Grantee Research Project Results
2019 Progress Report: Developing a Low-Cost Wireless Device for Real-Time Monitoring of Lead Levels in Drinking Water
EPA Grant Number: SU839450Title: Developing a Low-Cost Wireless Device for Real-Time Monitoring of Lead Levels in Drinking Water
Investigators: Tang, Xiaochao , Song, Xiaomu , Saha, Dipendu , Wang, Po-Yen , Buchter, Gabrielle , Nordfors, Ian , Kienbaum, Madeleine , Wajda, Alexander
Institution: Widener University - Main Campus
EPA Project Officer: Aja, Hayley
Phase: I
Project Period: November 1, 2018 through October 31, 2019 (Extended to October 31, 2020)
Project Period Covered by this Report: November 1, 2018 through October 31,2019
Project Amount: $14,935
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2018) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources , Sustainable and Healthy Communities
Objective:
The lead-detecting device consists of two primary components: lead ion-selective electrode (ISE) sensing membrane and a microcontroller-based unit. The following sections describe the preliminary results and outcomes of developing the two primary components of the lead-detecting device.
Progress Summary:
This study has selected ion-selective electrode (ISE) membrane as the sensing component because ISE membrane has proven to show low detection limits, good selectivity, low cost and potential to be integrated in a miniature device (Tang et al., 2018). The research team has adopted lead-selective copolyanilline (AN-co-HSA) ionophores embedded in PVC membrane to detect trace levels of lead. The research team conducted a series of tests on membranes fabricated with various ionphore/Na/TBP/PVC resin ratios in order to determine the optimal ratio that would lead to an acceptable sensitivity (Table 1).
Table 1. Testing Plan for ISE Membranes
| Membrane Group | Membrane Specimen | Ionophore (mg) | NaTPB (mg) | Resin (mg) | THF(mL) | Ratio |
| A | 1 | 150 | 300 | 4500 | 2 | 1:02:30 |
| 2 | 750 | 300 | 4500 | 2 | 5:02:30 | |
| 3 | 900 | 300 | 4500 | 2 | 6:02:30 | |
| B | 1 | 150 | 600 | 4500 | 2 | 1:04:30 |
| 2 | 150 | 900 | 4500 | 2 | 1:06:30 | |
| 3 | 150 | 1200 | 4500 | 2 | 1:08:30 |
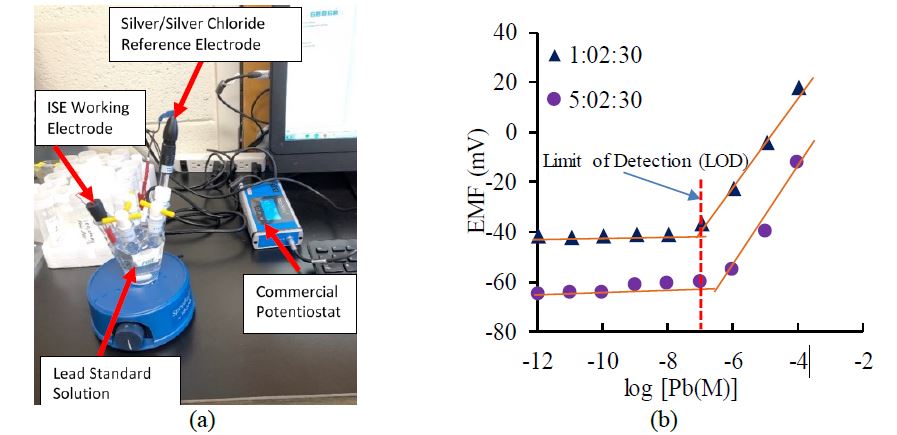
The membranes was subjected to open-circuit potential (OCP) measurements in Pb(NO3)2 standard solutions by a commercial potentiostat, PalmSens3 (PalmSens BV, GA Houten, The Netherlands) (Figure 1-a). As can be seen in Figure 1-b, two of the ratios show a limit of detection (LOD) of 10-7 mol./L (M) or 33 parts per billion (ppb) (Buchter, 2019). It is noted that the current limit of detection is still higher than the EPA action level (15 ppb) for lead concentration in drinking water (EPA, 1991). The research team will continue to work to enhance the sensitivity of the ISE membrane to lower its limit of detection.
Figure 1. Electromotive force (EMF) measurement: (a) setup for open-circuit potential (OCP) measurement using commercial potentiostat; (b) EMF responses of sensing membranes to lead solutions of different concentrations.
In addition to open-circuit potential (OCP) measurements to characterize the electrochemical responses of the sensing membranes to lead solutions, scanning electron microscope (SEM) and atomic force microscopy (AFM) were used to characterize and investigate the topography of the membranes (Figure 2). SEM image reveals that there are micro cavities and a striped pattern on the ISE membrane (Figure 2-a). A 2.5×2.5 μm AFM topography also shows striped pattern on the membrane and ionophores and their distributions. It can be seen from the AFM image that the ionophores are not necessarily disbursed and uniformed distributed, which may adversely affect the sensitivity and performance of the ISE membrane.
Figure 2. Ion-selective electrode (ISE) membrane: (a) scanning electron microscope (SEM) image; (b) atomic force microscopy (AFM) topography.
Development of Low-Cost Custom-Built Electrometer
Ion-selective electrode (ISE) sensing membranes are usually used in conjunction with a potentiostat to measure electromotive force (EMF). Commercial off-the-shelf potentiostats typically cost several thousands of US dollars or more. As one of the goals of this project is to develop a device that is low-cost and affordable to allow for widespread use, the project team has designed an electrometer to specifically measure the low potential without drawing any current from the circuit. A prototype of the electrometer has been built and encased with a LED display (Figure 3-a). A microcontroller with a wireless module is mounted on the circuit board (Figure 3-b).
Figure 3. Custom-built low-cost electrometer: (a) encased with LED display; (b) circuit board with a mounted microcontroller that has a wireless module.
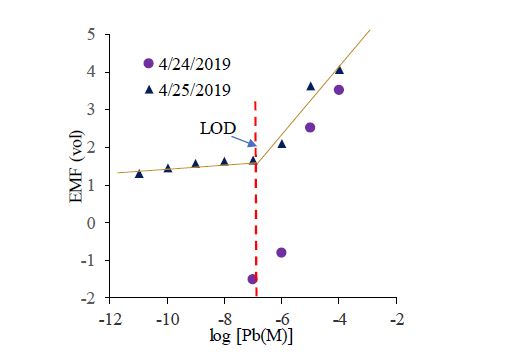
Preliminary testing of the custom-built electrometer shows that the device is able to capture the limit of detection (LOD) of the ISE sensing membrane (Figure 4). However, as can be seen in Figure 4, the electrometer shows reasonably good consistency only at higher lead concentrations. Further investigations and fine tuning are needed to ensure consistent and reliable measurements.
Figure 4. Open-circuit potential (OCP) measurements by custom-built electrometer.
Future Activities:
The research team will continue to work to move the project forward. Specifically, efforts of the research team will be centered on the following three primary tasks:
1) The research team will continue to improve the sensitivity of the ion-selective electrode new ionophores to examine their potential in enhancing sensitivity of the ISE membrane.
2) The research team will continue to work to improve the custom-built low-cost electrometer to improve its consistency and reliability for open-circuit potential measurements for the ISE membranes. The research team will review and fine tune the circuit design of the electrometer to enhance the precision of the device. The custom-built electrometer will be tested and validated by comparing testing result to that of a commercial potentiometer.
3) The lead-detecting device will be further examined by collecting actual water samples and using commercial services to analyze lead trace in the water samples. An established method such as inductively coupled plasma atomic emission spectroscopy (ICP-AES) will be used. Comparisons will be made between the testing results of the lead-detecting device and ICP-AES.
References:
Buchter, G. (2019). Developing an Ion-Selective Electrode Membrane for Lead Level Detection in Drinking Water, Master Thesis, Widener University, Chester, PA.
Environmental Protection Agency (EPA). 1991. Safe drinking water act lead and copper rule (LCR). Federal Register, 56: 26460-26564.
Tang, X.; Wang, P.-Y.; Buchter, G. (2018). “Ion-Selective Electrodes for Detection of Lead (II) in Drinking Water: A Mini-Review.” Environments, 5, 95,doi:10.3390/environments5090095.
Journal Articles:
No journal articles submitted with this report: View all 1 publications for this projectProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.