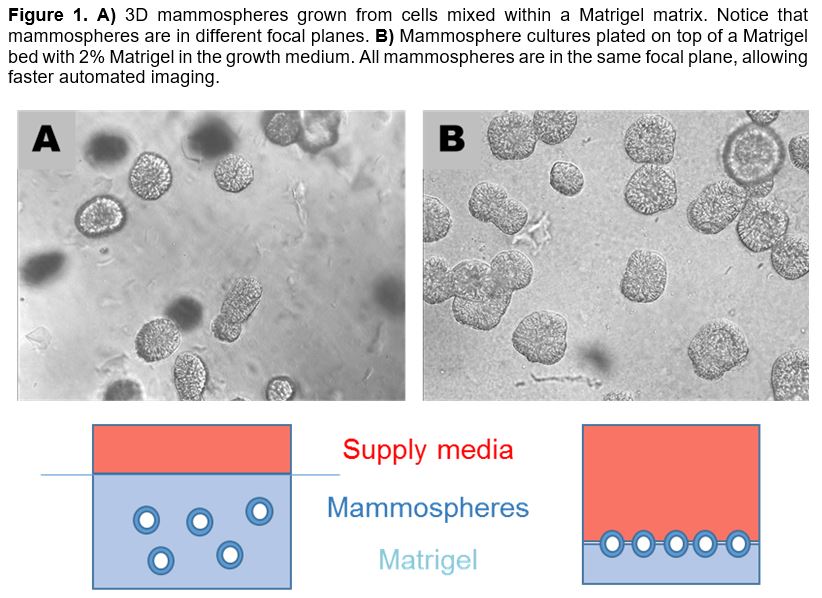
Grantee Research Project Results
2015 Progress Report: Mammosphere Bioreactor For Life-Stage Specific Toxicology
EPA Grant Number: R835736C001Subproject: this is subproject number 001 , established and managed by the Center Director under grant R835736
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Mickey Leland National Urban Air Toxics Research Center (NUATRC)
Center Director: Beskid, Craig
Title: Mammosphere Bioreactor For Life-Stage Specific Toxicology
Investigators: McCawley, Lisa J. , Auner, Alex , Markov, Dmitry , Schafer, Emily , Fryman, Phillip , Telange, Rahul
Current Investigators: McCawley, Lisa J. , Markov, Dmitry
Institution: Vanderbilt University
Current Institution: University of Pittsburgh , Vanderbilt University
EPA Project Officer: Aja, Hayley
Project Period: December 1, 2014 through November 30, 2018 (Extended to November 30, 2019)
Project Period Covered by this Report: December 1, 2014 through November 30,2015
RFA: Organotypic Culture Models for Predictive Toxicology Center (2013) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Taking advantage of unique collaborations afforded through VPROMPT, we will develop a microfluidic Mammary Gland organotypic culture model (MG OCM) for toxicant assessment, for monitoring dynamic toxicant-dependent changes to cellular functions and signaling cascades, and for elucidation of Adverse Outcome Pathways. In vivo the mammary gland is particularly sensitive to toxicant exposures during different life stages co-incident with times of active tissue growth and remodeling. We propose to extend our previously developed, microfabricated Mammary Gland Thick Tissue Bioreactor (MG-TTB) to support the combination of 3D culture methods with controlled exposure to investigate chemicals for potential mammary gland toxicity. We will utilize a simple mammosphere organotyic culture system and additionally evaluate a tri-culture system (normal human mammary epithelial cells + human mammary fibroblasts + subcutaneous adipocytes) for a more accurate recapitulation of mammary gland biology and crosstalk between heterotypic cells. We will validate this system for use as a medium throughput toxicology screening as well as for high information content analysis of targeted toxicant-dependent alterations to mammary formation. We will exploit this system for in vitro evaluation of potential environmental toxicants for effects on mammary development using both 1) chronic exposure and 2) acute exposures co-incident with various stages of gland development. Furthermore, this system will be used for high content analysis of putative toxicants with a focus on key biomarkers.
Our specific objectives to validate the microfluidic OCM for toxicants are to:
- develop and validate the predictive utility of self-contained, fully-automated MG OCM modules;
- investigate the effects of chemical exposures on MG OCMs for chemicals shown to reduce lactation index in only the F2 generation of multigenerational studies – strongly suggesting a role for life-stage specific exposure; and
- develop and validate toxicant assessment for compounds requiring metabolic activation using a paired Liver-OCM/MG-OCM.
Progress Summary:
For this first year, we focused on protocol development for medium throughput on-chip analysis using known toxicants of mammary development and the development of subchronic delivery of compounds. Specifically,
- We have upgraded a previously developed TTB to contain six culture chambers to be compatible with medium throughput screening systems and active pumping using miniature, custom-built pumps from Project 5.
- We have developed a protocol to assess toxicant interaction with PDMS, the primary material of our TTB, using BioTek NEO microplate reader at HTS core to screen for potential toxicant sequestration from the solution, complemented with FT-IR based measurements to probe PDMS surface directly.
- We have tested various PDMS treatments and their effect on toxicant-PDMS interaction such as: a) cross-linking; b) annealing; and c) state of oxidation with a subset of toxicants. These drug-PDMS assessments are useful to establish effective/actual drug concentration levels within the MC-OCM, prior to assessment of toxicant effect on mammary formation within MG OCM.
- As part of our medium throughput assessment of putative toxicant effect on mammosphere formation, we are applying Caspase-3 and histone 2B nuclear marker lentiviruses to monitor apoptosis and proliferation using MCF-10A and MCF-7 cell lines. We assessed using transduction of a subpopulation of cells (~15%) to prevent off-target influences of the lentiviruses; however, this proved to be below the usefulness of the system. We have analyzed stable expression of H2B in one cell line and determined that morphogenesis was unaltered when selecting and enriching expressing cells by Flow Sorting. We have initiated development of stable cell lines with simultaneous expression of both lentivirus systems.
Future Activities:
- We will continue collaboration with Project 5 on upgrading RPPM drive mechanisms to a smaller (NEMA 11 or 8) form factor in order to accommodate both PRRM and PRV onto the same well-plate footprint along with MG bioreactor cartridge.
- Complete toxicant profiling efforts in assessing their interactions with PDMS material.
- Complete production of stable cell lines with dual expression of both lentivirus systems (Caspase-3 & H2B) and determine the effect of transfection on baseline morphogenesis.
- Validate MG OCM reactor system for medium throughput for stage specific and acute toxicity assessment.
Journal Articles:
No journal articles submitted with this report: View all 19 publications for this subprojectSupplemental Keywords:
mammary development, mammary toxicology, organs on chip, mammary on chip, PDMS bioreactors, thick tissue bioreactor, PDMS interactions, organotypic cell cultureRelevant Websites:
The Vanderbilt-Pittsburgh Resource for Organotypic Models for Predictive Toxicology Exit
Progress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R835736 Mickey Leland National Urban Air Toxics Research Center (NUATRC) Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R835736C001 Mammosphere Bioreactor For Life-Stage Specific Toxicology
R835736C002 Organotypic Culture Model to Analyze DevelopmentalLimbMalformationsResulting from Toxicant/Teratogen Exposure
R835736C003 Validating a fetal membrane on a chip model for characterizing
reproductive toxicant exposure risks
R835736C004 Organotypic Liver Model for Predictive Human Toxicology and Metabolism
R835736C005 Systems Engineering & Analysis for Organotypic Culture Models
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
3 journal articles for this subproject
Main Center: R835736
169 publications for this center
56 journal articles for this center