxide for the reductive chlorination process. The sorbent has a high loading of chlorine (24%) and can be regenerated for many times without loss of capacity. Low levels of HCl leaving the reactor are attained and high concentrations of chlorine are produced during regeneration.
Using a 25% Fixed Charge Rate (FCR) (4 year payback) we estimated that the cost of chlorine, a wet oxidizing HCl waste gas feed (1996$) is $7.1 million, and the resulting cost of chlorine is $84/ton ($72/ton with 15% FCR). When operating with a dry reducing gas HCl waste stream, the cost for chlorine drops to $64/ton ($52/ton with 15% FCR). The economic analysis was conservatively performed with lower chlorine loadings and lower space veloCities than can be attained by our sorbent. We provided test data to our industrial partners, who conducted an independent cost analysis of the reductive chlorination process. The estimated capital costs and operating costs were essentially equal to those estimated by TDA, even though a different costing methodology was used.
Our industrial partners set a goal of $100/ton with a 15% capital charge rate (half of the current market price of $200/ton Cl2 ), which we could easily meet.
Given the excellent performance of our sorbent and the excellent economic potential of the reductive chlorination process, continued development of both are recommended. At this time, additional research on the operating conditions for the wet gas stream are being performed under funding by our industrial partners.
Supplemental Keywords:
Small business, SBIR, pollution prevention, engineering, chemistry, EPA., Sustainable Industry/Business, RFA, Air, Scientific Discipline, Water, Waste, Toxics, Hazardous, Technology for Sustainable Environment, Chemical Engineering, Engineering, Chemistry, Sustainable Environment, HAPS, Environmental Chemistry, Engineering, Chemistry, & Physics, Chemistry and Materials Science, Hazardous Waste, EPCRA, cleaner production/pollution prevention, Drinking Water, New/Innovative technologies, Environmental Engineering, chemical use efficiency, clean technology, process modification, long-life sorbent, chlorinated hydrocarbons (CHCs), treatment, Chlorine, pollution prevention, chlorinated hydrocarbons, hydrogen chlorine, low-cost sorbent, CuO sorbent, chemical contaminants, chlorine recovery, sorbents, sorbent based process, in-process changes, drinking water treatment, recovery, chemical byproducts, cleaner production, Hydrogen chloride, in process treatment, in process recycling, chlorinated solvents, Deacon reactionSBIR Phase I:
A Long-Life, Low-Cost Sorbent for the Conversion of HCl to ChlorineThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.