Grantee Research Project Results
Final Report: Iron-TAML/peroxide Cyanotoxin Degradation
EPA Grant Number: SU839461Title: Iron-TAML/peroxide Cyanotoxin Degradation
Investigators: Collins, Terrence J. , Ryabov, Aleksandr D , Somasundar, Yogesh , Nagarajan, Anantha , Denardo, Matthew
Institution: Carnegie Mellon University
EPA Project Officer: Page, Angela
Phase: I
Project Period: March 1, 2019 through February 29, 2020
Project Amount: $15,000
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2018) RFA Text | Recipients Lists
Research Category: P3 Challenge Area - Chemical Safety , P3 Challenge Area - Safe and Sustainable Water Resources , P3 Challenge Area - Air Quality , P3 Challenge Area - Sustainable and Healthy Communities , P3 Awards
Objective:
Cyanobacterial blooms are a troubling and an increasingly frequent1 occurrence. In humans, birds, and mammals (including cattle), exposure to the cyanotoxins produced in blooms can cause liver, digestive, and neurological diseases.2 Multiple classes of cyanotoxins have been identified; the most prominent family is the microcystin group of toxins, which are cyclic heptapeptides. Highly hepatotoxic,3,4 studies have also suggested chronic, low-level exposure to microcystins through drinking water is correlated with increased rates of cancer in humans.4 Additional research, compiled in a recent review by Svirčev and coworkers3 indicated microcystins may also be neurotoxic--damaging to reproductive health, and genotoxic. Bioaccumulative in some species, including garden lettuce5, microcystin-LR is possibly the most common member of this family of toxins and is known to be among the most toxic.6 Although the prevalence of microcystins in U.S. waters has not been comprehensively studied, with state monitoring varying widely, the US Geological Survey determined that microcystin was present in 32% of samples collected in 2007 from over a thousand US lakes, ponds, and waterways.7 This survey, among others, shows particularly high microcystin prevalence in Midwestern water samples. Lake Erie, the source of drinking water for eleven million people,8 suffered a record-setting algal bloom in 2011 that resulted in microcystin concentrations of up to 8.7 μg/L.9 Such expansive blooms are expected to become more common in the lake.9 In Ohio generally, 44% of drinking water source samples exceed the EPA’s children-under-six heath advisory level of 0.3 μg/L,10 and 2014, microcystin contamination of finished municipal water caused Toledo, Ohio officials to issue a three-day Do-Not-Drink/Do-Not-Boil order affecting 500,000 people.
Nationwide, lakes and ponds are invaluable sources of drinking water, environmentally important fish and wildlife habitats, and a driver of tourism and economic activity. A sustainable, safe method of microcystin-LR detoxification is therefore highly desirable, as it would support the health of shoreline communities, encourage continued tourism and fishing along lakeshores, and provide relief to fish, bird and mammal populations along and in affected natural waters.This Phase I project aimed to investigate just such a solution: the viability and effectiveness of bio-inspired NewTAML/H2O2 catalysis for oxidative degradation of microcystin-LR. TAML (tetraamido macrocyclic ligand) activators are the result of an iterative design protocol developed, starting in 1980,11 by Dr. Terrence J Collins, currently director of Carnegie Mellon’s Institute for Green Science. The first TAML activator to emerge from this process was invented in 1995.12 Iron-TAML activators are designed to mimic oxidizing enzymes found in living cells. They are superior function small molecule mimics of the oxidizing enzymes like peroxidase and cytochrome P450 at less than 1% the weight of these enzymes, making them vastly easier to synthesize, transport and deploy. NewTAML catalysts were invented as a solution for decomposition pathways that limited the performance of the original TAML catalysts. NewTAML activators surpass TAML activators in technical performance by at least a factor of ten, and we are certain that this advantage gap will expand, reducing manufacturing and operational costs, and improving environmental performances by reducing the already incredibly low concentration of catalyst required to achieve the rapid breakdown of troubling water pollutants. This leap in performance tied to the NewTAML innovation has been published in ACS Catalysis.1
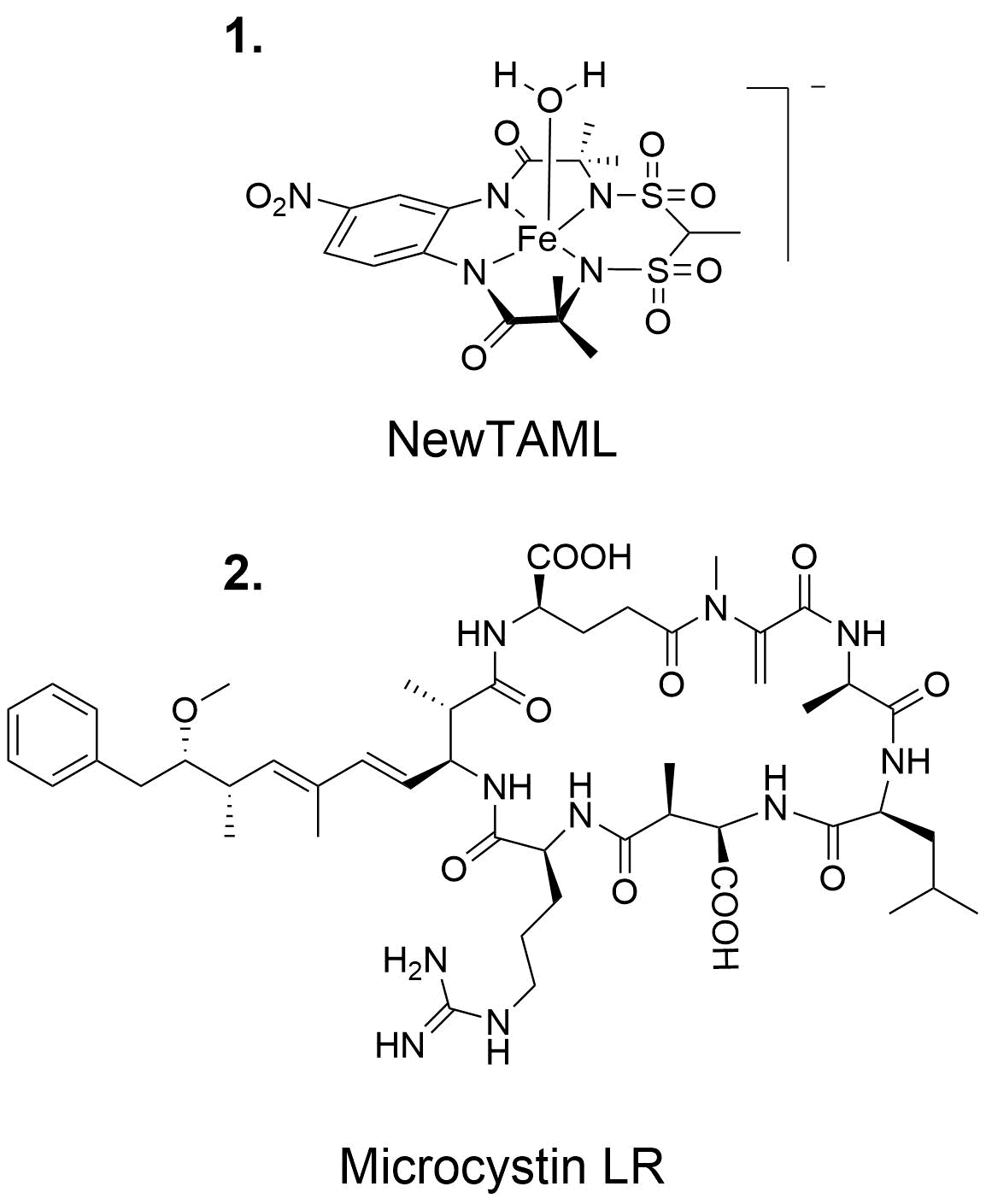
Figure 1. Structure of the NewTAML catalyst used in Phase I experiements (1) and microsystin-LR (2), the cyantoxin examined.
Summary/Accomplishments (Outputs/Outcomes):
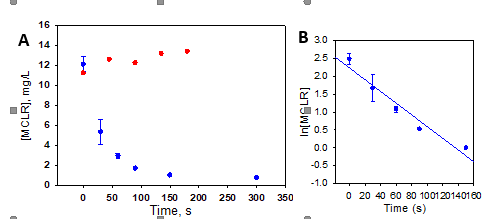
Outputs and outcomes The fundamental goal of this project was to evaluate the oxidation of microcystin-LR (MCLR, 2) under NewTAML (1) catalysis and to determine the values of chemical kinetic rate constants that govern that oxidation. Taken together, this information would help indicate whether 1 could be an effective treatment method for microcystin contamination of recreational waters and ultimately drinking water sources. To this end a LCMS method for quantification of 2 was developed, and an extensive series of reactions was run in order to evaluate the chemical kinetics of oxidation. (Methods are detailed below in ‘Methods and quality assurance.’) Experiments showed that oxidation of 2 is rapid and extensive under experimental conditions, as shown in Figure 2A. Linearization of the data (Figure 2B) reveals that the reaction is following pseudo-first kinetics. The slope of the linearization provides the information needed to calculate the rate constant governing the reaction between the activated TAML catalyst and 2, which was found to be (1.6±0.2) × 105 M-1 s-1, indicating that this reaction is fast. This value was confirmed by further experiments in which the concentration of 2 was systematically varied. These experiments, not presented here due to space constraints, gave a rate constant of (1.5±0.4)×105 M-1 s-1, representing very close agreement to the results of the experiment above. Furthermore, other experiments confirmed that the rate of reaction is linearly dependent on catalyst concentration, confirming that this reaction follows typical TAML kinetics (as described in previous scientific publications) under high concentrations of peroxide.
Figure 2. (A) Disappearance of MCLR signal over time under 1/H2O2 treatment (blue) and under control conditions (H2O2 only, red); (B) linearization of NewTAML treatment data. Reactions were conducted at pH 7, 25⁰C, 10 mM H2O2, and 100 nM 1. Control data not yet collected in triplicate.
Control experiments using only peroxide showed no measurable oxidation of 2, confirming that the results were due to NewTAML catalysis and not another chemical process. In experiments with catalyst, varying the concentration of H2O2 from 1 mM to 10 mM revealed that reaction rate is independent of peroxide concentration over the examined range. Further experiments will be conducted to determine at what concentrations of peroxide the reaction slows down. Removal of MCLR is extensive but not complete: 0.431±0.007 mg/L remained (3.6%) after 24 hours.
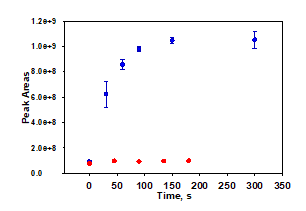
Figure 3. Formation of MCLR+16 oxidation product under catalysis by 1/H2O2 (blue), and lack of formation of the product using only H2O2 (red). The formation of this oxidation product kinetically mirrors the oxidation shown in Figure 2. Reactions were conducted at pH 7, 25⁰C, 10 mM H2O2, and 100 nM 1. Control data not yet collected in triplicate
Under treatment by 1/H2O2, 2 is quickly converted into an oxidation product which has a mass to charge signal of MCLR+16. This formation is illustrated in Figure 3. Without an analytical standard the concentration of this product cannot be assessed. However, the highest observed peak area, at 300 seconds, is 88.8% of the peak area observed for MCLR at time zero in the same experiment, suggesting that a majority of the original MCLR is converted into this initial oxidation product. That the formation of this MCLR+16 product is governed by rapid rate constant measured as (3.5 ± 0.3) ×105 M-1 s-1, again suggesting that almost all 2 is converted into this product. The MCLR+16 product persisted at 24 hours when its apparent concentration had slightly increased from the measurement taken five minutes after reaction initiation. The product is resistant but not completely impervious to further oxidation: spiking the 24 hour old reaction at with fresh 100 nM 1 and 20 mM H2O2 produced a 20.9±2.9% decrease in the observed peak area over eleven minutes of reaction. Additionally, the remaining MCLR decreased from 0.431±0.007 mg/L to 0.363±0.015 mg/L (a decrease of 15.7±2.6%) in the same amount of time. Future studies will further investigate the slow oxidation of this MCLR+16 product.
Methods and quality assurance
Instrumentation LCMS measurements were carried out in Carnegie Mellon University’s Center for Molecular Analysis using a Thermo Scientific Exactive Plus EMR Orbitrap Mass Spectrometer and Thermo Scientific Vanquish Flex Binary UHPLC using a 100 x 3.0 mm Phenomenex Kinetex 5 μM EVO C18 100 Å column. UV-Vis measurements were conducted on an Agilent 8453 diode array spectrophotometer. The pH of buffers was determined using a Fisherbrand accumet AB15 pH meter calibrated to pH 4, 7 and 10.
Reagent preparation Microcystin LR was obtained from Sigma Aldrich (as a 10 μg/mL analytical standard in MeOH) and from Enzo Scientific (as lyophilized powder). Purity and identity of the compound was determined via ESI-MS. Solutions were stored under refrigeration.The concentration of solutions prepared from lyophilized powder was compared to the Sigma Aldrich analytical standard using LCMS methods described below in order to quantify the concentration of MCLR in these stocks and ensure that the solutions had not degraded while in storage. All solvents (Fisher Scientific) were HPLC grade; other reagents were ACS reagent grade or better and were purchased from Fisher Scientific or Sigma Aldrich. All were used as received. NewTAML catalyst had been synthesized according to previously published methods.1
General Reaction Procedure
Every other day, the hydrogen peroxide concentration of H2O2 stock solution was checked using UV-Vis spectroscopy (ε=71.6 cm-1M-1 at 230 nm). Reaction buffer was re-prepared twice a week by adjusting the pH of a 10 mM ammonium carbonate solution to 7.0 using dilute formic acid. Buffer pH was confirmed daily. Buffer was stored refrigerated and warmed to 25 ⁰C in a water bath before use. Reactions were conducted in a 24.5-25.5 ⁰C room environment. The microcystin LR stock solution was combined with previously described pH 7 buffer and NewTAML stock in a 2 mL HPLC vial and mixed for ten seconds via mini-vortex machine. A single aliquot (100 or 150 μL) was then removed and placed in a 300 μL HPLC glass-insert vial pre-loaded with 10 or 15 μL10 mg/mL bovine liver catalase dissolved in the same buffer used for the reaction. The aliquot was mixed with the catalase via mini-vortex machine for five to ten seconds. Next, H2O2 stock solution, sufficiently concentrated so as to be <20 μL of addition, was added to the reaction vessel to a final concentration of 10 mM, a timer was started, and the reaction vessel was again mixed via vortexing. Then several further aliquots were collected, generally taken every 45 seconds and quenched as previously described—samples containing peroxide produced visible bubbles upon vortexing. Reactions used to determine kinetic rate constants were performed in triplicate.
LCMS Parameters
The elution gradient consisted of two mobile phases: a pH 5.5 10 mM acetate buffer (A) prepared weekly from ammonium acetate/acetic acid and MeOH (B). The flow rate was 0.3 mL/min with the following linear gradient: 10% B, RT 0.5 min; 80% B,1.5 min; 90% B, 2 min to 3.5 min; 10% B 5 min to 6.5 min. Injection volume was 10 μL. Column temperature was 30 ⁰C. At the HESI source, the sheath gas flow was 251 units, the auxillary gas flow was 5 units (138 ⁰C), and the sweep gas flow was 1 unit. Spray voltage was 4.00 kV with a capillary temperature of 250 ⁰C.
Microcystin LR Quantification
MCLR was quantified via creation of a calibration curve based on peak areas for m/z +992.00-1001.00 and +495.00-500.00, as measured at the elution time of at approximately 4.21 minutes. Peak areas were calculated using Thermo Scientific Xcalibur software (version 4.1.31.9). After initial creation of calibration curve, measured in triplicate, the slope obtained was used to calculate further concentrations. In addition, a diluted standard prepared from the Sigma Aldrich analytical standard was always run.
Evaluation of technical effectiveness, economic feasibility, and benefit to humans and the environment
As discussed in the research objectives above, Microcystin-LR is both prevalent and dangerous. In towns surrounding recreational waters, it damages tourism (including the valuable sport fishing industry). Additionally, MCLR and other cyanotoxins can kill pets, cattle, and wildlife that consume or live in affected waters. In drinking water sources, microcystins pose a serious threat to human health. Thus, there is a need for a safe and effective method of removal of microcystins (and other cyanotoxins) from natural waters and, ultimately, drinking water sources. Current treatment methods include chlorination, which can produce undesirable and potentially dangerous organochlorine byproducts, and ozone treatment, which is costly to implement and carry out in modestly sized water treatment plants. As TAML catalysis is a method already proven to remediate a diverse array of micropollutants present in urban wastewaters, it is a promising technology for this application as well. TAML and NewTAML catalysts are very stable in dried form, and are effective at the low concentrations that are practical for water treatment. Overall, we project that the cost of water treatment using TAML/H2O2 catalysis will be significantly less than the cost of ozone-based treatment methods.
As described above, NewTAML catalyst (1) rapidly oxidizes MCLR (2). In a sufficient excess of peroxide, the observed rate constant, (1.6±0.2) × 105 M-1 s-1 (pH 7, 25ºC), is comparable to or slightly faster than the literature value reported for ozone driven oxidation of 2 (5.22x104 M-ls-1, pH 7, 30ºC).2 However, and critically, the toxicity of the +16 oxidation product is unknown. It would be preferable to mineralize or severely degrade 2. Therefore, although these initial results are encouraging, they are preliminary, and we cannot yet determine whether NewTAML catalysis is a viable method for mitigation of MCLR induced toxicity. Future studies, as outlined below in ‘Future Activities,’ should allow for a more informed conclusion on the suitability of this method.
Conclusions:
TAML catalysts generally form a variety of oxidative products when presented with a substrate and are sometimes to mineralize their targets. Thus, the behavior of the TAML catalytic system in this case (the formation of one primary oxidation product) in the case of Microcystin-LR treatment is unusual and requires further analysis. First, it is critical to identify the structure of the primary oxidation product. This will provide crucial insight into the action of TAML catalysts in this interesting case. Next, phosphatase inhibition assays, which could provide a preliminary assessment of oxidation product toxicity, should be conducted. Methods for conducting such assays have been previously reported in scientific literature. Together, these two actions will provide insight into the identity and characteristics of the MCLR+16 species.
The studies conducted to date have examined significantly higher (ppm vs. ppb) concentrations of MCLR than prescribed by US EPA drinking water guidelines. This was due to the difficulty of experimental design and toxin quantification at lower concentrations. Certain kinetic considerations (i.e., second order catalyst degradation) may become considerably more important under the drinking-water-relevant, ultra-dilute substrate conditions that have proved are difficult to study via LCMS. In order to examine reaction characteristics at ppb concentrations, samples will most likely need to be concentrated or extracted before testing (as in EPA Method 544,3 among others4,5). Unfortunately, both methods of enhancing signal would be difficult or even impossible to use with current reaction volume and aliquot size and would be very cost prohibitive at scales above 10-20 mL reaction volume.
A possible, although non-ideal, method of studying whether TAML-catalyzed oxidative destruction occurs at all at low concentrations would be to employ commercially microcystin-sensitive test strips, which function similarly to ELISA tests but require no special equipment. Such strips are commercially available,6 but could only provide only basic proof-of-concept rather than conclusive results. HPLC quantification (sometimes coupled with colorimetric tests coupled with the colors are developed in a similar matter to ELISA tests or more directly via alkaline phosphatase inhibition; alternatively paired with solid phase extraction7) have also been reported in literature8 and represent a potentially more cost-efficient method of concentration monitoring compared to HPLC-MS.
To better understand the applicability of NewTAML catalysis for microcystin treatment, it is also important to study the degradation of MCLR in natural waters, which was not feasible during this study period. In natural waters, which are would need to be treated to improve public and environmental health, the toxin would never be found in isolation; it is usually present not only alongside other microcystins but also along with a large variety of other organic compounds. Therefore, it would be ideal to study natural waters contaminated with microcystins. It may be easier to spike natural water samples with MCLR or other microcystins, as the seasonality and fundamentally temporal nature of algal blooms means fresh samples from natural bodies of water are most available for a few select summer months. Furthermore, Carnegie Mellon University is not situated near a diversity of still, natural bodies of water (although cyanobacteria have previously been reported in the Allegheny Reservoir).9,10 Whether it is necessary to spike natural water samples or not, the study of microcystin degradation in more realistic water matrixes is an important goal.
References:
(1) Warner, G. R.; Somasundar, Y.; Jansen, K. C.; Kaaret, E. Z.; Weng, C.; Burton, A. E.; Mills, M. R.; Shen, L. Q.; Ryabov, A. D.; Pros, G.; et al. Bioinspired, Multidisciplinary, Iterative Catalyst Design Creates the Highest Performance Peroxidase Mimics and the Field of Sustainable Ultradilute Oxidation Catalysis (SUDOC). ACS Catal. 2019, 7023–7037. https://doi.org/10.1021/acscatal.9b01409.
(2) Shawwa, A. R.; Smith, D. W. Kinetics of Microcystin-LR Oxidation by Ozone. Ozone: Science & Engineering 2001, 23 (2), 161–170. https://doi.org/10.1080/01919510108961998.
(3) Shoemaker, J.; Tettenhorst, D.; Delacrus, A. Method 544. Determination of Microcystins and Nodularin in Drinking Water by Solid Phase Extraction and Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS). Environmental Protection Agency 2015.
(4) Draper, W. M.; Xu, D.; Behniwal, P.; McKinney, M. J.; Jayalath, P.; Dhoot, J. S.; Wijekoon, D. Optimizing LC-MS-MS Determination of Microcystin Toxins in Natural Water and Drinking Water Supplies. Anal. Methods 2013, 5 (23), 6796–6806. https://doi.org/10.1039/C3AY41328D.
(5) Zhang, L.; Ping, X.; Yang, Z. Determination of Microcystin-LR in Surface Water Using High-Performance Liquid Chromatography/Tandem Electrospray Ionization Mass Detector. Talanta 2004, 62 (1), 191–198. https://doi.org/10.1016/j.talanta.2003.07.001.
(6) Saxitoxin, Microcystin, Cylindrospermopsin & Bravetoxin Test Kits https://www.abraxiskits.com/products/algal-toxins/ (accessed Jan 27, 2020).
(7) Rivasseau, C.; Martins, S.; Hennion, M.-C. Determination of Some Physicochemical Parameters of Microcystins (Cyanobacterial Toxins) and Trace Level Analysis in Environmental Samples Using Liquid Chromatography. Journal of Chromatography A 1998, 799 (1), 155–169. https://doi.org/10.1016/S0021-9673(97)01095-9.
(8) Wirsing, B.; Flury, T.; Wiedner, C.; Neumann, U.; Weckesser, J. Estimation of the Microcystin Content in Cyanobacterial Field Samples from German Lakes Using the Colorimetric Protein–Phosphatase Inhibition Assay and RP-HPLC. Environmental Toxicology 1999, 14 (1), 23–29. https://doi.org/10.1002/(SICI)1522-7278(199902)14:1<23::AID-TOX5>3.0.CO;2-9.
(9) Schellhammer, M. Harmful Blue-Green Algae Found on Allegheny Reservoir. The Bradford Era. September 23, 2017.
(10) Harmful blue–green algal bloom detected at Allegheny Reservoir http://www.lrp.usace.army.mil/Media/NewsReleases/tabid/11552/Article/490654/harmful-bluegreen-algal-bloom-detected-at-allegheny-reservoir-in-new-york.aspx (accessed Jun 24, 2019).
Journal Articles:
No journal articles submitted with this report: View all 1 publications for this projectSupplemental Keywords:
Iron catalyst, green catalyst, blue-green algae, harmful algal bloom, water remediation, water treatment, drinking water, recreational waterRelevant Websites:
Institute for Green Science Department of Chemistry Exit
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.