Grantee Research Project Results
2015 Progress Report: Human Stem Cell-Based Platform to Predict Selective Developmental Neurotoxicity
EPA Grant Number: R835552Title: Human Stem Cell-Based Platform to Predict Selective Developmental Neurotoxicity
Investigators: Terskikh, Alexey V. , Farhy, Chen
Current Investigators: Terskikh, Alexey V.
Institution: Sanford-Burnham Medical Research Institute
EPA Project Officer: Aja, Hayley
Project Period: September 1, 2013 through August 31, 2017
Project Period Covered by this Report: October 1, 2014 through September 30,2015
Project Amount: $800,000
RFA: Development and Use of Adverse Outcome Pathways that Predict Adverse Developmental Neurotoxicity (2012) RFA Text | Recipients Lists
Research Category: Human Health , Chemical Safety for Sustainability
Objective:
The goal of this project is to develop and implement a novel approach to assay developmental neurotoxicity of ToxCast chemicals using a human embryonic stem cell (hESC)-based model of neuronal development. For the first step, we proposed to determine cytotoxicity of 1200 ToxCast compound library towards human fetal NPCs selective cytotoxicity towards human fetal neural precursor cells (NPCs). In fact, this has been partially accomplished during the second year (see detailed description below) and will be completed in the third year. Next, we will determine selective cytotoxicity of 1200 ToxCast compound library towards ventral and/or dorsal NPCs. In the proposed strategy, the partial or complete loss of a particular NPC population (e.g., ventral or dorsal NPCs) is a key event that precedes and predicts a specific adverse neurodevelopmental outcome both in vitro and in vivo. The objectives of this project are:
Objective 1: Develop an HTS platform based on hESC-derived NPC to identify ToxCast phase I chemicals that selectively affect ventral or dorsal NPCs.
Objective 2: Adapt the human NPC-based assays to 384-well format and identify ToxCast phase II chemicals selectively affect ventral or dorsal NPCs.
Objective 3. Investigate the cellular and molecular mechanisms of action of active ToxCast chemicals.
Progress Summary:
In the past year, we have made significant progress towards these objectives, which remain as originally proposed. Following the modifications and improvements incorporated in our strategies in the previous year we began screening of 1200 ToxCast compounds in 384-well format using automatic cell cytometry acquisition algorithm supported by the IC200 instrument at SBP Medical Discovery Institute. We have chosen to begin the screening efforts using human fetal neural progenitors (hfNPCS) for a number of reasons: (1) compared to hESC-NPCs that were developed in the dish with limited supply of growth factors, hfNPCs were developed in utero and thus represent in our opinion bona fide human neural precursors; (2) on the other hand, hfNPCs represent a sufficiently diverse range of developmental fates in respect to various types of neurons (including dopaminergic TH+ neurons) and astrocytes; and (3) we have a large repository of frozen hfNPCs making identical aliquots of these cells ideal for optimization and standardization purposes.
1. Characterization of human fetal neuronal progenitors.
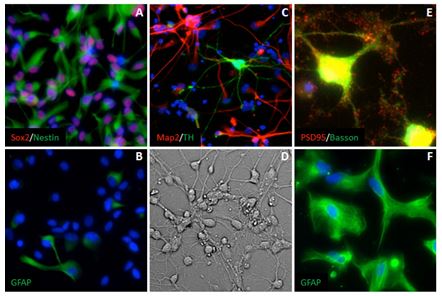
Multiple studies have demonstrated the close similarity of the human pluripotent stem cell-derived neuronal progenitors and their in vivo counterparts. However, we just began learning about their ability to recapitulate toxicity profiles(1). As mentioned above, prior to establishing selective toxicity of ToxCast compound to ventral and dorsal hESC-derived NPCs we have optimized the screening process to establish a baseline cytotoxicity profile using a naïve system, which closely mimics cell populations found in vivo. For this purpose we took advantage of commercially available hfNPCs (third trimester) that were previously used in the PI laboratory. First, we characterize hfNPCS in respect to the expression of key markers of neural stem/progenitor cells such as Sox2 and Nestin. As expected, the majority of hfNPCs were found to express Sox2 and Nestin albeit at various expression levels (Figure 1A). Only a small subpopulation (16%) was positive for the astrocyte marker GFAP (Figure 2B) consistent with the expression of this marker in some stem/progenitor cells(2). To ascertain that these primary derived cells maintained neurogenic and gliogenic potential in culture we propagated hfNPCs under differentiation conditions for period up to 2 months. For neuronal differentiation hfNPCs were cultured in basic neurobasal media supplemented with B27 and BDNF, GDNF, DAPT, Ascorbic acid and cAMP(3, 4). Within 3-4 weeks the majority of differentiating cells expressed the mature neuron marker MAP2 (Figure 1C; red). Due to their heterogeneous nature hfNPCs generate multiple neuronal lineages such as glutamatergic and dopaminergic neurons (see example of TH+ neurons Figure 1C; green). Additionally, hfNPCs derived neurons generated elaborate neurite arbors (Figure 1D) as well as complex synaptic networks as evident by colocalization of the presynaptic protein Bassoon (Figure 1E; green) and the postsynaptic PSD95 (Figure 1E; red). For glial differentiation, hfNPCs were cultured in basic DMEM/F12/Neurobasal media supplemented with 2% fetal bovine serum (FBS). After 2 weeks the cells acquired the typical flat morphology common to astrocytes and 73% of cultured cells expressed the mature astrocytes marker GFAP (Figure 1F).
Figure 1. Human fetal neuronal progenitors are heterogeneous and retain multipotency in culture. Third trimester neuronal progenitors express the pan NPC markers Sox2 and Nestin (red and green in A). Only 16% of hfNPCs cultured under passaging conditions express the glial marker GFAP (green in B). Human fetal NPCs derived neurons express the mature neuron marker Map2 (red in C). Differentiation of hfNPCs generates multiple subtypes such as dopaminergic neurons (green in C). Differentiated hfNPCs form comprehensive neurite arbors (D) and form complex synaptic networks, as shown by colocalization of the presynaptic protein Basson (green in E) and the postsynaptic protein PSD95 (red in E). Human fetal NPCs retain gliogenic potential in culture and can be differentiated to express the astrocytic marker GFAP (green in F).
2. Automated high-content screening of ToxCast phase-I compounds.
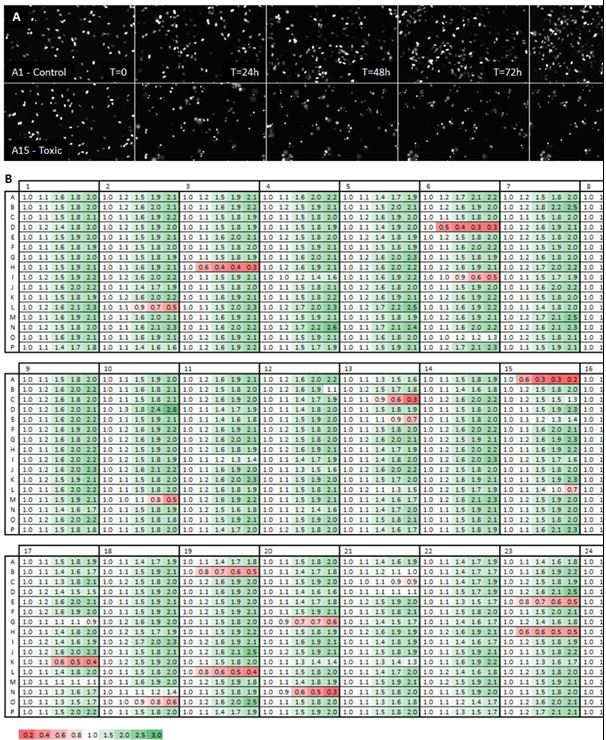
Next we determined the toxicity of 386 ToxCast phase-I compounds using primary hfNPCs in 384-well format. For this purpose, hfNPC were engineered with adenovirus delivering nuclear localized mCherry to enable automatic fluorescence tracking of cells. Live cell image acquisition using automated multiwall microscopes as well as accurate cell number scoring using batch image analysis programs enables multiple rounds of observation of the same cells across the entire time of the experiments. Fluorescence-tagged hfNPCs were seeded at 5000 cells per well and compounds were dispensed using an acoustic Liquid Handler (Echo 555, LabCyte) to a final concentration of 10 µM. Cells were cultured for 4 days in presence of ToxCast compounds and imaged daily. Images were acquired using IC200-KIC, C200-KIC™ a fully automated high-content screening system (Vala Sciences). The examples of fields of control and toxic compound shown in Figure 2A. The images were analyzed using Acapella Imaging Software (PerkinElmer) to score nuclei number as well as morphological parameters such as nuclear area and nuclear roundness. For initial statistical analysis the number of nuclei in each well was normalized to T=0 and presented as fold change in each following day (overview of growth dynamics in each well). The first column was dedicated to vehicle treated cells (DMSO; see also grey labeled wells in Figure 3A), green label indicates increased number of cells while red indicates reduce cell numbers (Figure 2B). Visual examination of the screening results revealed a number of obvious cytotoxic candidates (red stripes). However, such approach could be subjective and time consuming when large ToxCast libraries have to be screened. Therefore, we developed an objective statistical approach (based on nonlinear regression analysis) to analyze the growth behavior/cytotoxicity using the pilot 368 set of ToxCast compounds.
Figure 2. ToxCast compounds can alter growth of hfNPCs. Human fetal NPCS labeled with nuclear mCherry were treated with 368 phase-I ToxCast compounds. Growth was monitored daily for 4 days using automated microscopy (IC200-KIC, Vala Sciences). Image analysis was preformed using Acapella Imaging Software (PerkinElmer). (A) Pictures showing normal growth of vehicle treated hfNPC and rapid cell death and reduced cell numbers induced by a fast acting toxic ToxCast compound. (B) Control wells treated with vehicle (DMSO) showed approximately 2-fold growth during the 4 days while approximately 7% of all compounds displayed a significant change in growth pattern.
3. Nonlinear regression analysis of cell growth after administration of ToxCast compounds.
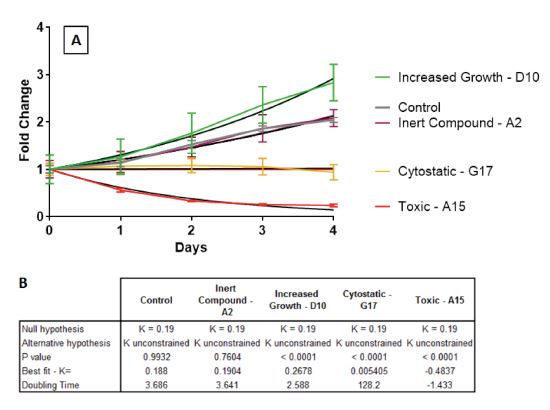
Nonlinear regression analysis is used widely in toxicology to assess the effect of chemicals on cell growth and differentiation(5-7). To identify compounds which significantly alter the growth of hfNPCs control data was fit to an exponential growth function (Y=eKX) and was found to match the mathematical model with K=0.188 and doubling-time of 3.7 days with confidence of greater than 95%. The model prediction of hfNPCs doubling time (3.7 days) is in good correlation with the observed growth rate of hfNPCs in our laboratory. Next, the growth curves of cells treated with 386 ToxCast compounds were fit to control derived function (Figure 3A,B). The data from the cells treated with about half of the tested compounds (195 out of 386) fitted well to the growth of the control cells, suggesting that these compounds had no observable effect during the time of our analysis. On the other hand, 191 ToxCast compounds did not fit the control equation with at least 5% confidence. Many of the 191 compounds revealed a rather minor growth deviation from the control cells. A more in depth analysis of these compounds will await the completion of the screening of 1200 ToxCast library. However, we have selected a set of criteria enabling us to visualize the most pronounced classes observed among the first batch of compounds.
Figure 3. Empirically growth curves can be fit to function using nonlinear regression. (A) Growth curves obtained for control and each compound treated cells was fit to an exponential growth function (Y=eKX). (B) Vehicle treated control was found to match the mathematical model of K=0.188 and doubling-time of 3.7 days with confidence of greater than 95%. Growth curves of cells treated with compounds which altered hfNPCs growth significantly deviate from control equation (green, yellow, red in A) while inert compounds which do not affect hfNPCs growth do not deviate from control equation (purple in A).
4. Multiple ToxCast compounds display diverse effects on growth of hfNPCs.
We have implemented several strategies to visualize the most obvious classes of compounds according to their effect on hfNPCs. Note that the examples of several types of behavior presented below could be extended by the compounds expected to be identified by screening the remaining part of the ToxCast library and also by changes in the stringency/selection criteria.
Cytostatic compounds.
We found that 10 compounds, which increased the doubling time of hfNPCs by at least 2-fold (ranged from 2- to 34-fold). Therefore we did not observed the change in cell numbers in the wells exposed to these 10 compounds during the 4 days of our assay. However, visual inspection and fluorescent imaging confirmed that the cells were present in the wells. Therefore, these compounds were considered cytostatic (Figure 4, yellow marked cells). Note that 2-fold change compared to control is an arbitrary call prompted by a common sense and visualization clarity.
Figure 4. ToxCast compounds display diverse effects on growth of hfNPCs. 34 of the 386 ToxCast phase-I compounds tested were found to statistically alter the growth pattern of hfNPCs. (A) A table displaying fold change in cell number after 4 days of treatment with ToxCast compounds. (B) Growth curves of significantly altered compounds identified. Compounds affecting growth of hfNPCs were divided into 3 groups: compounds which increase growth (green in A,B,C), compounds which halt growth (cytostatic, yellow in A,B,C) and toxic compounds (red in A,B,C).
Cytotoxic compounds.
We found 18 compounds that could not be fitted to the (Y=eKX) equation (due to the formula constraint). These compounds are shown in Figure 4, labeled in red. Visual examination showed that most of these compounds could be directly observed from Figure 2 as cytotoxic compounds with various degree of toxicity. However, our ability to identify these compounds using the nonlinear fitting algorithms enable us to analyze large data sets and select toxic compounds automatically.
Proliferation promoting compounds.
Curiously, we identified many compounds, which induced decreased the doubling time of hfNPCs to different extent and therefore promote proliferation of hfNPCs. Here we focused on seven compounds, which were defined as decreasing the doubling time of hfNPCs by 20% or greater. These compounds were considered growth promoting (Figure 4 cells labeled in green). As with the cytotoxic compounds the bar at 20% growth increase is rather arbitrary. In fact, there are large numbers of compounds that exert a subtle growth promoting effect on hfNPCs.
Along with the increase in the number of the compounds tested (the current ToxCast library in hands comprise 1200 compounds), we will focus our efforts to fit empirically derived growth curves with various biologically-relevant growth constrains and will attempt to identify additional classes of growth patterns. For example, we found that toxic compounds described above could be further classified into fast acting toxic compounds deviated from control as early as 24 hours after compound application (Figure 5 cells labeled in red) and slow-acting toxic compounds which required at least 48 hours to diverge from the control growth pattern (Figure 5 cells labeled in blue).
Upon the completion of the screening of 1200 ToxCast compounds and once the identities of these compounds will be revealed multiple statistical correlations will be computed based on a wealth of existing data sets at EPA. Namely, EPA has established a large portfolio of toxicology data and bioinformatics, including the activity of ToxCast chemicals in ~500 cell-based and biochemical assays (www.epa.gov/ncct/toxcast). In vivo data is also available for these compounds (www.epa.gov/NCCT/toxrefdb).
In close collaboration with the EPA scientists (Dr. Thomas Knudsen's group), we will calculate statistical associations between the results obtained in our study and the data from ToxCast and ToxRefDB. These analyses will correlate our data with information on the gene/biochemical pathway activities of the identified compounds, thus providing insight into the mechanisms of developmental neurotoxicity.
Future Activities:
In the coming year, we plan to determine the effect of the entire phase-II ToxCast compound library (1200 compounds) on hfNPCs, ventral/dorsal NPCs. After compound identification procedure, we will further investigate the compounds that exert marked effect on hfNPCs. In particular, we will calculate statistical associations between the results obtained in our study and the data from ToxCast and ToxRefDB. This work will be performed in close collaboration with Dr. Thomas Knudsen at EPA within the frame of the cooperative agreement established during the second year of this project. Further work includes engineering hfNPCs as well as ventral/dorsal NPCs with Fucci to follow the changes in cell cycle dynamics in addition to the cell growth.
References:
- Schwartz MP, et al. Human pluripotent stem cell-derived neural constructs for predicting neural toxicity. Proc Natl Acad Sci U S A 2015;112(40):12516-12521.
- Nakano I, et al. Maternal embryonic leucine zipper kinase (MELK) regulates multipotent neural progenitor proliferation. The Journal of Cell Biology 2005;170(3):413-427.
- Bajpai R, et al. Molecular stages of rapid and uniform neuralization of human embryonic stem cells. Cell Death and Differentiation 2009;16(6):807-825.
- Cimadamore F, et al. Human ESC-derived neural crest model reveals a key role for SOX2 in sensory neurogenesis. Cell Stem Cell 2011;8(5):538-551.
- Chandler KJ, et al. Evaluation of 309 environmental chemicals using a mouse embryonic stem cell adherent cell differentiation and cytotoxicity assay. PLoS ONE 2011;6(6):e18540.
- Kleinstreuer NC, et al. In vitro perturbations of targets in cancer hallmark processes predict rodent chemical carcinogenesis. Toxicological Sciences 2013;131(1):40-55.
- Sipes NS, et al. Predictive models of prenatal developmental toxicity from ToxCast highthroughput screening data. Toxicological Sciences 2011;124(1):109-127.
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.