Grantee Research Project Results
Final Report: Cryptosporidium Capture in Sewage Impacted Waters
EPA Grant Number: SU840143Title: Cryptosporidium Capture in Sewage Impacted Waters
Investigators: Elliott, Mark , Jellison, Kristen L. , Olson, Julie , Maxcy-Brown, Jillian , Gunaydin, Serhat , Abungu, Vivian , Stahl, Leigha , Davidson, Rose , Mayerberger, Elisa , Early, Tana
Institution: The University of Alabama , Lehigh University
EPA Project Officer: Spatz, Kyle
Phase: I
Project Period: December 1, 2020 through November 30, 2021 (Extended to November 30, 2022)
Project Amount: $24,999
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2020) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
In the Alabama Black Belt, widespread rural poverty, limited access to sewer, and shrink-swell clay soils that preclude the use of conventional septic systems lead to both failing septic systems and the discharge of raw sewage from homes through "straight pipes" (Maxcy-Brown et al., 2021) (Figure 1). Sewage that is discharged to the surface typically pools in a ditch or trench until rain flushes the sewage and sediment into local waterways. The degree to which fecal microbes are associated with the suspended sediment load (SSL) has important implications for the sustained transport of these contaminants within the waterway, with ~60% of fecal microbes in surface water during storms associated with particles.

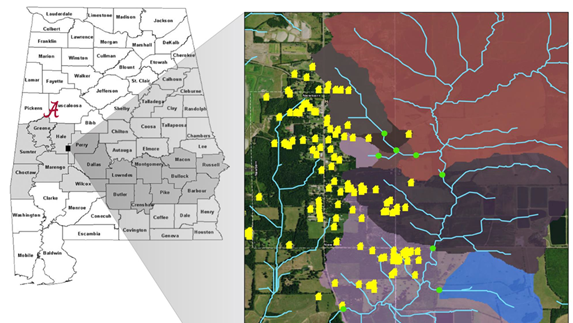
Therefore, an accurate and efficient way of capturing and monitoring suspended sediment and determining the microbial association is critical in understanding the potential impact that storms have on the fate and transport of fecal pathogens. Sampling surface water for fecal contamination typically involves occasional visits to a site, collection of a small grab sample, cold storage, incubation in the presence of a selective substrate and analysis for viable cells of fecal indicator bacteria (i.e. E. coli, thermotolerant coliforms, enterococci). While this approach has the advantages of being technically simple and broadly affordable, there are many drawbacks, including (1) microbial surface water quality is highly variable, with order of magnitude differences from one second to the next (e.g., Rao et al., 2015), (2) indicator bacteria concentrations can be poorly correlated with pathogens including Cryptosporidium (e.g., Wu et al., 2011); (3) The vegetative bacteria used as fecal indicators are far less environmentally robust than many pathogens (e.g., Savichtcheva and Okabe, 2006), with Cryptosporidium being especially environmentally robust as an oocyst-forming parasite. Therefore, a single grab sample tested for fecal indicator bacteria is completely inadequate to evaluate potential risk to human health from enteric pathogens in a body of water. Substantial progress has been made in molecular methods to detect multiple enteric pathogens in a single sample (e.g., Liu et al., 2013) and efforts to concentrate large samples in the field (e.g., Mull and Hill, 2012). However, there has been little to no effort to explore time-integrated sampling methods that have been used in sedimentology (e.g., Elliott et al., 2017a) and incorporate those into the detection of fecal microbes. This project uses novel time-integrated methodologies for capturing suspended sediment and Cryptosporidium in study three sites along Big Prairie Creek in Newbern, AL (Figure 2).
Figure 2: Map on left shows the state of Alabama ad location of Big Prairie Creek (BPC). Map on right shows zoomed in version, depicting the homes and sample locations (Maxcy-Brown et al., 2021)
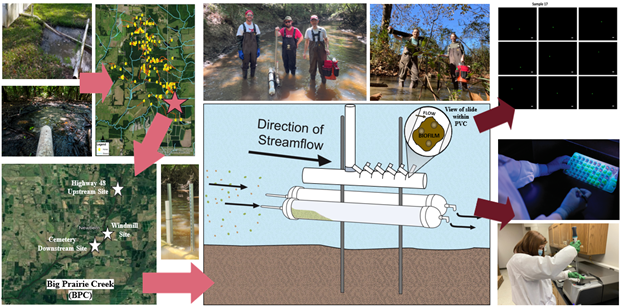
The novel time-integrated sampling procedure for Cryptosporidium takes advantage of the propensity of oocysts to stick to biofilms (Luo et al., 2017a). Growth of biofilm on glass slides yield a surface that can capture suspended oocysts, with addition of added calcium ions to enhance collection (Sarkosh et al., 2019); Cryptosporidium oocyst analysis and quantification is conducted at Lehigh University. The time-integrated mass sediment sampler (TIMS) method that has been used in sedimentology has been applied for the first time to the challenge of quantifying pathogens and other fecal microbes attached to suspended sediment (Perks et al., 2014; Elliott et al., 2017a). TIMS samples were analyzed for particle size distributions and analysis for fecal microbes (Figure 3).
In this P3 Phase I project, the laboratory and field research of two research groups was used to demonstrate that a simple, inexpensive time-integrated, biofilm-based surface can successfully capture oocysts from one of the most important human enteric pathogens, Cryptosporidium, in rural environments with a diverse set of point and non-point sources of human fecal contamination. Additionally, Cryptosporidium research capabilities at Lehigh University were leveraged to test the efficacy of increased calcium ion concentrations to enhance oocyst capture (Luo et al., 2017a) by designing, testing and deploying a tablet-based calcium dissolution device in the field. In parallel to the time-integrated biofilm-based sampling method, we integrated a time-integrated mass sediment sampler (TIMS)(Elliott et al., 2017a) to capture sediment for analysis of sediment-associated fecal microbes. Attachment of fecal indicator bacteria to sediment was evaluated using a vortexing and centrifugation-based method. E. coli, thermotolerant coliforms, and total coliforms were analyzed using IDEXX Colilert media and the Quanti-Tray 2000 system.
Figure 3: Workflow (left to right), showing effluent from straight pipes & flow to Big Prairie Creek (BPC); map of sample site locations; Field photos & diagrams of novel collectors, bi-directional TIMS & Cryptosporidium collector, showing how fecal-microbe associated SSL will be sampled both in TIMS (Elliott et al., 2017a) & biofilm on slides with crypto. attached; sample analysis for Cryptosporidium done at Lehigh University; analysis of fecal microbes via IDEXX & grain-size distribution conducted at the University of Alabama.
Summary/Accomplishments (Outputs/Outcomes):
Cryptosporidium samplers successfully collected oocysts during periods of 3-20 days. However, counts were low (0-9 oocysts) for every collection period, during both wet and dry weather, and showed no significant difference between any of the three sites (Table 1) and no difference between wet and dry conditions. Statistical hypothesis testing included parametric t-test and non-parametric tests, as dictated by Kolmogorov Smirnov Tests for normality, with paired analysis incorporated when appropriate (e.g., paired for sample date). The sampling process is somewhat time-consuming and labor-intensive and the high flow at times led to samplers being inaccessible or dislodged (dashes in Table 1 indicate that no sample was collected). The novel Cryptosporidium samplers show promise, but a more physically robust model and higher surface area for oocyst collection should be explored in future work.
| Date | Cemetery (downstream) | Windmill (midstream) | HWY48 (upstream) |
| 6/29/2021 | 5 | - | 3 |
| 7/1/2021 | 4 | 4 | 1 |
| 7/6/2021 | 0 | 0 | 5 |
| 7/8/2021 | 3 | 0 | 2 |
| 7/13/2021 | 0 | 8 | 4 |
| 7/15/2021 | 2 | 9 | 1 |
| 7/20/2021 | - | 0 | 0 |
| 7/22/2021 | - | 5 | 4 |
| 7/27/2021 | - | 1 | 1 |
| 7/29/2021 | 0 | 0 | 0 |
| 8/2/2021 | 1 | 0 | 0 |
| 8/5/2021 | - | - | 3 |
| 8/10/2021 | 0 | 1 | 0 |
| 8/16/2021 | 3 | - | - |
| 8/17/2021 | 0 | 0 | |
| 9/26/2021 | - | 1 | - |
| 2/18/2022 | 2 | 1 | 3 |
| 3/4/2022 | 0 | 3 | 1 |
| 3/11/2022 | 0 | 1 | 1 |
| 3/29/2022 | - | - | 4 |
Table 1: Cryptosporidium oocysts identified in biofilm samples collected from the three sites on Big Prairie Creek
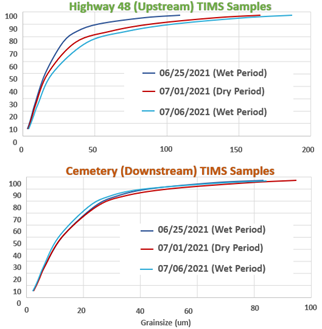
As expected, the concentration of fecal indicator bacteria in grab samples increased following rain events. However, the grain size analysis revealed a surprising finding: the grain size distribution following rain events was finer than under dry conditions (representative data in Figure 4). This is likely because the surface soil in our watershed is dominated by vertisol clay, whereas the creek bed incorporates larger grain soils. The increased surface area of finer sediment provides more opportunity for fecal microbes to become sediment associated.
Conclusions:
The TIMS and novel Cryptosporidium samplers are both made of PVC and provide inexpensive alternatives to conventional time-integrated or Cryptosporidium sampling methods in surface waters. This work reflects our efforts to enable affordable time-integrated approaches to the quantify Cryptosporidium in rural streams, differentiate sediment-associated fecal microbes, assess the potential of in-situ natural and engineered surfaces to capture pathogens as an alternative to conventional approaches to sampling for Cryptosporidium, and provide water managers with improved methods for evaluating the safety of water sources for drinking water supply and recreational use. While addition of calcium should be expected to increase attachment, the number of oocysts collected in Big Prairie Creek was insufficient to demonstrate that addition of calcium is effective in field settings. Further analysis of sediment for human-associated and cow-associated fecal markers is ongoing. This project demonstrates the potential for affordable time-integrated approaches to the quantify pathogens in rural streams, differentiate sediment-associated fecal microbes, assess the potential of in-situ natural and engineered surfaces to capture significant portions of pathogens that current conventional approaches fail to reflect, and provide water managers with improved methods for evaluating the safety of water sources for drinking water supply and recreational use.
Figure 4: Grainsize distributions for the three sampling periods (wet, dry, wet) at the upstream (Highway 48) and downstream (Cemetery) TIMS sampling sites
References:
Elliott, E. A., Monbureau, E., Walters, G. W., Elliott, M. A., McKee, B. A., & Rodriguez, A. B. (2017a). A novel method for sampling the suspended sediment load in the tidal environment using bi-directional time-integrated mass-flux sediment (TIMS) samplers. Estuarine, Coastal and Shelf Science, 199, 14-24.
Liu, J., Gratz, J., Amour, C., Kibiki, G., Becker, S., Janaki, L., & Haverstick, D. M. (2013). A laboratory-developed TaqMan Array Card for simultaneous detection of 19 enteropathogens. Journal of clinical microbiology, 51(2), 472-480.
Maxcy-Brown, J., Elliott, M. A., Krometis, L. A., Brown, J., White, K. D., & Lall, U. (2021). Making waves: Right in our backyard-surface discharge of untreated wastewater from homes in the United States. Water Research, 190, 116647.
Perks, M. T., Warburton, J., & Bracken, L. (2014). Critical assessment and validation of a time‐integrating fluvial suspended sediment sampler. Hydrological processes, 28(17), 4795-4807.
Rao, G., Eisenberg, J., Kleinbaum, D., Cevallos, W., Trueba, G., & Levy, K. (2015). Spatial variability of Escherichia coli in rivers of northern coastal Ecuador. Water, 7(2), 818-832.
Sarkhosh, T., Zhang, X. F., Jellison, K. L., & Jedlicka, S. S. (2019). Calcium-mediated biophysical binding of Cryptosporidium parvum oocysts to surfaces is sensitive to oocyst age. Applied and environmental microbiology, 85(17), e00816-19.
Wu, J., Long, S. C., Das, D., & Dorner, S. M. (2011). Are microbial indicators and pathogens correlated? A statistical analysis of 40 years of research. Journal of water and health, 9(2), 265-278.
Journal Articles:
No journal articles submitted with this report: View all 1 publications for this projectSupplemental Keywords:
Septic systems, rural wastewater, environmental justiceRelevant Websites:
Consortium for alabama Rural Water and Wastewater Management Exit
Dr. Elliott's Research Group Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.