Grantee Research Project Results
Final Report: Sustainable Utilization of Coal Combustion Byproducts through the Production Of High Grade Minerals and Cement-less Green Concrete
EPA Grant Number: SU835349Title: Sustainable Utilization of Coal Combustion Byproducts through the Production Of High Grade Minerals and Cement-less Green Concrete
Investigators: Mohanty, Manoj K. , Kolay, Prabir , Kumar, Sanjeev , Liu, Jia , Rimmer, Sue , Wiltowski, Tomasz
Institution: Southern Illinois University - Carbondale
EPA Project Officer: Hahn, Intaek
Phase: II
Project Period: August 15, 2012 through August 14, 2014 (Extended to August 14, 2015)
Project Amount: $89,943
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet - Phase 2 (2012) Recipients Lists
Research Category: Pollution Prevention/Sustainable Development , P3 Awards , P3 Challenge Area - Sustainable and Healthy Communities , P3 Challenge Area - Chemical Safety , Sustainable and Healthy Communities
Objective:
- To develop low-cost process flowsheets for extracting valuable metal oxides, such as Iron oxide and Aluminum Oxide from the waste products of combustion of high-sulfur coal typically found in the Illinois basin.
- To develop a suitable process to utilize majority of the coal combustion residues as a useful product in the form of a geopolymer-based concrete without the use of any Portland cement.
- To educate the present and future university students about the challenges behind the continued use of coal-based electricity and the commercialization potentials of various high-value end uses of coal combustion byproducts.
Summary/Accomplishments (Outputs/Outcomes):
Fly Ash Magnetite Recovery: Studies were conducted to investigate the extraction of magnetite from fly ash through the application of a 2 stage wet-magnetic separation circuit (rougher-cleaner). These experiments were conducted using the CWLP source fly ash. The processing scheme involved a pre-concentration stage-I of the feed sample and a second stage cleaning using a low intensity magnetic drum in between stages. Major identified process parameters that affect the 



Alumina Recovery from Coal Fly Ash: &xperiments were conducted to recover Aluminum from CWLP fly ash, A leaching reactor (Model: 1K A LRI 000) was used for this test which has the 



Representative samples were sent for ICP-MS analysis. An Al concentration of 2.01 g/l leachate was obtained. This translated to an Al recovery of 66.2% for the leaching process. The low extraction can be due to the co-dissolution of silica and the subsequent formation of an unfilterable silica gel. Yao et al, 2014 have proposed that the glassy phase of fly ash dissolves rapidly and that the more stable quartz—mullite phase which remains dissolves more slowly. Other methods of leaching are getting more common such as lime sinter process, lime—soda sinter process, predesilication and lime—soda sinter combination process.
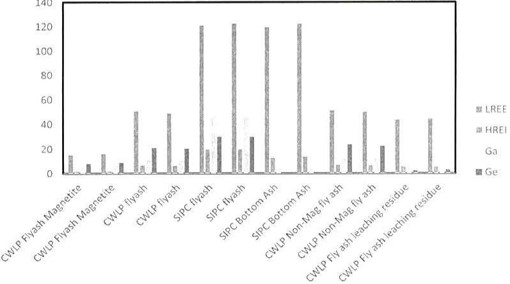
ICP-MS Study on Bottom Ash and Fly Ash: ICP-MS analysis was conducted on both bottom ash and fly ash to investigate the presence of precious and strategic metals. The results (Figure l) showed that the concentration of light REES in SIPC coal ash is in the range of 100-120 pm that is not very promising. The amount of heavy REES, however is not more than 20 ppm in any of the samples tested.
Figure 1. ICP-MS result for coal ash (CWLP and SIPC).
In addition, SIPC fly ash has a good concentration of silver (Ag), as high as 900 ppm, which can be further studied for its economic recovery. Other precious metal elements were not as significant as Ag as shown in Table l .
Table 1- Valuable metals concentration in different Coal Ash
| Fly ash and Bottom ash samples from different sources |
Precious Metals
|
Other Valuable Metals
| |||||||||
| Pd | Pt | Ag | Au | Re | Ga | Ge | In | W | Ti | Te | |
| PPB | PPB | PPB | PPB | PPB | PPM | PPM | PPM | PPM | % | PPM | |
|
| 10 | 2 | 2 | 0.2 | 1 | 0.1 | 0.1 | 0.02 | 0.1 | 0.001 | 0.02 |
| CWLP FIyash Magnetite | <10 | <2 | 32 | <0.2 | 2 | 6.2 | 8.1 | 0.02 | 0.8 | 0.042 | <0.02 |
| CWLP FIyash Magnetite | <10 | 2 | 32 | <0.2 | 1 | 6.7 | 8.9 | 0.02 | 0.9 | 0.043 | <0.02 |
| CWLP flyash | <10 | <2 | 76 | 0.9 | 57 | 11.4 | 21.3 | 0.06 | 2.1 | 0.074 | 0.07 |
| CWLP flyash | < 10 | <2 | 74 | <0.2 | 54 | 10.9 | 20.6 | 0.05 | 2.1 | 0.074 | 0.07 |
| SIPC flyash | 39 | 4 | 864 | 2.5 | 22 | 45.7 | 29.9 | 0.37 | 7 | 0.137 | 0.21 |
| SIPC flyash | 55 | 5 | 889 | 1.7 | 22 | 45.6 | 29.8 | 0.35 | 6.7 | 0.143 | 0.24 |
Coal Ash as an Effective Geopolymer Concrete Precursor: Ordinary Portland cement (OPC) generally used for preparation of concrete materials is known to have a large carbon footprint. Therefore geopolymer concrete testing was conducted using both fly ash samples collected for this study. Some of the important findings of the geopolymer concrete work completed using the two fly ash samples are summarized below:
From the sample characterization, both SIPC and CWLP fly ash samples are qualified as ASTM Class F type fly ash by examining Calcium content of close to or below 5 %. Particle size analysis indicated that 86 % of CWLP fly ash was finer 45 um while SIPC fly ash had 91% of -45 micron size fraction. The Loss on Ignition (1001) values examined for both SIPC and CWLP fly ash were 5.34% and 1.72%, respectively. The above-mentioned properties contribute in making the fly ash samples favorable for Geopolymer concrete production.
To optimize the strength of geopolymer concrete, a series of parametric studies were designed and conducted with both SIPC and CWLP fly ash type. The parameters targeted were 
The fractional factorial test was designed to investigate the effect of parameters on Geopolymer concrete strength. Mortar cube samples of 2 inch sides were made with SIPC fly ash. The mortar specimen with the highest compressive strength which reached 5063 psi at day 60 had NaOH concentration of 16 Molars, the silicate to hydroxide ratio of I : I , and were cured for 2 days at 90 O Celsius. The fractional factorial design test program indicated the concentration of hydroxide, curing time and temperature as the significant parameters with respect to the compressive strength of the specimen,
A full factorial design test was developed to determine the best possible combinations of hydroxide and silicate for the activator alkaline solution. The potassium hydroxide and silicate 

The optimal mixture design for SIPC geopolymer concrete was with 18 molar NaOH concentration and alkali to silicate ratio of 1:1. The compressive strength peaked at 6720 psi after 28 days of curing; it stabilized at 6530 psi after 60 days of curing. With the same mixture condition, the CWLP fly ash reacted differently. The hydroxide to silicate ratio of 1:2 was more favorable for CWLP geopolymer concrete.
A Box-Behnken test program was designed following the factorial design for the CWLP gepolymer, to determine the best process parameter vaues to achieve the maximum possible compressive strength of geopolymer concrete. The NaOH solution concentration, curing temperature and curing time were the three significant factors, and the three responses were the compressive strength at 7 th day and 28 th day after curing as well as the split tensile strength at 28 th day after curing. The levels for NaOH solution concentrations that were tested included 12, 14 and 16 Molars. For curing time, I, 3 and 5 days were chosen as variables. Curing temperature was varied in the range of 50 to 90 degrees Celsius.
Based on the Box-Behnken design test results, it was concluded that the alkali concentration and curing temperature are significant parameters affecting both compressive and tensile strengths of the CWLP fly ash-based geopolymer concrete. Lower alkali concentration and lower curing temperature resulted in high compressive strength of CWLP geopolymer concrete. The compressive strength of the CWLP fly ash based geopolymer increased with a decrease in alkali concentration, a trend that is exactly opposite to that observed with SIPC fly ash based geopolymer concrete.
The highest strength of CWLP concrete was achieved with 12 molar NaOH concentration and cured for 3 days under 50 Q C. The compressive strength and tensile strength reached 4613 psi and 388 psi at the 28 th day after curing. Although this compressive strength is not high as high as SIPC's, it certainly compares very favorably with the strength (—4000psi) of conventional OPC concrete.
The tensile strength refers to the greatest stress a material can tolerate when being stretched or pulled before failing or cracking. The tensile strength for the optimal SIPC concrete was examined to be 405 psi. High strength Geopolymer concrete sample were concluded to achieve higher tensile strength than the OPC counterpart.
Slump Test was conducted to measure the workability of fresh concrete. The slump height of 

Freeze-Thaw Tests were conducted on Geopolymer concrete with the optimal mixture of both SIPC and CWLP concrete. After 12 freezing and cooling cycles, there was over 5% mass loss on the Geopolymer concrete samples made with SIPC fly ash. The weight of CWLP based geopolymer concrete on the other hand increased by about I % of mass on average apparently 
An economic analysis was conducted to compare the cost of geopolymer concrete versus the OPC concrete. As detailed in the following table, the cost of Geopolymer concrete is compares very favorably with that of the OPC concrete cost. The costs/cubic yard in respectively order are $106.50 and $108.90.
| Geopolymer Concrete |
| Ordinary Portland Cement Concrete | |||
| Materials | Mass (kg) | cost ($/kg) | Materials | Mass (kg) | Cost ($/kg) |
| Fly Ash | 5.64 | - | Cement1 | 5.635 | 1.32 |
| Coarse A gregate | 18.46 | 0.41 | Coarse Aggre ate | 18.455 | 0.41 |
| Fine Aggregate | 13.86 | 0.38 | Fine Ag re ate | 13.862 | 0.38 |
| Sodium Silicate | 0.99 | 0.63 |
|
|
|
| Sodium Hydroxide | 0.99 | 0.64 |
|
|
|
| Water | 0.60 | - | Water | 3.32 |
|
| Total | 40.5 | 2.07 | Total | 41.3 | 2.11 |
| Specific Gravity= | 2.73 | Specific Gravity= | 2.78 | ||
| Cost per cubic yard = | 106.50 | Cost per cubic yard = | 108.90 | ||
Educational DVD Preparation: A step-by-step demonstration video of the main research components of this study was prepared to educate mostly high school students and teachers about the various beneficial uses of coal combustion wastes studied in this investigation. The video was shot at multiple locations in SIU Campus including external areas of the engineering building, the mineral processing laboratory, civil engineering concrete laboratory and our pilot scale facility at the Illinois Coal Development Park in Carterville, Illinois.
The twenty minute video, which has been uploaded to the project website (hup://www.usepap3.siu.edu/), presents an overview of the proposed flowsheet. The overview is then followed by stepwise demonstrations of the methodologies applied in the geopolymer concrete preparation including chemical preparation, geopolymer concrete mixing and preparation of cylindrical samples, demolding and strength tests conducted on the demolded specimen. The second half of the video demonstrates the extraction of magnetite minerals from fly ash. The video shows in real time, the extraction process using a low intensity drum magnetic separator at the 



The twenty minutes video in Mpeg 4 media format is hosted on YouTube and the link is also embedded on the project website. With an objective for public education on the topic being researched, hosting the video on YouTube presents a wider reach, and excellent shareability on social media channels. The chosen description of the video (using appropriate keywords) on the YouTube platform provides a detailed overview of the video's content to help viewers understand what they are watching and also plays a huge role in search engine optimization (SEO). In a little over two months of upload, the video has close to three hundred views from across the world.
Conclusions:
- Magnetite of high grade (Davis tube analysis of >96%) could be extracted from fly ash generated from the combustion of high sulfur coal found in the Midwestern US. Many other minerals of high value could be extracted from coal ash.
- F-class fly ash generated from the combustion of bituminous coal may serve as a good raw
material for geopolymer concrete. Geopolymer concrete specimen produced using two different F-class fly ash samples in this study provided compressive strength up to 6500 psi. The minimum strength achieved of about 4600 psi is better than that of the conventional cement concrete.
- The cost of geopolymer concrete compared very favorably with that of the conventional cement concrete. The respective cost figures on kg basis were $2.07 and $2.11; the respective costs/eubie yard were $107 and $109.
References:
- http://www.lowes.com/productDisplay?productld=4728843
- Illinois Ready Mix, Murpphysboro, Illinois
- Illinois Ready Mix, Murpphysboro, Illinois
- PQ Corporation (http://www.pqcorp.com/Portals/1/lit/04 SS 120112.pdf)
- http://www.buIkapothecary.com/raw-ingredients/other-ingredients-and-chemicals/sodium-hydroxide-lye/
Journal Articles:
No journal articles submitted with this report: View all 4 publications for this projectSupplemental Keywords:
Geopolymer, green concrete, metal extraction, coal ashProgress and Final Reports:
Original AbstractP3 Phase I:
Sustainable Utilization of Coal Combustion Byproducts through the Production of High Grade Minerals and Cement-less Green Concrete | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.