Grantee Research Project Results
Final Report: Human Safe, Near-Field Infection Protection (NIP) for Continuous Pathogen Inactivation in Air and on Surfaces
EPA Contract Number: 68HERC22C0017Title: Human Safe, Near-Field Infection Protection (NIP) for Continuous Pathogen Inactivation in Air and on Surfaces
Investigators: Rasansky, Richard
Small Business: XCMR Inc
EPA Contact: Richards, April
Phase: I
Project Period: December 1, 2021 through May 31, 2022
Project Amount: $99,771
RFA: Small Business Innovation Research (SBIR) Phase I (2022) RFA Text | Recipients Lists
Research Category: SBIR - Homeland Security , Small Business Innovation Research (SBIR)
Description:
XCMR is developing breakthrough respiratory protection from infectious diseases and infection control for pathogens by commercializing a new generation of devices for safe, continuous use utilizing Far UV-C radiation (222 nm). This relatively short wavelength offers effective microbial disinfection typically associated with conventional germicidal UVC lamps (254 nm) but with much-reduced human health hazards since it does not penetrate beyond the outer layer of human skin or outer tear layers of the eye. Expected uses of XCMR’s Near-field Infection Protection (NIP) device include biosafety protection during interpersonal interactions in a variety of stationary settings found generally in offices, schools, healthcare, hospitality, retail, transportation and many others.
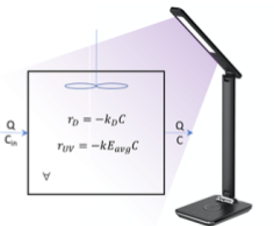
The central hypothesis examined in the Phase I was that Far UV-C radiation, specifically the radiation from a KrCl* lamp, could be applied in a manner that accomplished effective disinfection of a personal air space, while keeping exposure of human tissues below the American Conference of Governmental Industrial Hygienists (ACGIH) Threshold Limit Values (TLVs). The project involved experiment-based measurements and modeling to examine this hypothesis. The experiments and simulations involved a unit air volume (1 ft3). Experiments were conducted to first measure the spatial distribution of UV222 radiant energy from a device that resembles a desktop lamp configuration (Figure 1) and then quantify inactivation of viral surrogates at strategic points within that irradiation field. The device incorporated optically-filtered, commercially-available KrCl* sources that provide directional character for illumination. Numerical simulations were applied to examine the potential for these systems to achieve effective inactivation of airborne pathogens, while controlling human exposure to below ACGIH limits. The results of this work have provided data for the development of prototype devices for application in a variety of settings and provided a basis for a Phase II evaluation of a prototype for the disinfection of a more dynamic air space, including aerosolized viruses in a more complex and realistic fluid flow scenario.
Figure 1.
| No. | Performance Schedule of Milestone Objectives |
|---|---|
| 1 | Simulations of disinfection efficacy over a wide range of system configurations and operating conditions; will be compared with results of experiments described above and will inform CFD-based simulations |
| 2 | Quantitative measurements of the spatial distribution of UV222 energy around lamp fixtures; will inform potential for virus inactivation and design of spherical point disinfection experiments |
| 3 | Quantitative measurements of surrogate virus (phage) inactivation within a unit air volume; will inform model simulations |
| 4 | Detailed simulations of the physical behavior of the system; results will be used to inform prototype development |
| 5 | Prototype device to be developed for commercialization |
Summary/Accomplishments (Outputs/Outcomes):
Milestone 1: Mathematical Modeling
A mathematical model was developed for use as a tool to allow users to examine the effects of input parameters on the performance of a near-field infection protection system. The model was used to simulate behavior of the proposed system over a range of operating conditions. For simplicity, the irradiated volume (control region for the model) is assumed to be cubic in shape; as such, it is possible to define the geometry of the control region by a single number, the length of an edge (L). A definition sketch for the model is presented in Figure 2.
The results of this simulation indicated that achieving effective inactivation of airborne SARS-CoV-2 (and other airborne pathogens) depends on the volume of air that is irradiated and the average fluence rate within the irradiated zone (the control region for these simulations). For the input variables used in these simulations, it appears that an average fluence rate in the range of 1000 µ𝑊/cm2 to 10, 000 µ𝑊/cm2 will be required to achieve treatment that is comparable to the protection provided by an N95 mask. However, it is important to recognize that the results of these simulations will depend on the values of all input parameters. Another important aspect of the behavior of this system will be the balance between delivering a sufficient UV222 dose to achieve effective inactivation of the airborne pathogens, while maintaining human exposure below the TLVs.
Figure 2: Sketch to describe the dynamic behavior of air quality in a small irradiated volume.
Milestone 2: Fluence Rate Field measurements
To better represent the true UV fluence rate in a complex irradiation field, a micro-fluorescent silica detector (MFSD) was utilized and its output was correlated to a calibrated radiometer while measuring output from irradiation from two unique Far UV-C sources. It was discovered that the use of a planar radiometer did not accurately reflect the true irradiance delivered to the virus suspension and the MFSD was used to improve the accuracy during the testing.
Milestone 3: Air Volume Spherical Point Disinfection Measurements
Following a detailed mapping of the irradiance field in the 1 cubic foot volume with 2 and 3 lamps using the MFSD, the next set of tests measured inactivation at specific points within the field, using spherical quartz bulbs. The custom-designed bulbs were approximately 1 cm in inner diameter and filled with solutions of T1 phage for inactivation testing. The time required to achieve specific log reduction value (LRV) targets were determined using two different experimental setups with two and three Eden Park lamps, respectively. The time required to reach target LRVs at similar points in the cube was decreased by 30-40% with a 3 lamp configuration versus a 2 lamp configuration of Eden Park lamps. The amount of time required for target LRVs could be reduced further by using lamps with higher irradiance levels such as Ushio where the target LRV can be achieved in about 5-8% of the total time observed with the Eden Park lamps at the same points in the cube.
Milestone 4: CFD-based Simulations
A computational fluid dynamics (CFD) model of the fluence rate produced by Eden Park KrCl excimer lamps was performed to explore the impact of flow on the resulting pathogen inactivation within a one cubic foot area element. The model did predict similar spatial locations of the maximum fluence rate. However, there are differences depending on whether the cosine angle factor is applied. The values are different between the experimental results and are very dependent on the type of sensor used to record the local fluence rate (i.e., whether the sensor can detect 360 exposure of the fluence rate at the specific location). The model is also sensitive to the true power released at the measured wavelength. In the model, we used the value reported by the manufacturer, which is subject to manufacturing lot variability and age.
Significant pathogen removal was observed when the air was relatively still. However at higher local air flow velocities, there was a significant drop off in inactivation performance. These results suggest that higher lamp power may be required to allow for significant inactivation under local air flow velocities that are typical of human comfort in indoor rooms.
It should be noted that the air velocity condition was specific to a flow that was directed towards the UV lamp light ring and the potential worker sitting on the effluent side of that air flow. Potential real conditions will probably see non-sustaining air velocity movement in different directions that is a function of the room HVAC diffuser system configuration. To demonstrate this concept of a UV disinfection air replacement technology (UVDART), CFD simulations were performed with a reactor located below a desk that disinfects air and sends it out to the desk surface towards a human sitting using diffuser plates above the desk surface. The working occupant at the desk received disinfected air that has a removal fraction over 95 percent. The range of removal in the air surrounding the occupant is due to the mixing with the room air that has a high concentration of airborne pathogens. In this example, the occupant is not exposed to UV light directly and would not be subject to the TLV restrictions with the design concept,
Milestone 5: Prototype Development and Commercialization Progress
Constantly refining and iterating our design concepts and requirements, driven by our research in this Phase I effort, we progressed towards commercialization with the completion of a detailed Commercialization Readiness Assessment Report (CRAR) which we expect will lay the foundation for our advanced commercialization plan in our Phase II efforts.
In addition to this CRAR, and based on the results and learnings obtained from our research performed under this contract, we have delivered a detailed, reasonable and actionable execution plan for initial prototype design and delivery for a Phase II timeline, working closely with our contract manufacturing partner, Nortech Systems. We have further identified and signed MNDAs with other Far UV-C emission source OEMs for testing in response to our lab results suggesting the need for higher energy sources of emission.
Overall, we are confident from the work performed in this Phase I effort that we can successfully deliver an efficacious PPE device prototype configured for commercial application, and in full compliance with the objectives of this project and research topic in a Phase II period of performance.
Conclusions:
Summary Conclusions informing a path forward in experimental design:
1. T1 bacteriophage is an easy phage to work with, and representative of respiratory virus inactivation kinetics, for field and lab experiments.
2. The micro-fluorescent silica detector (MFSD) can accurately and predictably represent the irradiance in a complex irradiation field and should be used, correlated to a calibrated radiometer, for 3-D irradiation measurements.
3. The current generation of Eden Park lamps used in this study are not powerful enough to inactivate within the exposure window for effective personal protection, however next generation micro-plasma lamps are expected to deliver much greater fluence.
4. Light intensity produced by current Eden Park lamps is too low to produce reasonable disinfection of a moving air stream that is typical for human respiration
5. Adding additional lamps or more powerful lamps into the irradiation field would improve the inactivation of viruses.
6. Ushio lamps can produce reasonable disinfection of an air stream under human room comfort conditions. However, human exposure to UV fluence rates might exceed TLVs in a short time frame, unless mitigated by dose limiting means.
7. Design and development of Near-Field Infection Protection (NIP) is constrained by the conflicting requirements of delivering a sufficient UV dose to inactivate airborne pathogens (to an acceptable degree), while controlling exposure of human skin and eyes to below the TLVs.
8. Disinfection of an air stream while avoiding human exposure to UV fluence rates can be achieved using a UV disinfection air replacement technology (UVDART) that places UV lamps in an enclosed reactor that produces effluent pathogen free air.
9. Computer simulation and modeling demonstrated and confirmed similar spatial light patterns compared to the physical experimental measurements.
10. NIP devices will benefit from the inclusion of geometric and/or optical features that allow discrimination between airborne pathogen exposure and human exposure to UVC radiation, including Far UVC.
11. The inclusion of reflective and absorptive media in NIP devices may allow acceptable discrimination of airborne pathogen exposure and exposure of human tissues.
12. Inclusion of a quantitative microbial risk assessment (QMRA) tool will allow identification of NIP system operating conditions that effectively limit disease transmission.
All expected and anticipated, initial commercialization goals were achieved in this Phase I program pursuant to our work plan and associated lab-based bench work. Our lab-based prototype UV reactor systems were designed, built and operated with commercial use cases and requirements in mind. All components and design factors were tested in our bench top experiments. Further, all data collected will be used to inform the design and specifications for our Phase II commercial prototype to be delivered in conjunction with our manufacturing partner, Nortech Systems, Inc. (see above Milestone 5: Prototype Development and Commercialization Progress.) To reiterate, we are confident from the work performed in this Phase I effort that we can successfully deliver an efficacious PPE device prototype configured for commercial application, and in full compliance with the objectives of this project and research topic in a Phase II period of performance.
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.