Grantee Research Project Results
2019 Progress Report: Three- Step Scrubber for Ammonia Removal
EPA Grant Number: SV839356Title: Three- Step Scrubber for Ammonia Removal
Investigators: Barsanti, Kelley , Harake, Maggy , Milne, Joshua , Paul, Phartho , Armstrong, Maurice , Stamatis, Christos
Current Investigators: Rupiper, Amanda , Barsanti, Kelley , Limon, Gwyndolin , Flores, Alexis , Sanchez-Diaz, Nancy , Iqbal, Usman , Bang, Eric , Orozco, Omar , Chan, Dylon , Martinez, Dianna-Kristina
Institution: University of California - Riverside
EPA Project Officer: Page, Angela
Phase: II
Project Period: March 1, 2018 through February 29, 2020 (Extended to February 28, 2025)
Project Period Covered by this Report: March 1, 2019 through February 29,2020
Project Amount: $40,240
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet - Phase 2 (2017) Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources , Sustainable and Healthy Communities
Objective:
Agricultural producers are primary contributors to atmospheric ammonia (NH3) pollution throughout the US. Although NH3 is not a hazardous air pollutant regulated at the federal level, or a state-identified toxic air contaminant, its acute and chronic non-cancer health effects justify limiting its concentration in the atmosphere. Specifically, NH3 reacts with other constituents in the air to produce fine particulate matter (PM2.5), which is a federally-regulated criteria pollutant, and causes adverse health effects. Current conventional chemical scrubbers for NH3 do not represent a sustainable solution, as they have high economic and environmental costs. Therefore, we have designed a three-step NH3 scrubbing and filtration system for agricultural NH3 emissions that promotes sustainability by reusing water, recycling waste, and reducing costs.
Reducing the release of NH3 to the atmosphere contributes to improved air quality for individuals in close proximity to agricultural operations. The use of the collected NH3 as an amendment for soil, rather than releasing it in waste streams as ammonium sulfate (a salt) improves water quality, which has a benefit for people and the planet. The ability to recycle water leads to reduced waste, providing an environmental benefit; in addition, this reduces costs, providing an economic benefit and contributing to prosperity. It is in these ways that the design project addresses people, prosperity, and the planet.
Each academic year, a team of four undergraduate students in Chemical & Environmental Engineering lead the project design, implementation, evaluation, and iteration. Team members have included typically underrepresented minorities in STEM and first-generation college students. This often represents their first research experience. The students work closely with a graduate student or postdoctoral researcher in the PI’s group, which provides teaching and mentoring opportunities for these graduate students and postdocs. Finally, the students participate in at least one community outreach event, in which they share their project and findings with the general public.
Progress Summary:
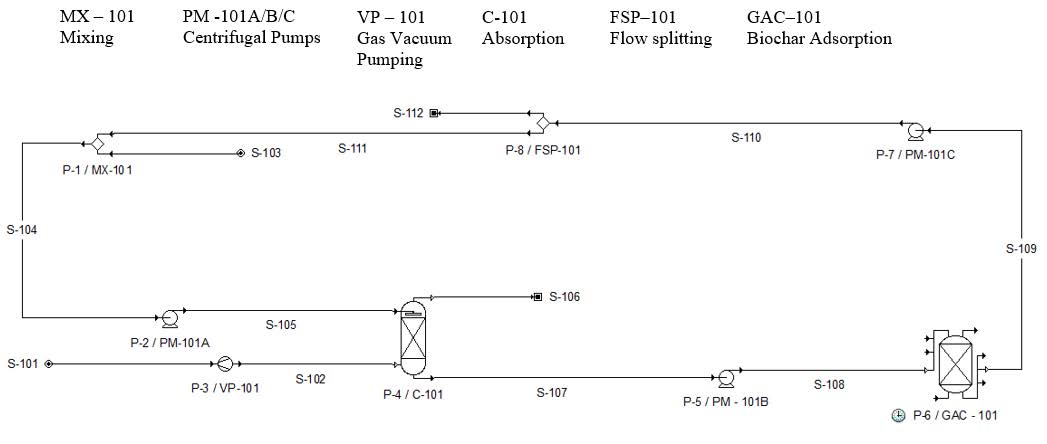
The primary objective of the P3 Phase II award was to build and test a field-scale prototype of the three-step NH3 scrubber. At the completion of the Phase I award, a laboratory-scale prototype of the second step, the adsorption column, was built and tested. In year one of the Phase II award, the team focused on building and testing steps one and two of the field-scale prototype under laboratory conditions. The team also tested the efficacy of the ammonia-enriched biochar as a soil amendment. In year two of the Phase II award, the team focused on moving the field-scale prototype from laboratory to the field, and on optimization and feasibility under field conditions. The revised skeleton process flow diagram is shown in Table. 1 and the associated stream tables in Table 2.
Figure 1. Process flow diagram (PFD) for ammonia removal system
| Time Ref: h | S-101 | S-102 | S-103 | S-104 | S-105 | S-106 | S-107 | S-108 | S-109 | S-110 | S-111 | S-112 | |
| Type | Raw Materials | Raw Materials | |||||||||||
| Total Mass flow | kg | 482.9000 | 482.9000 | 3500.0000 | 13984.9157 | 13984.9157 | 478.0020 | 13989.8139 | 13988.4820 | 13979.8876 | 13979.8876 | 10484.9157 | 3494.9719 |
| Temperature | c | 25.0 | 25.0 | 25.0 | 25.1 | 25.1 | 25.1 | 25.1 | 25.1 | 25.1 | 25.1 | 25.1 | 25.1 |
| Pressure | bar | 1.000 | 1.013 | 9.000 | 9.000 | 10.000 | 1.013 | 10.000 | 11.000 | 11.000 | 12.000 | 12.000 | 12.000 |
| Liq/Sol Vol Flow | L | 0.0000 | 0.0000 | 3518.6335 | 14056.2439 | 14056.3001 | 0.0000 | 14051.3753 | 14050.0923 | 14050.0923 | 14050.1485 | 10537.6114 | 3512.5371 |
| Total Contents | kg | 482.9000 | 482.9000 | 3500.0000 | 13984.9157 | 13984.9157 | 478.0020 | 13989.8139 | 13988.4820 | 13979.8876 | 13979.8876 | 10484.9157 | 3494.9719 |
| Ammonia | 5.4800 | 5.4800 | 0.0000 | 0.0000 | 0.0000 | 0.5820 | 0.2449 | 0.2449 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | |
| NH4OH | 0.0000 | 0.0000 | 0.0000 | 3.6678 | 3.6678 | 0.0000 | 13.2534 | 13.2532 | 4.9037 | 4.9037 | 3.6778 | 1.2259 | |
| Nitrogen | 366.2374 | 366.2374 | 0.0000 | 0.0000 | 0.0000 | 366.2374 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | |
| Oxygen | 111.1826 | 111.1826 | 0.0000 | 0.0000 | 0.0000 | 111.1826 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | |
| Saturated Air | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | |
| Water | 0.0000 | 0.0000 | 3500.0000 | 13981.2379 | 13981.2379 | 0.0000 | 13976.3157 | 13974.9839 | 13974.9839 | 13974.9839 | 10481.2379 | 3493.7460 |
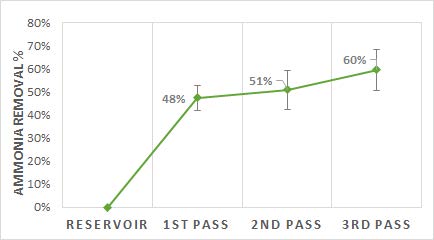
At the end of year one, the team determined that with three passes, the adsorption column achieved an average NH3/NH4 removal of 60% and a maximum removal of 78% (see Fig. 3). The flow rate under laboratory conditions was ~ 5 mL/s, with a retention time of ~150 seconds in the biochar column. The year two team concluded that the mass flow rates under laboratory conditions were too low to achieve significant reduction of NH3 emissions under field conditions. Thus the team used a mass flow rate of ~140 mL/s (500 kg/hr) as the starting point for SuperPro modeling simulations. The team focused on optimizing operating temperatures and pressures, flow rates, and mass:volume ratios in each of the reactors using SuperPro. The team found that for the base modeling conditions, the first step (absorption into a water stream) achieved 89% efficiency and the second step (adsorption onto the biochar column) achieved 63% efficiency (similar to the laboratory results of the prior team). The current team supported the suggestion that ultimately, multiple columns in parallel may be a better design.
Figure 3. Ammonia removal percentage with three passes through
a biochar column.
The team also more thoroughly evaluated the environmental and economic costs of operating the most recent version of the design at the field scale. The team calculated the fixed capital investment (FCI) using SuperPro and Capcost. The FCI included all processing equipment except for the flow splitter and the mixer as these may not even be needed in the actual build. The working capital chosen for the startup period was 20% of the FCI and the combined total capital investment (TCI) was $103,460. The assumed construction period was two years with 60% of the TCI in year one and the remainder in year two. Straight line depreciation over a period of 6 years was assumed. Since the NH3/NH4-saturated biochar can be used as a fertilizer, this was considered direct revenue. The price point for the saturated biochar product was assumed to be $2000/ton, with a production rate of 0.09 tons per year. The offset in emissions fines based on state and federal emissions standards was also considered revenue. The estimate year one team determined that at 53% reduction in emissions would results in a savings of $1504; this savings rate was used by the year two team in cost evaluation.
Future Activities:
The year two team concluded that overall, implementation of the three-phase NH3 scrubber is not economically sustainable, based on the current design and current costs. However, the team did not include the cost of water or water mitigation, should that become a factor in the future. The team did conclude that use of the three-phase NH3 on dairy farms and perhaps other agricultural operations would minimize NH3 emission from these hot spots, thus reducing haze, odor, eutrophication, and improving human and environmental health near. The team recommended getting more accurate measurements of NH3 emissions as a function of facility type and size, evaluating how to include costs of water and water mitigation, and identifying opportunities for technological improvements (equipment and/or design) that would decrease costs.
Journal Articles:
No journal articles submitted with this report: View all 3 publications for this projectSupplemental Keywords:
ammonia, agricultural biproducts, water conservation, emission control technologies, reuseProgress and Final Reports:
Original AbstractP3 Phase I:
Three-Phase Ammonia Air Scrubber Recycles Water | 2017 Progress Report | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final
- 2023 Progress Report
- 2022 Progress Report
- 2021 Progress Report
- 2020 Progress Report
- 2018 Progress Report
- Original Abstract
- P3 Phase I | 2017 Progress Report | Final Report