Grantee Research Project Results
2019 Progress Report: Synthesis and Characterization of Fluorinated Hydrocarbon Anion Exchange Resins for the Extraction of Perfluorinated Chemicals
EPA Grant Number: SU839454Title: Synthesis and Characterization of Fluorinated Hydrocarbon Anion Exchange Resins for the Extraction of Perfluorinated Chemicals
Investigators: Danielson, Neil , Almquist, Catherine B , Berberich, Jason , Kuehl, Emily , Spilker, Robert , Smith, Abby , Wourms, Hannah , Xie, Ruichao
Current Investigators: Danielson, Neil , Berberich, Jason , Almquist, Catherine B , Smith, Abby , Wourms, Hannah , Beckman, Matthew , Kuehl, Emily , Spilker, Robert
Institution: Miami University - Oxford
EPA Project Officer: Page, Angela
Phase: I
Project Period: December 1, 2018 through November 30, 2019 (Extended to November 30, 2021)
Project Period Covered by this Report: December 1, 2018 through November 30,2019
Project Amount: $14,999
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2018) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
We propose to design and test a continuous-flow water purification device that uses a novel fluorinated anion exchange sorbent for removal of perfluoroalkyl compounds including perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS) from drinking water. Our idea improves upon adsorption technologies such as activated carbon and anion exchange resins, which are not specific for perfluoroalkyl compounds (PFCs). While it has been demonstrated that these adsorption materials are effective in removing perfluoroalky compounds, they are prone to reduced effectiveness in the presence of co-existing organic matter in water. Organic compounds can outcompete PFCs for binding sites reducing effectiveness. We have preliminary data showing the synthesis of a mixed-mode fluorinated polymer-anion exchange resin is straightforward and the ratio of fluorocarbon/anion exchange functionality can be tailored. The fluorous affinity plus the anion exchange interaction should result in improved selectivity for PFCs and reduced competition by organic matter in water. Adsorption kinetic studies in both the batch and column modes are outlined. Because of the complete polymeric nature of our adsorbent, facile cleaning and long term stability permitting reuse of the material is expected. Our ultimate goal is to develop an adsorbent material than can be used as a point-of-use filter, like a Brita® filter, or as an adsorbent material in columns for reliable removal of PFCs from drinking water.
Perfluoroalkyl compounds (PFCs) are anthropogenic industrial compounds used in many applications, including surface coatings, surfactants, and flame retardants. They are, however, emerging contaminants due to their recalcitrant nature; they are persistent and very stable in air, water, and soil environments. PFCs have been detected in water and soil samples all over the globe, as well as in humans and in wildlife and even in areas as remote as the artic. They have been reported as bioaccumulative, with detectable concentrations in humans and in both aquatic and terrestrial animals. For reasons of their stability and bioaccumulative nature, and for possible health effects on humans, efforts have been made to restrict their production and use worldwide.
This design will have a significant impact on people through removal of PFCs such as PFOA and PFOS which have been linked to increased risks in cancer, obesity, and immune suppression. Reducing PFCs will improve people’s health to allow them to build prosperity within their communities. The implementation of improved, PFC selective fluorinated anion exchange adsorbents will promote a sustainable planet since the fluorinated adsorbents can be regenerated and reused for many years.
Students working on this project are from the Departments of Chemistry/Biochemistry and Chemical, Paper and Biomedical Engineering. Our design team currently consists of one graduate student and four undergraduates of diverse science and engineering backgrounds. Students on the design team will learn to develop sustainable solutions for removal of PFCs from water in impacted communities by considering a range of economic, and environmental inputs to ensure the device’s long-term viability. The design team will also educate the Miami University student body about using this technology to decrease water pollution by presenting their work at national and regional conferences, University Undergraduate Research Forums, and Senior Design and Capstone courses.
Progress Summary:
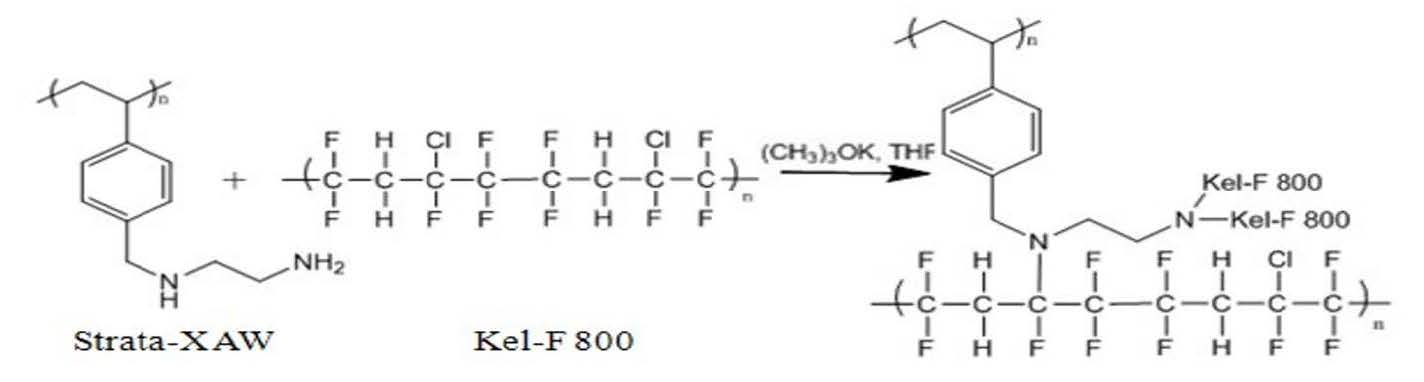
We proposed an approach to prepare an adsorption resin that will bind PFOA and PFOS using different modes. The first mode is through attraction of the fluorocarbon substituent on PFOA and PFOS to a fluorocarbon surface on the adsorbent material. The second mode uses anion exchange (charge interactions) to attract the anion of PFOS and PFOA to the adsorbent interface. Toward this goal, we modified a commercially available anion exchange resin (Strata-XAW aka Sepra-ZT-WAX) with a highly fluorinated polymer (Kel-F800) (Scheme 1). The modification of the resin was confirmed by IR spectroscopy and elemental analysis. However, modification of the starting resin with the fluorinated polymer did reduce the surface area of the resin by approximately 25%.
Scheme1. Modification of Strata-XAW anion exchange resin with Kel-F 800 fluoropolymer.
How well the newly synthesized adsorbent bound PFCs was determined by measuring the adsorption capacity and potentially kinetic parameters of the adsorbents for PFOA and PFOS using a continuous flow adsorption column. The adsorption columns were also prepared to eventually investigate how representative co-contaminants present in the water affect adsorption capacity and adsorption kinetics of the PFCs. PFOA was observed to adsorb strongly to the Sepra-ZT-WAX anion exchange column and to the Kel-F modified columns. Using the breakthrough times from column experiments, flow rate, PFOA concentration, and mass of the column packing, a loading capacity of 1.1 x10-3 meq/g-m2 was found for both the fluorinated and unfluorinated resins. Hence, the unmodified and fluorous modified anion exchange resins show similar binding capacity for PFOA per unit of surface area.
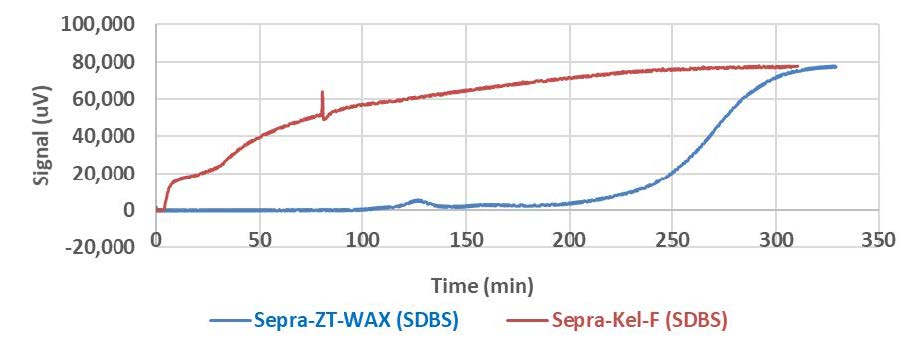
One problem of many commercially available anion exchange resins is poor selectivity of the resin for PFCs over other organic compounds. If the goal is to remove all PFCs, it is important for the resin to selectively adsorb PFCs over other organic materials that may be present in groundwater or wastewater. An example breakthrough curve of an adsorption column study is shown in Figure S1. In this study, a non-fluorinated commercial detergent that is commonly used in laundry detergents is pumped through the column. If the detergent binds to the column, the signal will remain close to zero. If the detergent flows through the column without binding, the signal will increase. What is observed is that the non fluorinated detergent binds tightly to the Sepra-ZT-Wax adsorbent whereas little binding is observed with the Kel-F modified resin. This indicates the fluorinated anion exchange resin should have good binding capacity for PFCs even in the presence of organic compounds such as detergents.
Figure S1: Breakthrough curves for the detergent sodium dodecylbenzene sulfonate (SDBS) in water using the Sepra-Kel-F column and Sepra-ZT-WAX column in water at a flowrate of 1 mL/min.
Future Activities:
Based on our Phase I work, we have developed a straightforward, flexible approach to synthesize a fluorocarbon polymer weak anion exchange resin. Both ion exchange and fluorphilic retention for PFOA can be differentiated on the Sepra-Kel-F polymer. Both the Sepra-Kel-F and the Sepra-ZT-WAX polymers have the same meq/g-m2 capacities. Retention selectivity of the Sepra-Kel-F column for PFOA as compared to the commonly used detergent dodecylbenzene sulfonate is significantly better as compared to the Sepra- ZT-WAX column. These polymers are pH and mechanically stable for potentially unlimited recycle and reuse (unlike silica based materials). This combination of weak anion exchange and fluorocarbon polymers should produce an adsorbent material that can be used to facilitate detection of PFCs in environmental samples at the sub ppb level and also be applied for large scale-purification of water to make it free of PFCs.
Proposed Phase II Objectives and Strategies: The major goal of the Phase II effort is to use what was learned from the Phase I work to develop an anion exchange resin for large scale purification efforts. The resins used in the preliminary studies were designed for small scale analysis. In the Phase II effort, larger particle resins will be modified and optimized to maximize surface area and adsorption capacity. For best performance, the surface area of the resins should be large, and should bind high levels of PFCs. Furthermore, the EPA limit for PFCs in water is 70 ppt. We will develop methods for quantification of PFCs at levels well below the EPA limit so that adsorption studies can be performed at concentrations relevant to drinking water. Finally, a lab-scale system will be built to simulate an industrial scale system. This system would be similar to what would be found for the softening of drinking water. With this scaled-up system, studies would be performed to determine the adsorption capacity of the resin, regeneration procedures, selectivity for PFCs in the presence of common contaminants, and resin stability.
Journal Articles:
No journal articles submitted with this report: View all 5 publications for this projectSupplemental Keywords:
PFCs, PFOA, PFOS, water treatment, water purification technologies, drinking water treatment technologies, adsorption, drinking water, water purification, potable water, sustainable technology.Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.