Grantee Research Project Results
Final Report: One in a Billion: Living Filters for Arsenic Removal
EPA Grant Number: SV840018Title: One in a Billion: Living Filters for Arsenic Removal
Investigators: Colvin, Vicki L.
Institution: Brown University
EPA Project Officer: Aja, Hayley
Phase: II
Project Period: July 1, 2020 through June 30, 2022 (Extended to June 30, 2023)
Project Amount: $75,000
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet - Phase 2 (2020) Recipients Lists
Research Category: P3 Awards
Objective:
This project aimed to develop a living filter using engineered organisms to remove arsenic from drinking water. One goal (Aim 1) was to improve the performance of these living filters and the second goal was to explore magnetic labeling as a means to remove the organism from produced water. We achieved the first goal by engineering a novel protein based on the Arsenic regulatory protein (ArsR) already known in bacteria to include two additional ‘loops’ or short peptide sequences identified to have strong binding to arsenic oxoanions. An outcome was a bioacummulation of 1 mg/g biosorbent using the modified protein termed ArsR-Loop was overexpressed an order of magnitude larger than that measured for non-engineered bacteria; notably this biosorbent exhibited stoichometric removal of arsenic even at the very low concentrations (< 5 ppb) of concern in drinking water. Another outcome was the demonstation that this whole-cell filter was also insensitive to a range of different water chemistry conditions making its action highly predictable. We also demonstrated that more complex samples, such as apple juice and wine, could be treated with living filters to remove sub-ppm levels of arsenic. Additionally to further improve the performance of these living filters we also showed a bioengineered living sensor for Arsenic which through an optical fluorescent reporter could detect arsenic concentrations as low as 10 ppb. Finally we demonstrated effective arsenic-removal properties even when engineered microbes were grown from shelf-stable and freeze-dried samples.
For the second objective (Aim 2), we evaluated ways to remove the whole-cell sorbents from produced water. We attached magnetic particles to the microbes to enable magnetic separation of the bacteria from produced water. While this approach was successful in the laboratory, there is a cost and complexity of adding non-renewable magnetic materials to the whole-cell filtration system that is incompatable with our strategy of using renewable materials for water treatment. As an alternative we explored hollow-fiber filtration and found a tandem filter system was able to remove bacteria from water. For this measurement we applied the highly sensitive qPCR EPA methodology (EPA Method 1603) to confirm the effectiveness of this post-processing polishing of drinking water and found that this treatment produced water that had no measurable biological contamination. However, our chemical analysis of the produced water from the whole-cell filtration system revealed that some anaerobic respiration may occur under the non-stirred reactors we employed. Either further treatment by activated carbon or aeration of the living filters would be needed to remove these nuisance species.
This project developed eleven young researchers able to work on sustainable solutions to water treatment and leveraged support from corporate partners (Agilent) as well as the university’s summer programs. Dr. Yidan Bi’s thesis research centered on the application of engineered organisms as described in this project. This PhD thesis will result in three peer-reviewed publications. Zahra Ahmed received her master’s degree based on work from this project, as did two undergraduate students who wrote original honor’s research theses (Bonilla, Ayers). Another seven undergraduates were supported by this project in part with all winning competitive grants from Brown University to extend their research over the summer. Agilent corporation also supported this work and awarded the research team a loaner analytical instrument (ICP-MS) during late 2021 which made it possible to quantitate precisely the arsenic bioaccumulation properties at very low concentrations of arsenic. Collaboration with USGS in New Hampshire on well water analysis and samples extended the impact of this work by using real world examples.
Summary/Accomplishments (Outputs/Outcomes):
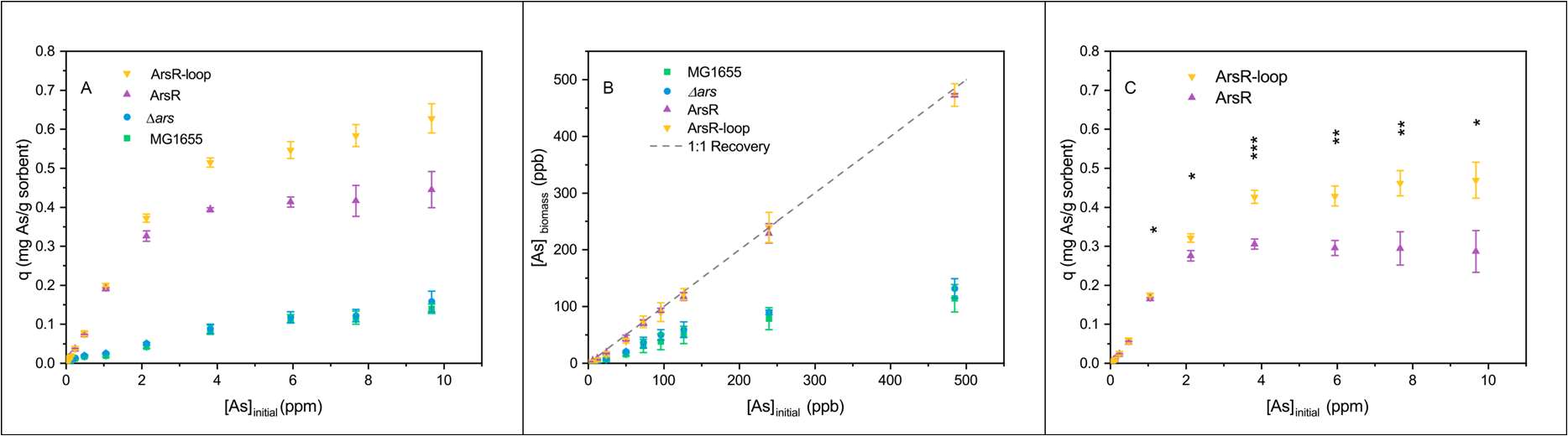
Aim 1 of this work was to improve the bioaccumulation of arsenic in engineering microbes. The overexpression of arsenic-binding proteins leads to a 3- and a 4.5-fold increase in As(III) bioaccumulation capacity in E. coli (ArsR) and E. coli (ArsR-loop), respectively (Figure 1). E. coli overexpressing ArsR has a binding capacity of around 0.4 mg/g dry bacterial weight, while bacteria overexpressing ArsR-loop has a higher capacity of 0.6 mg/g dry bacterial weight. This result is among the best arsenic treatment efficiency as compared to published work on arsenic bioaccumulation. The accumulation capacity in wild-type E. coli and E. coli (Δars) was only at ~ 0.1 mg/g dry bacterial weight. As mentioned in the above section, we hypothesize that this background accumulation level could either result from As(III) non-specifically binding to the cysteine residues in bacterial proteins or the upregulation of the E. coli chromosomal ars genes. When zooming into low-level As(III) accumulation, the engineered E. coli can volumetrically remove 500 ppb of As(III) to < 3 ppb and can completely recover from 6.25 to 125 ppb As(III) to sub-ppb levels with high fidelity (Figure 1B).
Figure 1. Comparison of arsenic accumulation of different E. coli strains and the corresponding arsenic-binding proteins. (A) Arsenic bioaccumulation capacity of different E. coli strains. 6 g/L of four types of E. coli were exposed to 0-10 ppm As(III) solutions for 16 hours. The bioaccumulation capacity was measured in the mass of arsenic per gram of dry bacterial weight. E. coli MG1655 and E. coli (Δars) were used as the background controls. The bioaccumulation of As(III) by cells overexpressing ArsR and the ArsR-loop arsenicbinding protein is considerably enhanced. (B) Volumetric arsenic recovery by different E. coli in the low arsenic concentration regime. E. coli containing arsenic-binding proteins were able to remediate water containing up to 125 ppb of As(III) to sub-ppb levels. The dashed gray line shows 1:1 arsenic removal. (C) Net As(III) bioaccumulation contributed from the arsenicbinding proteins. The arsenic binding capacity of ArsR and ArsR-loop is calculated by subtracting the bioaccumulation capacity of the E. coli (Δars) from the bioaccumulation capacity of the E. coli (ArsR) or E. coli (ArsR-loop). The arsenic binding by ArsR-loop is significantly higher than ArsR at saturated levels. ICP-MS was used to analyze samples with arsenic levels lower than 1 ppm, while ICP-OES was used for higher concentrations.
The ability to bioaccumulate trace-level arsenic is a valuable advantage of living sorbents, as capturing low-level arsenic (<10 ppb) is usually challenging for inorganic sorbents and the treatment cost grows exponentially as the arsenic MCL decreases. By subtracting the background arsenic accumulation enabled by E. coli (Δars) from the overall arsenic accumulation capacity of the engineered E. coli strains, we could obtain the net arsenic bioaccumulation capacity enhancement contributed from the arsenic-binding protein overexpression. The arsenic bioaccumulation capacity contributed from ArsR is approximately 0.3 mg/g dry bacterial weight, while arsenic binding resulting from ArsR-loop is about 0.45 mg/g dry bacterial weight. Compared to the native E. coli ArsR proteins, the extra pair of binding sites on the ArsR-loop fusion protein improved the arsenic bioaccumulation capacity by 50%. The discrepancy from our hypothesis that the extra binding sites in the ArsR-loop would double the arsenic bioaccumulation capacity can be explained by the higher binding affinity toward As(III) of ArsR sites (Kd ~ 6 𝜇M) compared to the AfArsR sites (Kd = 12±2𝜇M).
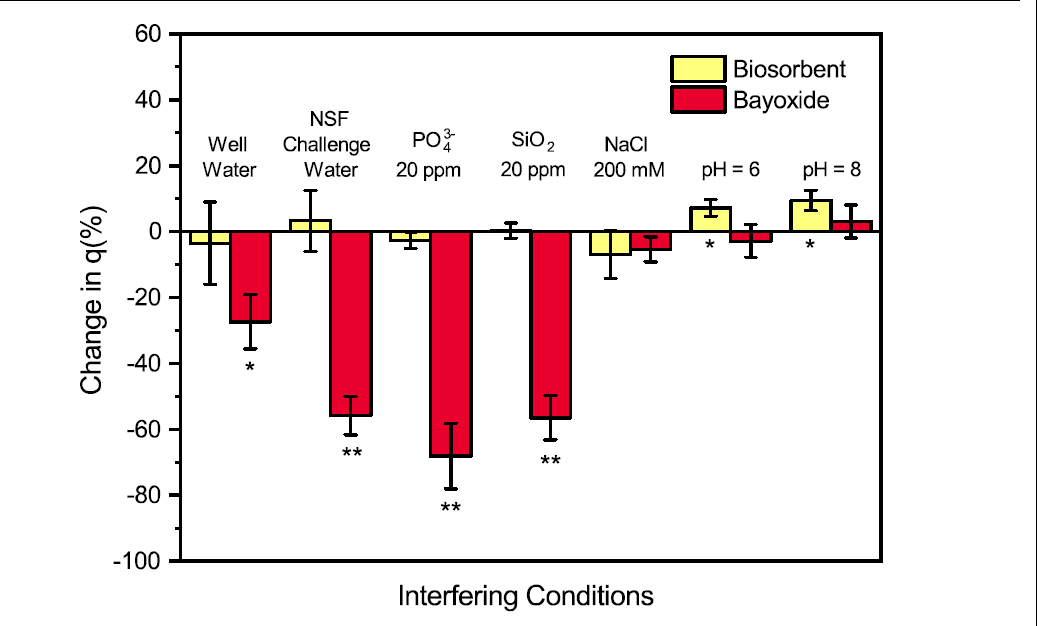
Figure 2. Impact of interfering conditions on the sorption capacities of biosorbents and Bayoxide. Sorbents were added into solutions containing 100 ppb of As(III) and the interfering species. Solutions containing PO4 3- (20 ppm, **p < 0.01) and SiO2 (20 ppm, **p < 0.01) significantly impaired the sorption performance of Bayoxide. NSF challenge water, a synthetic water matrix that mimics ion-abundant drinking water, and the well water obtained from Maine both led to significant sorption capacity decrease of Bayoxide, *p < 0.05 and **p < 0.01, respectively. In contrast, the performance of the living sorbents remains unaffected in these conditions. Operation conditions: 6g/L for biosorbents, 0.05g/L for Bayoxide. The samples were analyzed with ICP-MS.
Living sorbents can overcome the selectivity barriers faced by the inorganic sorbents and recognize As(III) among a variety of interfering ions in water (Figure 2). Although Bayoxide possesses a high capacity for arsenic remediation (25 mg As(III)/g), it can also respond to a broad range of ions, such as phosphate and silicate. As shown in Figure 2, the sorption performance of Bayoxide dropped by over 50% when 100 ppb As(III) co-exists with 20 ppm of phosphate or silicate; and its sorption capacity decreased by 50% and 30% when exposed to synthetic water (the NSF challenge water) or well water, which contains a variety of ions. The susceptibility of Bayoxide to interfering ions makes it unpredictable when treating groundwater with complicated chemistry. In contrast, the high selectivity of thiol groups on arsenic-binding proteins to As(III) prevents phosphate and silicate from occupying the As(III) binding sites, and the bioaccumulation performance remained stable across different water matrices. Besides being insensitive to common interfering species for arsenic treatment, the living sorbents also exhibit relatively stable sorption capacity in different ionic strength and a wide pH range (pH = 3-9), except that in extremely basic conditions where the bacteria cells lysed and lost the capability to carry arsenic.
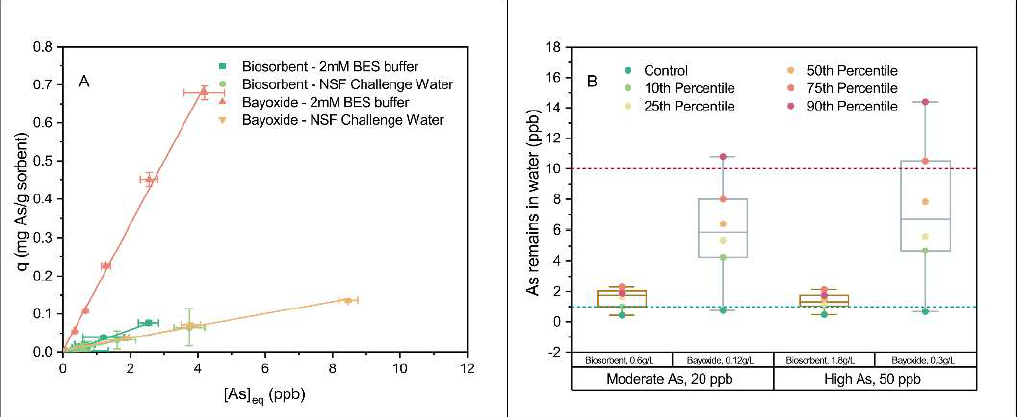
Because the biosorbents use the whole bacterial cell as a carrier of arsenic-binding proteins to achieve the optimum performance, the maximum arsenic loading capacity for them in deionized water at high arsenic levels is significantly less than the commercial inorganic Bayoxide (0.6-0.8 mg/g for biosorbents and 25 mg/g for Bayoxide, Figure S3.7). However, their stable performance makes them a robust candidate for arsenic treatment in complex matrices (Figure 3). To further evaluate the comprehensive performance of these two sorbents for arsenic removal, low-level As(III) sorption isotherms were tested in buffered pure water and the NSF challenge water. The isotherms are fitted with a linear model, and the extracted slopes of the fitted curves were used to compare the apparent binding affinities (K, equilibrium constant, L/mg) change of the sorbents under different conditions (Fig. 3.6 A and Table 3.3). Although the apparent binding affinity of living sorbents is 5-fold lower than the apparent binding affinity of Bayoxide in ideal conditions because of the bacterial mass burden, in interfered conditions, the K value of the biosorbents remains consistent and emulates the K value of Bayoxide. To treat complicated water matrices such as NSF challenge water, the required biosorbents dose remains at a comparable level while the required Bayoxide dose increases significantly. Second and most importantly, the water chemistry variation due to location and season change poses less impact on the arsenic treatment performance of biosorbents, and therefore, they will offer more reliable treatment results compared to Bayoxide. For example, if we aim to reduce a 20 ppb As(III) (considered as moderate contamination compared to the EPA regulation) to 1 ppb from drinking water with different matrix intensity with the minimum sorbent dose calculated from pure water treatment (Figure 3B), arsenic levels after biosorbents treatment exhibited a narrow distribution within 3 ppb, while the water treated with Bayoxide has broad a range of treatment result and could exceed the 10 ppb regulation level. Similarly, when treating high-level As(III) (50 ppb), the treatment result of biosorbents stays in a low and narrow range, while the arsenic remaining in Bayoxide-treated water spreads broadly. This result indicates that engineered biosorbents could serve as universal sorbents for treating different drinking on different sites and seasons with reliable outcomes.
Figure 3. Low-level isotherm fitting and the arsenic remediation predictability of biosorbents and Bayoxide in different water matrices.(A) Arsenic low-level isotherms for biosorbents and Bayoxide in 2 mM BES buffers and NSF challenge waters. The slope (equilibrium constant) of Bayoxide isotherm dropped significantly in the presence of interfering species, while the slope of biosorbents remained unchanged. (B) Arsenic remediation performance of biosorbents and Bayoxide treating moderate level (20 ppb) and high level (50 ppb) arsenic in water with 10 - 90th percentile synthetic water. The synthetic water is made according to the occurrence of co-occurring parameters in national groundwater arsenic database, which artificially arranged arsenic co-occurrence water quality from low to high levels and selected 10, 25, 50, 75, and 90th percentile cut-off concentrations.
Aim 2 of this project centered on the removal of the biosorbent from the produced water. The concentration of engineered E. coli in arsenic-bioaccumulation post-treatment permeate was quantified to evaluate the performance of hollow fiber membrane facilitated bacterial removal. Protein overexpression usually results in ribosome damage in engineered bacteria and the loss of reproducing ability on nutritious agar. Therefore, resting protein laden bacteria quantification with membrane filtration in cope with plate enumeration methods are not trustworthy and often leads to false negative results. Here we apply real-time PCR for E. coli quantification, and in the meantime analyze the wildtype E. coli copy numbers with both plate culture and qPCR methods. In this approach, the drawback of reporting false negative results of plate culture method is avoided, the disadvantage of qPCR not discriminating living and dead bacteria could be corrected. According to our results, the hollow fiber membrane cartridge is effect to filter treated water with low sorbent dose, while high sorbent dose will require more units in parallel to achieve satisfying removal rate. Adding an extra HFM filter would result in 3 to 5-fold of genetic copies in treated water, however, this number is not necessary corresponding to the living bacteria removal efficiency. Resting bacteria in water without nutrient would lead to wildtype death according to the qPCR results, while engineered sorbents are more resistant to nutrient deficient environment. However, our chemical analysis of the produced water from the whole-cell filtration system revealed that some anaerobic respiration may occur under the non-stirred reactors we employed. Either further treatment by activated carbon or aeration of the living filters would be needed to remove these nuisance species.
| Permeated Samples | E.coli copies via qPCR (CFU/50mL) | WEildtype E.coli m-TEC methods (CFU/50mL) | Wildtype E.coli copies via qPCR (CFU/50m>) | NH4+ (ppm) | NO3- (ppb) | PO43- (ppm) |
|---|---|---|---|---|---|---|
| [As] = 5 ppb Stock solution | ND | ND | - | <LOQ | 164.5 | ND |
| [As] = 50ppb Stuck Solution | ND | ND | - | <LOQ | 127.7 | ND |
| 0125 g bacteria/L 1HFM | 220± 1.5 | ND | - | 0.114±0.009 | 242.1±18.2 | 0.076±0.010 |
| 0.125 g freshly cultured bacteria/L 2HFM | 42 ± 39 | - | - | 0.411±0.048 | 260.2±14.5 | 0.134±0.019 |
| 1.56 g freshly cultured bacteria/L 1HFM | 1.0X104 ± 3.4X102 | Detected | 1.2X105 | 4.977±0.286 | 264.1±30.7 | 1.683±0.161 |
| 1.56 g freshly cultured bacteria/L 2HFM | 3.7X102 | ND | 4.3X104 | NA | NA | NA |
Besides passively confining living sorbents behind the membrane filters, active magnetic mediated bacterial isolation was also explored. The E. coli cells were magnetically labeled with magnetic nanoparticles via a known conjugation chemistry. Iron oxide nanoparticles coated with amine groups were chosen because in neutral pH positively charged amine groups electrostatically attract negatively charged bacterial cell walls, which facilitates the progress of conjugation. Optical density was measured for magnetic bacterial removal efficiency calculation. Although the electrostatic attraction between cells and nanoparticles could partially remove the bacteria from the system, the removal efficiency increased by 50% as the concentration of coupling agent increased. While these results were favorable, the application of iron oxide nanoparticles was not aligned with the project’s focus on renewable sorbents for arsenic removal. Future work will explore reversible attachment and detachment strategies to permit recover and reuse of magnetic materials for this application.
Quality Assurance
Table 2 describes the analytical instrument used during the report period and their calibration procedure and frequency. The arsenic level was analyzed with ICP-AES and ICP-MS, while the E. coli quantification was achieved by membrane filtration. The quality assurance methods for these two measurements are discussed below in detail.
| Instrument | Purpose and application | Calibration Procedure | Frequency |
|---|---|---|---|
| Balance (METTLER TOLEDO) | Use to measure mass of chemicals. ALso used is calibration standards preparation and pipette calibrations | Calibrated by service technician during annual maintenance | Yearly |
| Automated pipettes (Eppendorf) | Use to adjust the pH of prepared solutions | Calibrated by service technician during annual maintenance | Yearly |
| pH meter (METTLER TOLEDO) | Use to adjust the pH of prepared solutions | Calibrated using standard buffers | Every session |
| UV-vis spectrophotometer (Molecular Device) | Uesd to determin the bacterial concentration in terms of g/L | Calibrated using background solvent | Every session |
| ICP Automic Emission Spectrometer (ICP-AES)(ThermoFisher) | Elemental analysis. THe quantification of the amount of arsenmic remediated by biosorbents | Calibarated by introducing a series of standards over expected range of quantitation | Every session |
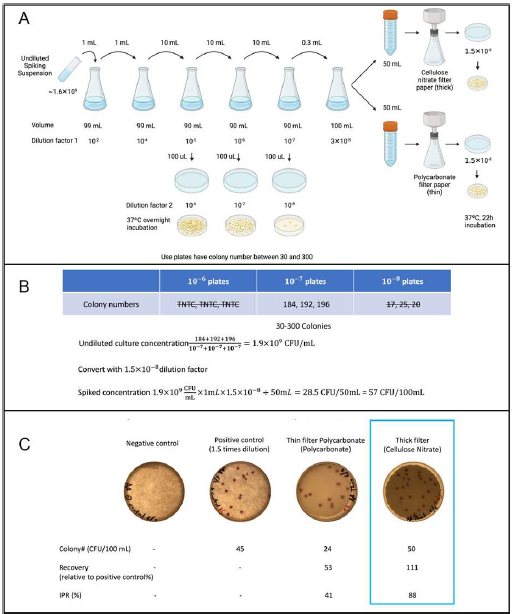
Before applying the membrane filtration method to quantify E. coli colony forming units in the permeate obtained from prototype 3, the initial precision and recovery (IPR) analysis were conducted according to EPA method 1603. Figure 5 shows the experimental procedures in detail. We also compared the performance of two recommended membranes for bacterial retention and selected the one with the higher IPR. For E. coli quantification in each permeate batch, negative control and positive control were analyzed along with the samples. For ICP-AES and ICP-MS analysis, we referred to Method 200.7 and Method 200.8 to assure the data quality for arsenic measurement. We confirmed the linear dynamic range (LDR) and the method detection limits (MDL) of the instrument could fulfill our application purposes. The MDL were lower than eight ppb for ICP-AES and were in sub-ppb level for ICP-MS. We periodically verified if the instrument performance was acceptable with the quality control samples, and if a problem occurred, we would identify and correct it before proceeding with sample analysis. In each batch of sample analysis, the laboratory reagent blank (LRB) and the laboratory fortified matrix (LFM) were measured immediately following the matrix-matched calibration standards, after every ten samples, and by the end of the sample runs. The LRB should not exceed the MDL, and the LFM recovery range should be within 80-120%. In addition, internal standards (ISTD) were used to monitor throughout the sample analysis to evaluate the impact of the matrix effect. If the ISTD stability went beyond 80-120%, samples had to be diluted and reanalyzed.
Figure 4. IPR procedures of the EPA Method 1603. (A) experimental procedure. (B) Calculation procedure for spiked bacterial concentration. (C) Initial precision.
Conclusions:
In conclusion, the engineered E. coli harboring fusion protein that carry more arsenicbinding motifs can achieve higher As(III) bioaccumulation capacity. The engineered microbes can behave as living sorbents and selectively bioaccumulate As(III) in water containing interfering ions such as phosphate and dissolved silica. In heavy water matrices, compared to inorganic sorbents, the living sorbents performance is predictable, and the treatment results retain below the 10 ppb drinking water limit. The sorbent dose required is estimated to be comparable to inorganic sorbents under complicate water conditions. Moreover, the living sorbents maintain their selectivity toward As(III) in more complicated beverage matrices, and they can be freeze-dried for sorbent storage and transportation. In future work, a fluorescence protein expression mechanism will be integrated to the living sorbents for reporting the saturation of the biosorbents based on real-time arsenic level in treated water.
We constructed renewable living for selective low-level As(III) bioaccumulation. By labeling the biosorbents with magnetic nanoparticles and iteratively designed prototypes, we explored the potential to apply them to point-of-use drinking water treatment. This work meets the need for developing sustainable and selective materials that can remove trace levels of arsenic from drinking water. It also fulfills the P3 program's goal of improving our planet, the health of people, and the prosperity of our nation. The living sorbent design provides an opportunity to provide As(III) drinking water to rural households and small communities inaccessible to expensive and energy-consuming arsenic treatment technologies.
Journal Articles:
No journal articles submitted with this report: View all 2 publications for this projectProgress and Final Reports:
Original AbstractP3 Phase I:
Magnetic Nanocomposites for Water Remediation | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.