Grantee Research Project Results
2015 Progress Report: Cell-Free Neurochemical Screening Assays to Predict Adverse Effects in Mammals, Fish, and Birds
EPA Grant Number: R835170Title: Cell-Free Neurochemical Screening Assays to Predict Adverse Effects in Mammals, Fish, and Birds
Investigators: Basu, Niladri , Murphy, Cheryl A. , Head, Jessica Anne , Neitzel, Richard L.
Current Investigators: Basu, Niladri , Head, Jessica A , Murphy, Cheryl A. , Neitzel, Richard L.
Institution: University of Michigan , Michigan State University
Current Institution: University of Michigan , McGill University , Michigan State University
EPA Project Officer: Aja, Hayley
Project Period: September 1, 2012 through August 31, 2015 (Extended to August 31, 2017)
Project Period Covered by this Report: March 1, 2014 through February 28,2015
Project Amount: $1,199,222
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
- Objectives – The overall objective of this proposal is to advance an existing in vitro, cell-free neurochemical screening assay platform, and model data outputs to predict adverse, individual-level reproductive effects associated with toxicant exposure in mammalian, fish, and avian wildlife. The overall hypothesis is that several environmental toxicants will emerge to interact with and disrupt the function of neurotransmitter receptors, enzymes and transporters that have critical roles in vertebrate reproduction.
- Approach – We will study 60 toxicants (various metals, pharmaceuticals, personal care products, polycyclic aromatic hydrocarbons, halogenated aromatics, pesticides, real-world environmental mixtures) and 16 neurochemicals (receptors, enzymes, transporters) important in the neurotransmission of dopamine, GABA, glutamate, and acetylcholine, and 20 organisms (2-3 different freshwater fish, marine fish, terrestrial mammals, marine mammals, Arctic marine mammals, domestic birds, piscivorous birds, songbirds, rats and humans). All assays are cell-free and will be performed in 96-well microplates and are thus capable of being performed in a “high-throughput” manner. We have published widely on our approach, and here we adapt our screening platform to address the goals of the present RFA and mirror the U.S. EPA’s ToxCast program. In terms of research approach, first, a single concentration (50 µM or 0.5 mg/mL) of each toxicant (n = 60) will be evaluated in triplicate in each cell-free in vitro assay (n = 16) for each species (n = 20), thus resulting in 19,200 mean datapoints, plus many more after various controls are accounted for. Second, 120 toxicant-assay pairs that show most significant changes will then be further evaluated in each organism to derive concentration-response relationships. Third, a series of statistical and simulation models will aim to integrate results from cell-free assays to reproductive outcomes at the individual-level, which is an apical endpoint that can be used to predict population-level impacts.
- Expected Results – This work will result in the establishment of a high-throughput screening assay that can be used to predict reproductive impairment across multiple ecologically relevant species (birds, fish, mammals). Resources exist to adapt this platform to screen 1,000s of toxicants. It will generate important data on several toxicant classes (incl. real-world mixtures) that are of ecological relevance. Data outputs will be modeled to predict adverse outcomes at the individual level. Several publications, presentations and student dissertations will be produced.
Progress Summary:
Very few in vitro toxicity testing tools exist for the most standard ecotoxicological test species, and there is almost nothing for native species of ecological relevance. This research has advanced an in vitro, cell-free (acellular) neurochemical screening assay platform that can study chemical interactions against 4 neurotransmitter receptors and 3 enzymes critical to animal reproduction, behaviour and health. The assays were developed for more than 20 different organisms of multiple taxa, including freshwater and marine fish, birds, terrestrial and marine mammals. The tests were applied to study a diverse set of 80 potentially neurotoxic chemicals (such as metals, pesticides, personal care products, flame-retardants), and are currently being used to screen the U.S. EPA’s E1K library of ~800 endocrine disrupting chemicals. The work has established a new species-agnostic, high-throughput screening assay that can be used to study chemical-ligand interactions across ecologically relevant species (birds, fish, mammals) that have previously not been easily studied.
Results – Phase #1 (Primary Screen). We conducted a series of in vitro screening assays assessing 4 neurotransmitters receptors and 3 enzymes. The selected neurochemical receptors and enzymes were isolated from different organisms (n = 20) of multiple taxa, including freshwater and marine fish, birds, terrestrial and marine mammals, and biomedical species including human. The isolated neurochemical receptors and enzymes were dosed in vitro with a diverse set of 80 potentially neurotoxic chemicals (such as metals, rare-earth elements, pesticides, personal care products, flame-retardants, at 50 uM), and effluent extracts. Samples were assayed in triplicate and pooled control samples were used to monitor variability between plates. For receptor binding assays, the non-specific binding was determined by incubating samples with an unlabeled specific ligand and averaged 37%. For the enzymes assays, specific enzyme inhibitors were used to monitor the specificity of the observed reaction.Primary screen results indicate that high-quality data was collected across multiple organisms not easily studied in the lab, or in the field, with consistent ‘hits’ found across species (such as bisphenol A, Hg, B(a)P). We generated important data on several toxicant classes (including real-world mixtures) that are of ecological relevance. This work resulted in the establishment of a mid-throughput screening assay (up to 10,000 data points per week) that can be used to predict neurochemical effects across multiple ecologically relevant species (birds, fish and mammals).
Overall, 95% of the enzymes and receptors isolated from the 20 species were impacted by at least one chemical. On average, each chemical resulted in a significant decrease of receptor binding or enzyme activity in 32% of the species, with the lowest impacts measured on the GABA BZ receptor (18%), and the highest effects measured on the Glutamine synthetase activity (64%). From these results it was possible to shortlist some chemicals that showed the most harmful effects. For instance methyl-mercury, mercury, prochloraz, and benzo(a)pyrene showed significant effects on enzymes and receptors in 55 to 65% of the species. There was no clear sensitivity pattern among species with no species being more sensitive than another.
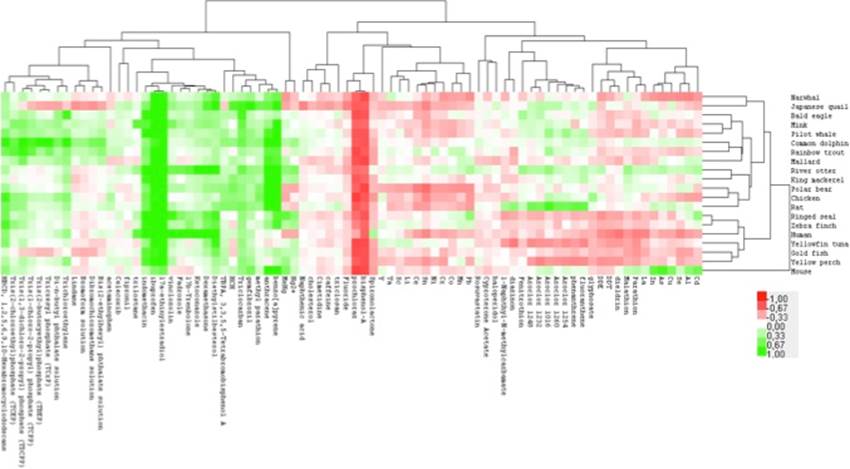
An example of results is presented in Figure 1.
Figure 1. Hierarchical cluster of all species and all chemicals tested for NMDA (receptor binding relative to control (100%); -1 = 0%, 0 =100%, 1 = 200%).
Future Activities:
Conduct secondary screening assays on selected chemicals and develop computational models.
Journal Articles:
No journal articles submitted with this report: View all 19 publications for this projectSupplemental Keywords:
Alternative testing, screening, in vitro, chemical testing, chemical prioritization, wildlife, cell-free assays, high-throughput, computational models;Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.