Grantee Research Project Results
2021 Progress Report: Urban Food Lab: Composting Plastic in Aquaponics
EPA Grant Number: SU840144Title: Urban Food Lab: Composting Plastic in Aquaponics
Investigators: Silverman, Andrea , Gowayed, Omar , Acklin, Josh , Moosa, Tahany , Moratos, Angelica , Sookchan, Savannah , White, Gianna , Charytan, Natan , Ryu, Chaehyun , Gupta, Yams
Institution: New York University , Mount Sinai School of Medicine
EPA Project Officer: Spatz, Kyle
Phase: I
Project Period: December 1, 2020 through November 30, 2021 (Extended to November 30, 2022)
Project Period Covered by this Report: December 1, 2020 through November 30,2021
Project Amount: $24,972
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2020) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Sustainable and Healthy Communities
Objective:
Three objectives were outlined in the project proposal: (1) build fungi-, bacteria-, and superworm-based composting systems to compost polyurethane, polyethylene terephthalate, and polystyrene foam, respectively, mixed with food waste, and monitor the relative decomposition of plastics within the these systems; (2) conduct a systematic evaluation to determine the toxicity of the resulting compost products, through germination tests and an evaluation of kale growth within an aquaponic farm system; and (3) evaluate aqueous compost solutions and plant matter grown from these solutions for the presence of microplastics, using spectroscopic methods [e.g., Fourier-transform infrared spectroscopy (FTIR)].
Progress Summary:
Our investigation into the toxicity of plastic degraded with biological specimens such as superworms, or Zophobas morio (a cousin of the mealworm, Tenebrio molitor), fungi, and PETase genetically modified bacteria has been challenging but fruitful. Due to the COVID-19 pandemic, we had to reduce the scale of our proposed research to accommodate for social distancing requirements. Therefore, we elected to investigate the role of superworms in the degradation of polystyrene foam ("Styrofoam") and PETase genetically modified bacteria degradation of Polyethylene terephthalate (PET). We opted to not continue the fungi portion of the investigation due to personnel changes and pandemic-related challenges.
Styrofoam Degradation Experiments
We iterated our experimental design several times to create an experimental set up and enclosure for the superworms that limits escape, mold, and mass death of the superworms; each iteration of the design was tested and lessons learned were used for the design iteration. An additional goal of the design is to separate the frass (superworm waste) generated from their feed which consisted of a combination of oats, kale, or PS foam. To accomplish this goal, three superworm bins were built to foster three frass producing conditions based on the superworm food sources: Oat+Kale (food frass), Oats+Kale+PS foam (plastic + food frass), and PS foam
(plastic frass ). Frass is collected by separation using a 100 µm mesh. While this methodology is able to separate frass from larger pieces of food waste and plastic, small pieces of PS foam were also able to pass through the 100 µm mesh. This presented a challenge given that we observed during experimentation conducted thus far that the superworms' eating habit entails ripping apart their food into micron sized pieces. This eating behavior has not been previously reported in studies conducted on analogous, mealworm-based, PS foam degradation systems. Due to this style of eating, the resultant frass contained a portion of undigested PS that is difficult to segregate in a scalable system. This inability to separate undigested, micron-sized, PS foam indicates a hindrance to the goal of this project which is to determine the feasibility of scalable utilization of vermi-degraded/vermi-composted plastics as a nutrient source for closed aquaponic systems.
Analysis of Degraded and Vermi-Composted Styrofoam
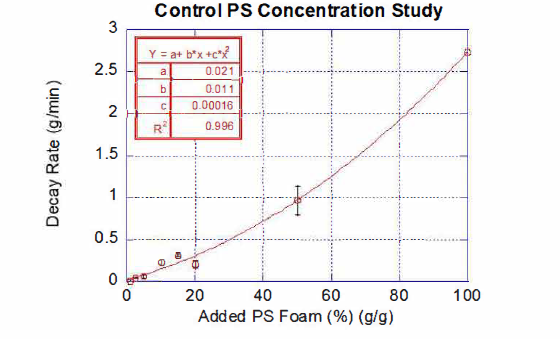
During the first year of the project, we conducted method development to spectroscopically characterize nano/micro-plastics and compost with the use of Si-wafer based Total Internal Reflection Fourier transform infrared (FTIR) spectroscopy techniques. While this methodology was successful in the detection of 50 nm diameter PS nanoplastics; we were unable to resolve plastic composition in mixed systems. We therefore pivoted the methods employed, and explored the use of thermogravimetric analysis (TGA) to measure the percentage of plastics within collected vermi-compost samples. By using known values of polystyrene foam added to samples of vermi-composted oats and kale to create a standard curve for TGA analysis, the concentration ofundegraded/undigested polystyrene foam in vermi-composted oats, kale, and PS foam was interpolated to be 21 % of the resultant compost.
Figure 1: Thermogravimetric analysis (TGA) of added PS foam to vermi-composted oats and
kale compost. TGA continuously measures the change in mass of a sample as it is heated
overtime. The decay rate (g/min) observes the change in mass of PS foam at a characteristic
temperature above 300 °C. Increased decay rates correspond to larger PS concentrations. This
plot shows the correlation between increased controlled PS foam additions (gig) to non-plastic
frass and their corresponding maximum decay rates. The data fits, with a variance (R2) of 0. 996,
to the polynomial curve: y= 0. 021 +0. 011 *x+0. 00016*x2 where y is the decay rate and xis the %
of PS added to fras
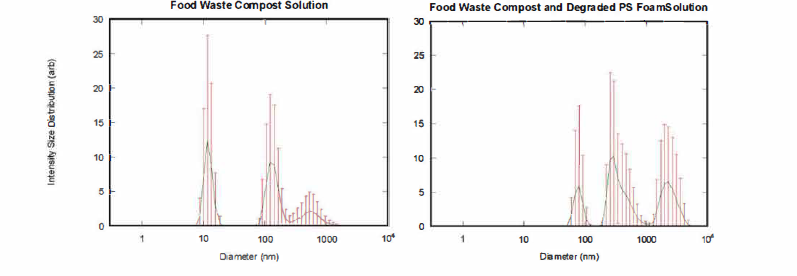
Figure 2: Dynamic light scattering results of Left: aqueous solution of foodfrass
(vermi-composted oats and kale) (peaks: 10 nm, 200 nm, 600 nm). Right: aqueous solution of
food+plastic frass (vermi-composted oats, kale, and PS foam) (peaks: 80 nm, 400 nm, 3000 nm).
We plan to corroborate this preliminary size distribution data with SEM and, if necessary, TEM images in the next few months to determine the relative sizes of plastic (undegraded and undigested) to non-plastic organic material.
Toxicity evaluation of Superworm Degraded PS Foam
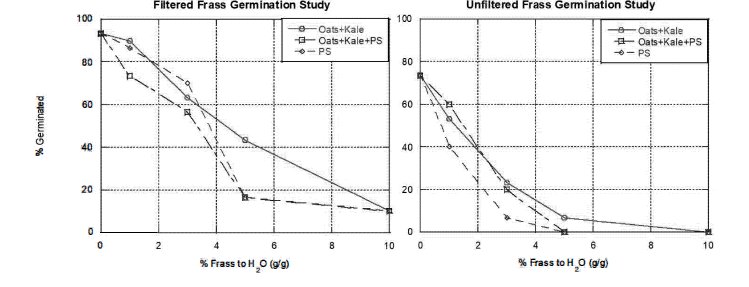
Germination studies are being conducted to determine the toxicity of the resultant vermi-composted materials. To do so, 30 tomato seeds are placed in a petri dish with aqueous solutions made with vermi-composted materials at various solution concentrations (0-10% frass/tap water). The petri dishes are exposed to indirect, full spectrum grow lights at a temperature of 25°C for a period of 5 days. After this, the samples are frozen to cease further growth. Germination rates are measured by counting the number of seeds with any form of sprouting. To evaluate the toxicity of the food, plastic, and food+plastic frass, three aqueous solutions were evaluated: a negative control solution of food frass and tap water, a solution of food+plastic frass and tap water, and a positive control solution of plastic frass and tap water; for each solution we evaluated both a 1-µm filtered solution and an unfiltered solution. Tap water was selected as a solvent due to its ease of access and scalability. As seen in Figure 3, preliminary studies indicate that larger concentrations ofundegraded and vermi-composted PS foam show an increase in toxicity relative to the plastic-free vermi-composted solution. It also shows that filtration enhances overall germination rates even in the tap water control (0% frass to H 0 on both plots in Figure 3). Seeing that this is preliminary data (N=l), further experimentation is needed to statistically analyze the data and determine the differences between filtered and unfiltered frass solutions with and without degraded plastics.
Figure 3: Right: Filtered (1 μm) germination study of 30 tomato seeds. Left: unfiltered (1 μm)
germination study of 30 tomato seeds. Tap water was used for the 0% .frass to H20 sample. Food
.frass (Oats+ Kale) (Red markers), food+plastic .frass (Oats+ Kale+ PS foam) (Blue markers),
plastic .frass (PS foam) (Green markers) .
Finally, due to the reduced scale of our food waste and plastic degradation systems, we were not able to produce enough material to conduct the planned growth toxicity tests in which we would assess growth of kale plants in aqueous compost materials after germination. This allowed us to focus on and more robustly develop a more established seed germination-based toxicity assays that are still ongoing.
PET Degradation Experimentation
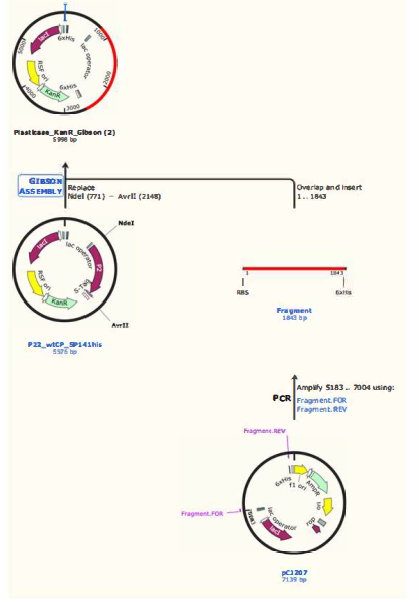
We've successfully cloned the MHETase gene into a KanR expressing cassette. First, the MHETase coding region was PCR amplified from the MHETase plasmid. The amplicon was then digested with the restriction enzymes NDEL and AVRIi to prepare for insertion into a plasmid expressing KanR. In parallel, NDEL and AVRIi were used to digest the KanR donor plasmid and generate ends overlapping our insert fragment. Finally, Gibson Assembly was used to reconstruct the KanR plasmid with MHETase. Individual colonies were rescued from kanamycin treated agar plates, indicating successful insertion into the KanR cassette.
Resultant colonies were sequence validated for correct insertion of the MHETase coding region. Sequence alignment revealed a perfect sequence identity with the sequence of MHETase. We are now in the process of optimizing the expression of our newly constructed MHETase plasmid with our PETase plasmid. MHETase expression will be optimized by expressing varying plasmid concentrations in a minimal media system to promote PET as the sole carbon source. Subsequent experimentation will be done to determine the optimal culture conditions for the biodegradation of PET utilizing this method. Once we have optimized an expression system for MHETase and PETase, we will conduct the proposed toxicity tests and seed-based toxicity assays to assess the impact of our PET compost on germination. Finally, we will utilize mass spectrometry to determine the composition of our compost, identifying any microplastics formation in the process of enzymatic degradation.
PET biodegradation studies were delayed by the insufficient commercial supply of the MHETase plasmid (pCJ207). As an alternative, we decided to generate the proposed plasmid construct ourselves via molecular cloning. Optimization of methods to evaluate PET concentrations in this system is ongoing. TGA analysis for microplastic detection and germination studies will follow.
Thus far we have done the groundwork to establish controlled experimental design and methods needed to accomplish proposed
objectives. Specifically, we have optimized the experimental design to evaluate two plastic degradation systems: GMO PETase bacteria and superworm composting bins. We also developed spectroscopic and thermal analysis methods such as DLS and TGA, that we will continue to use to evaluate the potential size distribution and concentration of microplastics in superworm degraded PS foam. We have begun evaluating the toxicity of filtered and unfiltered aqueous frass generated by superworms fed plastic and nonplastic nutrients. Preliminary results indicate the toxicity of vermi-composted PS foam as well as an overall reduced toxicity from filtering aqueous compost solutions with a micron filter.
Figure 4: PETase Cloning Diagram
Future Activities:
SEM imaging of food+plastic frass to assess size of PS particles. Reproduction of germination studies of food frass, plastic frass, and food+plastic frass with and without filtration. TGA analysis ofvermi-composted oats, kale, and PS frass with and without (0.45, 0.75, 1.0-µm) filtration to determine nanoplastic concentration. TGA and germination studies of bio-degraded PET. Professional videography of project and team for promotional presentation. Evaluate the ability of bacteria containing the PETase and MHETase plasmids to degrade PET. Development of new plasmids to metabolize ethylene glycol produced from MHETase reaction. Analyze microbially degraded PET with TGA, DLS, and FTIR.
Journal Articles:
No journal articles submitted with this report: View all 1 publications for this projectSupplemental Keywords:
Microplastics, compost, toxicity, sustainable development, clean technologies, waste reduction, waste minimization, bioremediation, community-based, aquaponic agricultureProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.