Grantee Research Project Results
2020 Progress Report: Green and Sustainable Water Purification Membranes
EPA Grant Number: SU839961Title: Green and Sustainable Water Purification Membranes
Investigators: Poler, Jordan C , Sahu, Abhispa , Rollins, Rachel
Current Investigators: Poler, Jordan C , Sahu, Abhispa , Rollins, Rachel , Jones, Anna , Oliver, Sydney , Reid, Christopher
Institution: University of North Carolina at Charlotte
EPA Project Officer: Page, Angela
Phase: I
Project Period: October 1, 2019 through September 30, 2020 (Extended to September 30, 2021)
Project Period Covered by this Report: October 1, 2019 through September 30,2020
Project Amount: $24,867
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2019) RFA Text | Recipients Lists
Research Category: Urban Air Toxics , Heavy Metal Contamination of Soil/Water , P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
This project proposes to develop a green, sustainable and eco-friendly membrane system that removes chemicals that persist following traditional water treatment. To achieve this, a cellulose nanoscale anion-exchange resin will be developed. Since cellulose nanofibers are abundant and inexpensive, this proposal, if successful, will provide affordable water purification solutions to rural communities that have high concentrations of contaminants found in water systems.
Progress Summary:
Our current work status has been slowed significantly by the Coronavirus pandemic. Only one project participant and the PI have been able to do in-lab work since the university shut down in March 2020. Fortunately the first five months of the project were very productive. The proof-of-principle results for a novel green and sustainable water purification membrane, are summarized here and described below. (1) microcrystalline cellulose was oxidized to nanofibers of cellouronic acid. This purified material was functionalized with a vinylic anchor point using EDC catalyzed amine linkage. The purified product was then functionalized with short brush anion exchange polymer strands. (2) the subsequent materials were fully characterized by all major spectroscopic and microscopic methods. (3) the materials were tested for purification performance using surrogate compounds.
Our university is scheduled to reopen on 1/20/2021. Most of the undergraduate participants will restart their laboratory work as soon as the recent surge in COVID cases lowers to a safe positivity rate.
- All key personnel completed their work on this project. Due to the Corona virus pandemic we had to shut down most of the in-lab experiments and all of our field work and travel from March 2020 onward. The participants on this project continued their research virtually and some of the project was shifted to literature research and computational research.
- The Graduate student stipend and participant stipends were expended from the budget as expected. Most of the budget on this project was for travel. Since the pandemic cancelled the EXPO and all of the conferences we planned to attend, and we were not allowed to travel to do the field work, most of the funds allocated for travel have not been expended. UNC Charlotte still has a travel ban for funded projects and we have not been informed of when that may be lifted. Since it is unlikely to be lifted through May 2021, we will be requesting a budget change to reallocate the funds from the travel line of the budget.
- All hazardous waste from this project will be collected and disposed of in accordance with Federal guidelines. Our laboratories are inspected by our Office of Environmental Health and Safety on an annual basis. All students and participants received extensive Safety and Chemical Hygiene training by the PI, departmental safety staff and online modules. All experiments have been carried out with the high quality that is typical of our lab. Our data management plan includes several safeguards to ensure high quality and ethical reporting of results. During the course of this project we have synthesized and characterized various polymers and nanomaterials. Experimental results are recorded in laboratory notebooks as well by PC’s attached to analytical instruments. All data files generated during characterization of samples and processes are saved on the PC’s. These files include mostly proprietary data formats. However, most of these applications can export data in common .xls or .csv formats.
Samples prepared for AFM, SEM, and TEM analysis are kept in small boxes, which are uniquely labeled with a notebook number. These samples are kept in cabinets away from common airborne contamination. Following analysis of these samples, all images and measurement data from AFM, SEM, and TEM studies will be saved. These are typically high resolution .tif files. Some of the applications can export a .jpg or .pdf formats.
All laboratory notebooks are maintained according to standard scientific practice. Notebooks are written in permanent ink and written as a contiguous record of all procedures, observations, hypotheses and conclusions. All entries are signed and dated by the researcher each lab period. All sample and file names are associated with where the data was taken and on which instrument and in what drive and folder it is stored.
All physical samples and solutions or dispersions made from them are labeled with a descriptive name and a notebook number. Every sample has its own unique notebook number. A standard sample number is; JCP-25D-16A which signifies the PI’s group, the notebook in a series, the page number and the sample on that page respectively.
All spectroscopic, microscopic and analytical data files are named with a unique notebook number. All AFM, SEM, and TEM image files and analyses are named with a unique notebook number (e.g., JCP-35D-124A3.tif).
- Results to date are described here. In this annual report we describe our results on the functionalized cellulose. As mentioned above, due to the pandemic, we also included our work on functionalized fluorographene. Our results on that part of the project are described in the manuscript submitted here.
The hydrodynamic diameter of pristine cellulose nanofibers measured using dynamic light scattering (DLS) zetasizer was in the range of ~250 nm as shown in Figure 1. After purification, the successful debundling of -COO-Na+ modified cellulose nanofibers were confirmed using DLS with a hydrodynamic diameter of 24 nm. The oxidation of cellulose was confirmed using Fourier transform Infrared Spectroscopy with a strong peak around 1575.85 cm-1 due to presence of -COO-Na+ group. Zeta potential of cellouronic acid solution was measured to be -28 mV. The amide reaction and functionalization of allyl amine hydrochloride was confirmed using Fourier Transform Infrared Spectroscopy (FTIR) where we observed shifting of C=O stretch at 1640.01 cm-1, appearance of N-H in plane vibrations at 1534.6 cm-1 and C-N stretch at 1429.6 cm-1. Zeta potential of amide modified cellulose nanofibers were measured to be -19 mV. To confirm allyl amine hydrochloride is covalent attached to cellulosic scaffold, we filtered away the cellulose material and found no peaks in the filtrate, which confirms there is no presence of any residual unreacted components. Zeta potential of poly(vbTMAC) functionalized cellulose nanofibers was measured to be +17.2 mV which confirmed the covalent attachment of polyelectrolyte to cellulose nanofibers. Adsorption loading, qe, measured from incubation experiment was measured to be 12.6 mg-C NaFL/g polymer functionalized cellulose. Preliminary analysis of adsorption isotherm was performed to understand the adsorption behavior (Figure 2). Ongoing studies include complete adsorption isotherm analysis and kinetics studies. We would also target the emerging contaminants enlisted by US EPA to show the efficacy of functionalized cellulose nanofibers to remove contaminants in a fast and high capacity manner.
Table 1. Hydrodynamic diameter of cellulosic fibrils. The cellulose nanofibers were dispersed in water and sonicated at 45 W cm^-2 for 3 h. The dispersion was centrifuged at 3000 g at 20 °C for 20 minutes. Transparent aqueous solution of cellulose fibrils was collected and measured for DLS analysis.
| Sample Name: | cellulose supermatant 20 | ||||
|---|---|---|---|---|---|
| SOP Name: | size cellulose sop | ||||
| File Name: | Size cellulose supermatant 1_9_2020... | Dispersant Name: | Water | ||
| Record Number: | 20 | Dispersant RI: | 0.330 | ||
| Record Material RI | 1.47 | Viscosity (cP): | 0.8872 | ||
| Material Absorbtion: | 0.000 | Measurement Date and Time: | Wednesday, January 08, 2020 7:00... | ||
| Temperature (oC): | 25.0 | Duration Used (s): | 120 | ||
| Count Rate (kcps): | 38.8 | Measurement Position (mm): | 4.65 | ||
| Cell Description: | Low volume disposable sizing cuv... | Attenuator: | 11 | ||
| Size (d.nm) | % Number | Width (d.nm) | |||
| Z-Average (d.nm): | 488.9 | Peak 1: | 258.6 | 100.0 | 46.74 |
| Pdl: | 0.476 | Peak 2: | 0.000 | 0.0 | 0.000 |
| Intercept: | 0.607 | Peak 3: | 0.000 | 0.0 | 0.000 |
| Result quality: | Refer to quality report | ||||
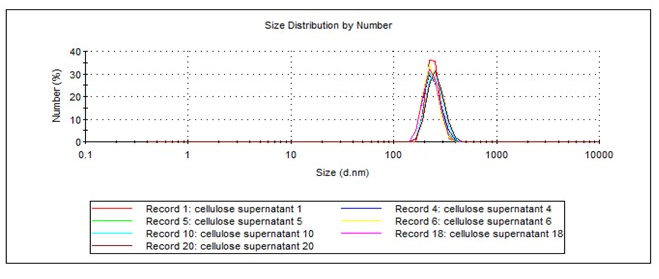 | |||||
| Figure 1 | |||||
Future Activities:
In the next reporting period, we plan to complete the milestones laid out in our No-Cost-Extension plan. As described above, we have prof of principle that our objective to functionalize nanofibril cellulous with anion exchange polymers was a success. We have identified a few synthetic challenges and we plan to solve these issues in the next reporting period. By the end of the reporting period, we will have optimized the water purification materials synthesis and fully characterized the optimized materials. All objective #4, Point-of-use water purifiers, is incomplete and will be completed in the next reporting period. We started completing Objective #3, Outreach teaching and training, during the last reporting period. Unfortunately, this effort was shut down due to the pandemic. It is not clear when we will be allowed to start the activities in this objective again.
Fortunately, the Ph.D. student on this project was allowed to continue in the lab through the pandemic. Due to her efforts, we have created a novel water purification material and these results are being submitted to ACS Nano shortly. I have included a draft of this manuscript with this annual report. The results of this manuscript “Aqueous defluorination and covalent grafting of fluorographite with anion exchange resins for water purification” are described therein.
Journal Articles:
No journal articles submitted with this report: View all 3 publications for this projectSupplemental Keywords:
Cellulose, Water Purification, Membrane, Ion Exchange, NanomaterialRelevant Websites:
Progress and Final Reports:
Original AbstractP3 Phase II:
Restoring Public Confidence in Drinking Water Safety - Education, Engagement, and New Sustainable Technology | 2022 Progress Report | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
